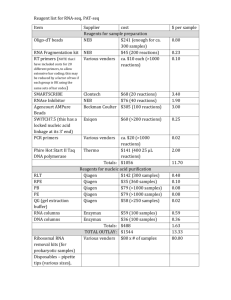
Gene expression 實驗
Suggest method and kit
RNA isolation
species or tissue
Purification of
total RNA (2nd)
total RNA
polyA-RNA
Yeast
Hot phenol protocol.
(Schmitt, et al.,1990, Nucl
Acids Res, 18:3091-3092.)
Qiagen's Oligotex
mRNA Kit
Qiagen's
RNeasy Mini
Kit
Arabidopsis
TRIzol Reagent (Invitrogen
Life Tech.)
Qiagen's Oligotex
mRNA Kit
Qiagen's
RNeasy Mini
Kit
Mammalian cells
(cultured cells and
lymphocytes)
Qiagen's RNeasy Mini Kit
Qiagen's Oligotex
Direct mRNA Kit
(directly from cell) or
Qiagen's Oligotex
mRNA Kit (from total
RNA)
N/A
Mammalian tissue
TRIzol Reagent from
Invitrogen Life Tech.
poly-A mRNA isolation
commercial kit (from
total RNA)
Qiagen's
RNeasy Mini
Kit
Albino
or polysacchoride rich
in plant
Pine tree method
N/A
N/A
(Adapted from GeneChip® Expression Analysis Technical Manual, Affymetrix company)
RNA quality check by electron gel
1
2
3
4
5
For Electrophoresis:
Please refer to the image at the left for the layout of the gel
Lane 1:100 bp DNA ladder
Lane 2: acceptable quality
Lane 3: heavily degraded total RNA
Lane 4: slightly degraded and DNA- contaminated sample
Lane 5: 1 Kb DNA ladder
Prokaryotic Total RNA Isolation
As starting material for the cDNA synthesis procedure, total RNA can be isolated by using standard
procedures for bacterial RNA isolation or various commercial RNA isolation kits.
For Pseudomonas aeruginosa and E. coli, we have successfully used the QIAGEN® RNeasy Mini
Purification Kit. Caution should be used to minimize chromosomal DNA contamination during the isolation,
due to the high sensitivity of the assay. It is suggested that no more than 1 X 109 cells are applied to a single
purification column. Also, use the lysozyme at a concentration of 1 mg/mL, and not the recommended 400
μg/mL. Additional DNase I treatment may be required to eliminate DNA contamination when the bacterial
culture is grown at high density.
After purification, RNA concentration is determined by absorbance at 260 nm on a spectrophotometer (1
absorbance unit = 40 μg/mL RNA). The A260/A280 ratio should be approximately 2.0, with ranges between
1.8 to 2.1 considered acceptable. We recommend checking the quality of RNA by running it on an agarose
gel prior to starting the assay. The 23S and 16S rRNA bands should be clear without any obvious smears.
Any indication of the presence of chromosomal DNA contamination (high molecular weight bands or
smears on the gel) would require additional DNase treatment before proceeding to cDNA synthesis.
 0
0