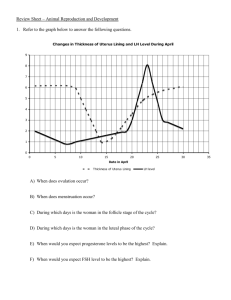
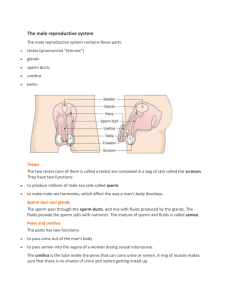
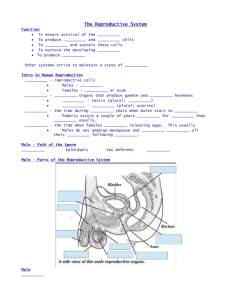
Evaluation of semen 1-Volume The graduated collection tube was
advertisement

Evaluation of semen 1-Volume The graduated collection tube was utilized to determine the volume of ejaculate to the nearest 0.1ml, just after collection. 2-Hydrogen ion concentration (pH) and color: Values of seminal pH determined by using pH paper. 3-Motility a)Mass motility A drop of fresh semen was deposited on a warm glass slide (37°C) and its motility was evaluated according to wave motion table under microscope . Table : Evaluation of mass motility. % Wave motion 0 Total immobility 10 Individual movement 20-40 Very slow movement 45-65 General wave motion ,slow amplitude of waves 70-85 Rapid wave motion ,no eddies 90-100 Rapid wave motion, eddies b) Individual motility Individual sperm motility was evaluated by depositing a few drops of diluted semen by normal saline on a warm glass slide (37°C), covered by cover slip(22mmX22mm). Evaluation was made according to progressively foreword motility as in Table under microscope . Table : Evaluation of sperm individual motility % Individual motility 0 Total immobility 10 1/5 of sperms motile 20-40 2/5 of sperms motile 45-65 3/5 of sperms motile 70-85 4/5 of sperms motile 90-100 All sperms motile 4-Dead sperms percentage: A drop of fresh semen was mixed with one drop of eosin stain then with two drops of nigrosin stain and smear made and dried. The slide was examined under microscope, uncolored sperm head(white) is considered as life sperm ,while pink head(colored) due to dead sperm . 5-Abnormal sperms percentage: Determination of abnormal sperm percentage was made using the same smear of live and dead. 6-Sperm concentration: Estimation of sperm concentration was made by adding 0.1ml of fresh semen to 19.9 ml of sperm counting solution (2.9 sodium citrate +few drops of eosin to facilitate distinguishing of sperm under microscope +Mercuric chloride (Hgcl2) as killing agent for sperm). The solution components were mixed for 3 minutes, after that using Neubauer chamber (RBCs square) sperms were counted to determine sperm concentration using the following equation: C=n X200 X 400 X 10 X1000/ 80 -n=No .of sperm in five square. -200= converse dilution rate. -80=No .of small square in five large squares. -10= converse depth between slide and cover slide. -400= converse square area of cover slide. Sperm function tests Semen analysis provides enough information to recognise sires of very low fertility, but has been increasingly considered to be a poor discriminator between moderate and high fertility levels . In order to attempt to improve the accuracy of semen assessments, a number of tests of sperm function have been employed, with varying success. The simplest of such tests incubate semen at various temperatures (typically 4 or 40°C) and, by relating the duration of sperm survival under these conditions to survival in the female genital tract, produce reasonable correlations with fertility. Other tests utilise additional measurements upon the semen, such as pH, adenosine triphosphate (ATP) content or aspartate transaminase concentration . These have been moderately successful, but have not been of sufficiently greater value than conventional semen assessment to justify their use. In medical practice, much value is placed upon the ability of sperm to penetrate cervical mucus and the behaviour of the sperm at the interface between semen and mucus . Failure of mucous penetration is frequently a sign of failure of sperm function and occurs in sperm that have been damaged by cryopreservation or in the presence of anti-sperm antibodies. Of more widespread use in veterinary practice is computer-assisted analysis of sperm swimming characteristics. In medical practice, such analyses are regarded as a useful prognostic tool in assessment of fertility because high correlations have been demonstrated between such measurements and fertility. The most important swimming characteristics are rate of forward progress, lateral movement of the sperm head and characteristics of the flagellar beat. Although the use of sperm motility analysis in veterinary practice at present is largely confined to thoroughbred stallions and AI stud bulls, it is probable that the use of such systems will rapidly increase as the cost of analysis programs decreases. Assessments of sperm viability have also been improved in recent years. Fluorescent markers that stain live, but not dead, sperm have been used and high correlations with fertility demonstrated . Assessment of the proportion of sperm with intact acrosomes has been highly correlated with fertility . The most recent innovation in assessment of sperm function has derived from the development of in vitro fertilisation (IVF) procedures, in which sperm from different sires were observed to have widely differing fertilisation success rates. Subsequently, the ability of sperm to undergo acrosome reaction in vitro was identified as a critical stage in the IVF procedure and, in the bull, tests of sperm function based upon in vitro induction of acrosome reactions have been found to have very high correlation with fertility in the field