Topic 9 - Reactions of acids
advertisement

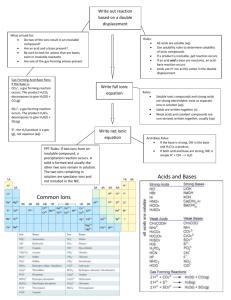
Topic 9 - Reactions of acids What Type of Substance Can Neutralise an Acid ? There are four categories of substance which react with acids and bring their pH towards 7. We will call these substances neutralisers. They can be recalled in an exam by remembering the letters CAMO !!! C A M O stands for metal or ammonium stands for stands for reactive stands for metal or ammonium Carbonate Alkali Metal Oxide Be careful - not all metals react, only those which we consider to be reactive (i.e. above Hydrogen on the electrochemical series) Everyday examples of Neutralisation Farmers add lime, calcium oxide, to soil help neutralise the soil as most plants and vegetables grow best in neutral or slightly alkaline soil. The lime can neutralise acidity of soil caused by acid rain. Lime is also used to neutralise lakes made acidic by acid rain. Milk of magnesia, magnesium oxide/hydroxide, is used to neutralise excess stomach acid. Dental Care - bacteria react with the carbohydrates in the mouth to form acids capable of dissolving the enamel, causing tooth decay. Toothpaste should be alkali to neutralise the acid produced by bacteria which helps to decay teeth. Insect Stings - A wasp sting is alkaline and a weak acid such as vinegar can be rubbed onto it to alleviate pain. In contrast the sting of a bee is acidic and an alkali such as bicarbonate of soda can be used to treat it. Effect of neutralisation on pH The pH of acids rise toward 7 when being neutralised, the pH of alkalis fall towards 7 when being neutralised. Neutralisation moves the pH of a solution closer to 7 (neutral). Base versus Alkali We have looked at four substances which will neutralise an acid ie. CAMO substances. Each of these react with an acid by accepting the H+ ion from the acid. Every substance which reacts with an acid in this way is called a base. So a base by definition is a hydrogen ion acceptor. Bases can be soluble or insoluble in water. Alkalis are those bases which are soluble in water. This can be visualised using the diagram below : Bases insoluble alkalis soluble Making a Salt If we remove the hydrogen from each acid and replace it with a metal or ammonium ion from one of the 4 neutralisers in the previous page this would form a salt. The last part in the name of the salt formed would be determined by the acid used in the reaction. Each acid gives rise to a family of salts Acid Hydrochloric Sulphuric Nitric e.g. Salt Chloride Sulphate Nitrate Hydrochloric acid + Potassium hydroxide Potassium chloride + water sulphuric acid + sodium hydroxide sodium sulphate + water Ammonia + nitric acid ammonium nitrate + water There are many different methods used for making a salt but the method you use depends on two factors: the solubility of the base used and the solubility of the salt to be made. The following table summarises information found on page 5 of your data booklet regarding the solubility of various compounds which you may find useful: compound metal compound soluble in water metal compound insoluble in water hydroxide sodium, potassium, ammonium, calcium all others oxide sodium, potassium, calcium all others carbonate sodium, potassium, ammonium all others nitrate all possible none sulphate all others lead, barium chloride all others silver, lead If the new salt is insoluble a precipitate is obtained. Salts by Precipitation Insoluble salts are best made by precipitation, in this method two soluble salts one containing the positive ion and the other containing the negative ion are mixed together, an insoluble precipitate containing the desired salt is formed. e.g. barium sulphate barium nitrate + sodium sulphate barium sulphate (precipitate) + sodium nitrate Writing Ionic Equations These equations are carried out in the same way as before. Assuming that you will want to go through all of the various steps then they are outlined below : Write a word equation from an account of the reaction. Turn your word equation into a formula equation. Put in state symbols. (these are important in this case) Balance your equation. Turn it into an ionic equation by putting in charges on any ions which exist. Separate ions which are free to move away from each other. Example 1 Write an ionic equation for the reaction between calcium hydroxide and nitric acid. Step 1 Word Equation calcium hydroxide + nitric acid calcium nitrate + water Step 2 Formula Equation Ca (OH)2 + HNO3 Ca ( NO3 )2 + H2O Step 3 Put in state symbols ( look them up in the solubility table on page 5 of your data booklet) Ca (OH)2 (aq) + HNO3(aq) Ca ( NO3 )2 (aq) + H2O(l) Step 4 Balance the Equation (see rules in topic 5) Ca (OH)2 (aq) + 2HNO3(aq) Ca ( NO3 )2 Step 5 Put in ionic charges Ca 2+ (OH-)2 (aq) + 2H+ NO3-(aq) Ca Step 6 Separate ions which are free to move Ca 2+(aq) + 2OH- (aq) + 2H+(aq) + 2NO3-(aq) 2+ (aq) 2H2O(l) + ( NO3 -)2 Ca 2+ (aq) (aq) 2H2O(l) + + 2NO3 -(aq) + 2H2O(l) If we examine the final ionic equation above we notice that there are two ions which appear as both reactants and products, namely Ca 2+(aq) and NO3 -(aq) . They appear to be doing nothing to take part in the reaction. For this reason we call ions which appear on both sides of an equation and in the same state spectator ions. We can rewrite the equation with spectator ions omitted as follows: Ca 2+ (aq) + 2OH- (aq) + 2H+(aq) i.e. 2OH- (aq) + 2NO3-(aq) Ca + 2H+(aq) 2+ (aq) + 2NO3 -(aq) + 2H2O(l) 2H2O(l) Without including the spectator ions, the reaction appears to be simply the reaction between H+ and OH- ions to form H2O. This is true for all neutralisation reactions between acids and alkalis. Example 2 Write a balanced ionic equation, omitting spectator ions, for the reaction between aluminium carbonate and hydrochloric acid. Jumping straight to Step 4 Al2(CO3)3 (s) + HCl Step 5 (aq) Balance the equation Al2(CO3)3 (s) + 6HCl (aq) AlCl3 (aq) + CO 2AlCl3 (aq) + 3CO 2(g) + 2(g) + H2O(l) 3H2O(l) Step 6 Put in Ionic Charges (Al 3+ )2(CO32-)3 (s) + 6H+ Cl- (aq) Step 7 Separate ions which are free to move (Al 3+ )2(CO32-)3 (s) + 6H+ (aq) + 6Cl- (aq) Step 8 Score out spectator ions (Al 3+ )2(CO32-)3 (s) + 6H+ (aq) + 6ClStep 9 (aq) 2Al 3+ (Cl-)3 (aq) + 3CO 2(g) + 3H2O(l) 2Al 3+(aq) + 6Cl- (aq) + 3CO 2(g) + 3H2O(l) 2Al 3+(aq) + 6Cl- (aq) + 3CO 2(g) + 3H2O(l) Rewrite the equation without the spectator ions (Al 3+ )2(CO32-)3 (s) + 6H+ (aq) 2Al 3+(aq) + 3CO 2(g) + 3H2O(l) Volumetric Analysis Using volumetric analysis the molarity or the volume of an alkali can be determined if the molarity and volume of the acid are known along with either the molarity or volume of the alkali. The results of a titration are used. Acid Rain For causes and effects of acid rain see topic 8 notes.