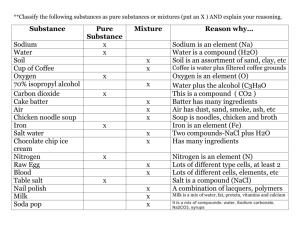
Chemical Formulas and Reactions Chemical formulas are an easy
advertisement

Chemical Formulas and Reactions Chemical formulas are an easy way to know the components of a compound. They are basically shorthand compound names. Just like scientists use e- instead of writing out the word “electron”, scientists use chemical formulas instead of writing out the name of the compound. Ex: sodium chloride is the name of table salt, but the chemical formula is NaCl. Knowing the above example, how do you think scientists write chemical formulas? The chemical (atomic) symbols o In compound names, the metal is always written first and the non-metals written last. o When writing compound names, the last 3 letters (usually –ine) are dropped and replaced with –ide. Example: Chlorine becomes Chloride, oxygen become oxide o Sometimes, the last 2 letters of the non-metal are replaced with –ate. These are usually non-metals that end in –ur or -on like sulfur. Sulfur becomes sulfate o Remember: mono=1, di=2, tri=3, tetra=4, penta=5, hexa=6, hepta=7, octo=8 Practice: Write the chemical formulas for the following compounds. (*hint: don’t forget about valence electrons!) 1. Sodium chloride NaCl 6. Potassium hydroxide KOH 2. Potassium bromide KBr 7. Sodium hydroxide NaOH 3. Carbon dioxide CO2 8. Calcium iodide CaI2 4. Carbon monoxide CO 9. Potassium sulfate K2S 5. Hydrogen chloride HCl 10. Magnesium carbonide Mg2C Part 2: We can actually figure out the elements and number of atoms in the compound from the chemical formula. How do we know which elements are present? What tells you this information? The chemical (atomic)symbols How do we know the number of atoms present in the compound? What tells you this information? The subscripts and coefficients Example: Sodium chloride has 2 atoms present, one of sodium and one of chlorine. Looking at the chemical formula, NaCl, we see that there is only 1 Na (sodium) written and 1 Cl (chlorine) written. Therefore, there are 2 total atoms that make up this compound and 1 is sodium, 1 is chlorine. o Remember that subscripts are small and below the element and are only attached to the element directly in front of the subscript o Remember that co-efficient are applied to the whole compound using the distributive property o Remember that parenthesis mean you have to use the distributive property and the subscript applies to everything within the parenthesis Practice: List the total number of atoms present in the compound and how many of each type of atom the compound contains. 1. LiCl Li - 1 Cl - 1 2. CaCO3 Ca – 1 C - 1 O-3 3. Ca(OH)2 Ca - 1 O–2 H-2 4. Na2CO3 Na – 2 O - 3 C-1 5. CaSO4 Ca – 1 O - 4 S-1 6. (NH4)2SO4 N–2 H–8 7. 2H2O H-4 O-2 8. 3NH3 N-3 H-9 9. 4Cl2 Cl - 8 10. KNO3 K–1 N–1 O-3 S-1 O-4 Part 3: Chemical reactions There are 2 parts to a chemical reaction: the reactants and the products. Label the reactants side and the products side of the equation below. Reactants Products HCl + NaOH H2O + NaCl The reactants are the things you are putting together, or reacting. They are your starting materials. The products are the things that you end up with. What does the law of conservation of mass state? Mass or matter cannot be created or destroyed, only transformed or changed Looking at the above reaction, how many atoms are on the reactants side? 2 hydrogen, 1 Chlorine, 1 Sodium, 1 Oxygen, = 5 total Looking at the above reaction, how many atoms are on the products side? 2 Hydrogen, 1 Oxygen, 1 Sodium, 1 Chlorine = 5 total Are there the same number of each atom on both sides of the reaction? yes The above reaction is called a chemical equation. The arrow is like the equal sign. Both sides of the equation have to be equal, just like in math (Please try and suppress your groan ) Since the law of conservation says that we can’t create or destroy matter, then we just have to rearrange them. This is why both sides of the chemical equation have to be the same. Part 4: Rules for balancing equations 1. Rewrite the equation with enough room for you to change the coefficients (the big number before the molecule). 2. Draw a line down from the arrow dividing the equation into 2 sides. 3. List all the elements on the reactants side down below the first element with the total number of that element for the side. 4. List the elements and the totals on the products side IN THE SAME ORDER AS THE REACTANTS. 5. Balance all the elements. Balance Oxygen (O) last and Hydrogen (H) second to last! Example: Fe + K2S K + Fe2S3 Fe: 1 Fe: 2 K: 2 K: 1 S: 1 S: 3 2Fe + 3K2S 6 K + Fe2S3 Fe: 12 Fe: 2 K: 2 6 K: 1 6 S: 1 3 S: 3