3.1.8 POLICIES POLICY
advertisement

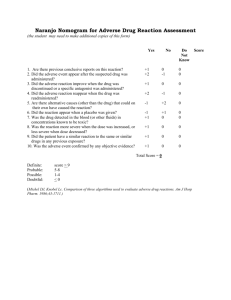
Peninsula Health Human Research Ethics Committee P O L I C Y ADVERSE EVENT REPORTING 1. INTRODUCTION Investigators in clinical trials report serious adverse events or reactions to Human Research and Ethics Committees and to trial sponsors to meet the requirements of regulatory agencies such as the Therapeutic Goods Administration. Sponsors of clinical trials report serious adverse events or serious unexpected suspected adverse reactions to regulatory agencies to meet the conditions of approval and to investigators to enable them to fulfil the conditions of an ethical approval by a Human Research and Ethics Committee (HREC). The responsibility of Human Research and Ethics Committees and the institutions they advise is to protect the safety of participants in clinical trials and in order to do this they need to receive sufficient reliable information about the implications of adverse events or reactions. The National Statement on Ethical Conduct in Human Research authorises institutions to establish mechanisms for reporting and reviewing serious adverse events, serious adverse drug reactions and serious unexpected suspected adverse drug reactions, as a condition of ethical approval of clinical trials. HRECs must ensure that the frequency and timing of safety reporting reflects the degree of risk to participants in the clinical trial. 2. PURPOSE The purpose of this policy is to clarify the responsibilities of investigators in relation to the reporting of serious adverse events and serious adverse reactions occurring in clinical trials at Peninsula Health. 3. DEFINITIONS Adverse Drug Reaction Any noxious and unintended response to an unapproved medicinal product, related to any dose. The phrase “response to an unapproved medicinal product” means that a causal relationship between the product and an adverse event is at least a reasonable possibility, ie the relationship cannot be ruled out. (Unapproved medicinal product here includes approved products used at levels or in ways that are unapproved). OR A noxious and unintended response to a drug that occurs at doses of marketed medical products normally used in humans for prophylaxis, diagnosis or therapy of diseases or for modification of physiological function. Serious Adverse Event Any untoward medical occurrence that: results in death is life-threatening requires inpatient hospitalisation or prolongation of existing hospitalisation results in persistent or significant disability/incapacity is a congenital anomaly/birth defect or is a medically important event or reaction. Suspected Unexpected Serious Adverse Reaction (SUSAR) A Serious Adverse Event for which there is some degree of probability that the event is an adverse reaction to the administered drug, and the adverse reaction is unexpected. Date Created / Revised Next Revision Executive Sponsor Co ordinator Approved by March 2008 Dr Sara Watson HREC 2 April 2008 Page 1 of 3 Peninsula Health Human Research Ethics Committee P O L I C Y ADVERSE EVENT REPORTING Unexpected Adverse Drug Reaction An adverse reaction, the nature or severity of which is not consistent with the applicable scientific information (eg Investigator’s Brochure for an unapproved investigational product or Product Information (PI) document or similar for an approved, marketed product). 4. LEGISLATION Regulations made under the Therapeutic Goods Act 1989 require that clinical trials of unregistered therapeutic goods are to conform with the requirements of the approving HREC and the National Statement on Ethical Conduct in Human Research (NHMRc 2007) (the National Statement). In turn, the National Statement requires HRECs to be satisfied that trials conform, where relevant to CPMP/ICH Note for Guidance on Good Clinical Practice (CPMP/ICH-135/95) as adopted by TGA (EU GCP guideline). Further, in relations to reporting of adverse events, CPMP/ICH-135/95 refers to Note for guidance on clinical safety data management: definitions and standards for expediting reporting (CPMP/ICH-377/95). 5. POLICY The Peninsula Health HREC in consultation with the Peninsula Health Drugs and Therapeutics Committee has agreed on the following policy; Serious Adverse Events involving participants at Peninsula Health (Internal Events) must be reported to the Research Program secretariat within 24 hours of occurrence. Expected Adverse Reactions including Serious Adverse Events (SAE’s) must be reported at least six-monthly or more frequently if so stated in the individual study protocol. The annual summary is therefore not required. Suspected Unexpected Serious Adverse Reactions (SUSARs) occurring in Australia must be reported as per the TGAs requirements – within 15 calendar days of first knowledge; or for fatal or life-threatening events, an initial report within 7 days and a follow-up report if required within 15 calendar days. Comments regarding action taken/planned are also required. Suspected Unexpected Serious Adverse Reactions (SUSARs) occurring in Australia and overseas must be provided quarterly as a listing with comments regarding action taken/planned. Significant New Information about a trial must be provided to the HREC in a timely manner. Such information may arise from monitoring or analysis of product safety information overseas based on individual safety reports or reports from Data Safety Monitoring Committees. It is not necessary, nor is it sufficient to provide individual safety reports without contextual information. 6. RESPONSIBILITIES Date Created / Revised Next Revision Executive Sponsor Co ordinator Approved by March 2008 Dr Sara Watson HREC 2 April 2008 Page 2 of 3 Peninsula Health Human Research Ethics Committee P O L I C Y ADVERSE EVENT REPORTING 6.1. HREC is responsible for reviewing individual reports and listings and making recommendations to investigators for further action where appropriate. 6.2 Investigators/Researchers are responsible for reporting adverse events as described in Section 5 and responding to HREC recommendations promptly. Individual reports must be submitted on the Peninsula Health Research Adverse Event Report Form. 7. EVALUATION Monitoring of compliance with this policy will be undertaken by the Research Program Secretariat through monitoring of Serious Adverse Events (SAEs) and Serious Unexpected Serious Adverse Reactions (SUSARs) reports from investigators. 8. REFERENCES NHMRC HREC Alert No 1 18 April 2007 National Statement on Ethical Conduct in Human Research (NHMRC 2007) Date Created / Revised Next Revision Executive Sponsor Co ordinator Approved by March 2008 Dr Sara Watson HREC 2 April 2008 Page 3 of 3