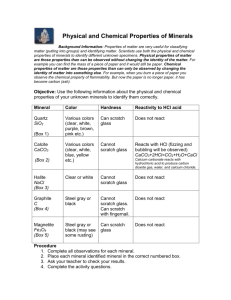
Objective: We created an in-class lab to assist students in identifying
advertisement

Objective: This lab is designed to assist students in identifying the differences between chemical and physical changes through a series of lab station experiments and follow-up questions. At each station, students are presented with a substance or process. They are asked if it represents a chemical or physical change scenario and the reasons for their answer. The third “follow-up” question is directly related to the definitions or characteristics of physical and chemical changes and is meant to assist or to confirm the students’ decisions. Follow-up questions may be used as a basis for classroom discussion or additional homework. We recommend use of this lab during the fall (mid-late October) for easy access to materials. Materials Needed for 9 Lab Stations: 1. 2. 3. 4. 5. 6. 7. Glo-sticks Burning candle (for melting wax) Changing leaves (IE, leaves which are changing color) Carved mini-pumpkin (may just be cut into pieces) Safety matches (for striking) Container of popcorn kernels and container of popped popcorn Sample of calcite (mineral) and vial of hydrochloric acid (Vinegar may be used for a safer acid.) FYI, the bubbles fizzing result from the chemical reaction between HCl and CaCO3 (Calcite) to give Carbon Dioxide gas. 8. Dissolved sugar and water 9. Alka-Seltzer and water (tablets will be dropped in by the students) Solutions to questions are as follows: 1. The snap in the stick is a chemical reaction caused by the mixing of two components. Descriptions of these materials may be found at the following sites: http://daily.stanford.org/Daily96-97/11-6-96/NEWS/NEWbug06.html http://www.extremeglow.co.uk/faq.htm#q7 2. Burning candle is a physical change. The wax is still wax; it is now in a liquid state of matter rather than a solid. 3. Leaf decomposition is a chemical change caused by chemical reactions within the leaf structure. 4. Pumpkin carving is a physical change—the pumpkin is now just in pieces; it is still of its original composition. 5. Striking a match is a chemical change caused by the reaction of chemicals on the tip of the match head instigated by the friction between the match head and the surface. 6. Popcorn popping is a physical change. It looks different but is the same thing. 7. Calcite + HCl is a chemical change. The bubbles result from the chemical reaction between HCl and CaCO3 (Calcite) to give Carbon Dioxide gas. 8. Dissolving sugar and water is a physical change. The sugar macromolecules are broken down into smaller molecules; but their form remains the same, which is shown through the descriptive taste Alka-Seltzer in water is a chemical change. The fizzing indicates a chemical change, and the substance dissolving does not maintain its original chemical structure. Questions for Lab Stations for Chemical vs. Physical Changes Lab Station 1: Halloween glo-sticks Is it a chemical or physical change? Why do you think so? When you snap the stick, what causes the change in color (the glowing)? Station 2: Melting Wax Is it a chemical or physical change? Why do you think so? Is this a reversible change? Station 3: Leaves Decomposing Is it a chemical or physical change? Why do you think so? What is causing the decomposition? (Consider how the leaf is changing colors from green to brown. Eventually, the leaf will break down into carbon, nitrogen, and other organic substances.) Station 4: Carving Pumpkin Is it a chemical or physical change? Why do you think so? By carving the pumpkin, what are you changing about the pumpkin? Station 5: Striking the match Is it a chemical or physical change? Why do you think so? What causes the flame? Station 6: Popcorn popping Is it a chemical or physical change? Why do you think so? How is the popcorn different before and after it is popped? Station 7: Calcite + HCl Is it a chemical or physical change? Why do you think so? How do you get a gas (the bubbles) from combining a solid and a liquid? Station 8: Dissolving sugar and water Is it a chemical or physical change? Why do you think so? Can you still taste the sugar after it is dissolved in water? Why is that important? Station 9: Alka-Seltzer Is it a chemical or physical change? Why do you think so? How is this different from dissolving sugar in water?