Percentage Yield Chemistry Worksheet
advertisement

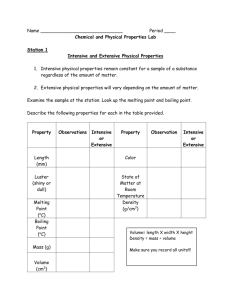
Name__________________________________Period_______Date_________ Percentage Yield Directions: Answer each of the following questions. You will need to use your periodic table. Example calculations are in section 9.3 of your textbook. 1. Methyl alcohol, CH3OH, is used as a fuel in alcohol-burning race cars. It can be manufactured according to the following reaction: 2H2(g) + CO(g) → CH3OH(l) When 34.5 g of hydrogen gas reacts with excess carbon monoxide gas, the actual yield of methyl alcohol is 252.2 g. What is the percentage yield of methyl alcohol? 2. Milk of magnesia is used as an antacid. It is an aqueous suspension of magnesium hydroxide, a mild base. It acts in the stomach according to the following reaction: Mg(OH)2(s) + 2HCl(aq) → 2H2O(l) + MgCl2(aq) When 28.0 g of magnesium hydroxide react with excess hydrochloric acid solution in an experiment to simulate the action of magnesium hydroxide in the stomach, the actual yield of magnesium chloride is 35.8 g. What is the percentage yield of magnesium chloride? 3. Hydrogen cyanide gas is produced by reacting ammonia gas, oxygen gas, and methane gas according to the following reaction: 2NH3(g) + 3O2(g) + 2CH4(g) → 2HCN(g) + 6H2O(g) If the typical yield is 89.8%, what mass of hydrogen cyanide gas should be expected if 324 g of ammonia gas are reacted with excess oxygen gas and methane gas? 4. The fuel used in the booster rockets attached to the space shuttle contains a mixture of aluminum and ammonium perchlorate. The following equation represents one proposed reaction for the “burning” of this fuel: 3Al(s) + 3NH4ClO4(s) → Al2O3(s) + AlCl3(s) + 6H2O(g) + 3NO(g) If the typical yield is 83.4%, what mass of aluminum chloride should be expected if 3.50 kg of aluminum are reacted with excess ammonium perchlorate?