Laboratory Exercise 2
advertisement

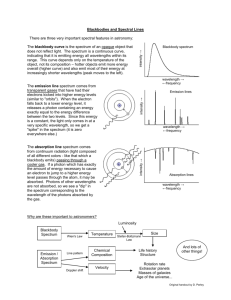
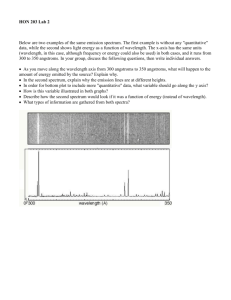
Laboratory Exercise 2 In this laboratory exercise students will examine the spectrum of various light sources and the action of various filters using spectral glasses and an Ocean Optics Red Tide Spectrometer. They will identify the atoms or molecules of an unlabeled hot gas using the measured emission spectrum. The spectrometer: The Ocean Optics Red Tide Spectrometer uses a diffraction grating to deflect light with different wavelengths through different angles. Light enters a fiber optic probe through a tiny hole at one end. It travels down the fiber optic cable into the spectrometer. The grating deflects light with different wavelengths onto different positions of a position-sensitive detector. The output of the detector is passed to a computer via a USB cable. The Ocean Optics SpectraSuite software provides real-time readout of the measured spectrum on the computer screen. Equipment: Incandescent lamp Mercury Lamp Gas Discharge Lamp Laser LEDs Filters Spectral Glasses Procedure: Look at each light source through the spectral glasses and measure its spectrum with the spectrometer. Make notes of your observations. Examine the spectrum of the incandescent light source with and without filters. What wavelength range does this spectrum cover? What wavelength range is absorbed by the yellow filter? What wavelength range is absorbed by the green filter? What wavelength range is absorbed by the "pink" goggles? They protect your eyes from which type of laser light? What wavelength range is absorbed by the "blue-green" goggles? They protect your eyes from which type of laser light? Examine the spectrum of the Mercury Lamp. What are the wavelengths of the three most prominent lines? If you click on a line the software displays the wavelength in nanometer (nm) in a box. Approximately how wide are those lines? Point the fiber optics probe towards the room lights. Do you observe discrete lines on top of the continuous spectrum? Can you identify the source of those lines? Examine the spectrum of the gas discharge lamp with the hydrogen tube. What are the wavelengths of the three prominent lines? Examine the spectrum of the gas discharge lamp with the tube containing an unlabeled gas. What are the wavelengths of some of the most prominent lines? Can you identify the gas using the attached tables? Examine the spectrum of the laser. What is the wavelength of this laser? What is the approximate width of the line? Examine the spectra of the four LEDs. (The longer leg has to be connected to the positive side (red wire) of the power supply.) Describe these spectra? Laboratory 2 Report: Name: E-mail address: In a few words, describe the experiment. Comment on the procedure. Did you encounter difficulties or surprises? Present your observations.