File
advertisement

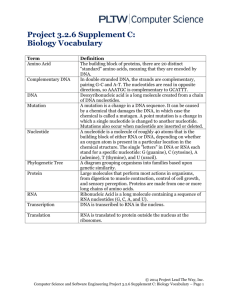
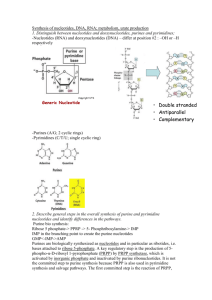
Week 15 Learning Objectives Synthesis of nucleotides, DNA, RNA; metabolism, urate production 1. Distinguish between nucleotides and deoxynucleotides, purines and pyrimidines; Nucleotide – These are the building blocks of DNA and RNA. DNA and RNA are both essentially long chains of nucleotide units. A nucleotide consists of a base (one of four chemicals: adenine, thymine, guanine, and cytosine) plus a molecule of sugar and one of phosphoric acid. Deoxynucleotide – This is a type of nucleotide and is a specific component of DNA. In the Triphosphate form, they undergo DNA polymerase for DNA synthesis. Compared to RNA (ribonucleic acid) which is made of nucleotides that contain an additional oxygen atom, deoxynucleotide lacks one oxygen atom hence the term *deoxy* and DNA – deoxyribonucleic acid. Purine – This is one type of nucleotide base that is present within each nucleotide. The Purine base in DNA and RNA has two variants; adenine and guanine. Pyrimidine – This is the other type of nucleotide base. The Pyrimidine base in DNA and RNA has three variants with two being exclusive to either DNA or RNA; cytosine, thymine (DNA) and uracil (RNA). 2. Describe general steps in the overall synthesis of purine and pyrimidine nucletotides and identify differences in the pathways. Synthesis of the first fully formed purine nucleotide, Inosine Monophosphate (IMP) begins with 5phospho-α-ribosyl-1-pyrophosphate, PRPP. Through a series of reactions utilizing ATP, tetrahydrofolate (THF) derivatives, glutamine, glycine and aspartate this pathway yields IMP. The rate limiting reaction is catalyzed by glutamine PRPP amidotransferase, enzyme indicated by 1 in the Figure. The structure of the nucleobase of IMP (hypoxanthine) is shown. Synthesis of the fully formed pyrimidine nucleotide, Uridine Monophosphate (UMP) starts from carbamoyl phosphate. Carbamoyl phosphate utilized in pyrimidine nucleotide synthesis differs from that synthesized in the urea cycle; it is synthesized from glutamine instead of ammonia and is synthesized in the cytosol. The reaction is catalyzed by carbamoyl phosphate synthetase II (CPS-II). Subsequently carbamoyl phosphate is incorporated into the pyrimidine nucleotide biosynthesis pathway through the action of aspartate transcarbamoylase, ATCase (enzyme #1) which is the rate limiting step in pyrimidine biosynthesis. Following completion of UMP synthesis it can be phosphorylated to UTP and utilized as a substrate for CTP synthase for the synthesis of CTP. Uridine nucleotides are also the precursors for de novo synthesis of the thymine nucleotides. The synthesis of pyrimidines differs in two significant ways from that of purines. First, the ring structure is assembled as a free base, not built upon PRPP. PRPP is added to the first fully formed pyrimidine base (orotic acid), forming orotate monophosphate (OMP), which is subsequently decarboxylated to UMP. Second, there is no branch in the pyrimidine synthesis pathway. UMP is phosphorylated twice to yield UTP (ATP is the phosphate donor). The first phosphorylation is catalyzed by uridylate kinase and the second by ubiquitous nucleoside diphosphate kinase. Finally UTP is aminated by the action of CTP synthase, generating CTP. The thymine nucleotides are in turn derived by de novo synthesis from dUMP or by salvage pathways from deoxyuridine or deoxythymidine. 3. Outline steps in the formation of deoxynucleotides from nucleotides; Formation of deoxynucleotides originate from ribonucleotides (a type of nucleotide). Deoxynucleotide Triphosphates (dNTPs) are formed by reducing Ribonucleotide Diphosphates (rNDPs) with the enzyme Ribonucleotide Reductase (RR). dNTPs are then phosphorilated by the same nucleotide diphosphate kinases that phosphorilate rNDPs. 4. Describe briefly how nucleotides are metabolised and how urate is formed. Urate is formed from the breakdown of purines by different enzymes. The two types of purines are acted upon by two different enzymes to produce xanthine: Guanine is oxidised by Xanthine Oxidase while Adenine is oxidized by Xanthine Oxidoreductase. The two purines each spontaneously deanimate into a product called hypoxanthine. This is oxidized by the two oxidants mentioned above to xanthine. Xanthine is further oxidized by the same two oxidants to form uric acid (urate). 5. Describe the effects of methotrexate and mercaptopurine on nucleotide metabolism. Methotrexate (rINN) (pronounced /mɛθəˈtrɛkseɪt/), abbreviated MTX and formerly known as amethopterin, is an antimetabolite and antifolate drug used in treatment of cancer and autoimmune diseases. Antimetabolites can be used in cancer treatment, as they interfere with DNA production and therefore cell division and the growth of tumors. Because cancer cells spend more time dividing than other cells, inhibiting cell division harms tumor cells more than other cells. Anti-metabolites masquerade as a purine (azathioprine, mercaptopurine) or a pyrimidine - which become the building blocks of DNA. They prevent these substances becoming incorporated in to DNA during the S phase (of the cell cycle), stopping normal development and division. Folate is important for cells and tissues that rapidly divide. Cancer cells divide rapidly, and drugs that interfere with folate metabolism are used to treat cancer. The antifolate methotrexate is a drug often used to treat cancer because it inhibits the production of the active form of THF from the inactive dihydrofolate (DHF). Unfortunately, methotrexate can be toxic, producing side effects such as inflammation in the digestive tract that make it difficult to eat normally. Also, bone marrow depression (inducing leukopenia and thrombocytopenia), acute renal and hepatic failure have been reported. Mercaptopurine is converted to the corresponding ribonucleotide. 6-MP ribonucleotide inhibits purine nucleotide synthesis and metabolism. This alters the synthesis and function of RNA and DNA. Mercaptopurine interferes with nucleotide interconversion and glycoprotein synthesis.