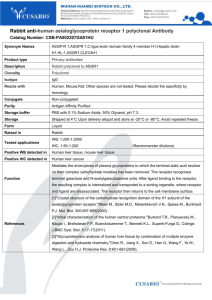
Nutritional regulation of antimicrobial peptide gene - GT-Plus
advertisement

Nutritional regulation of antimicrobial peptide gene expression: Boosting the innate immune response The recent discovery that vitamin D induces antimicrobial peptide gene expression has greatly renewed interest in using nutritional approaches to improve immune function. An examination of published work indicates that numerous nutritional compounds are capable of modulating the expression of genes that encode antimicrobial peptides, supporting the role for proper nutrition in optimal function of the innate immune system. These studies also provide mechanistic explanations for how nutritional deficiencies may lead to susceptibility to bacterial and viral infections. The list of compounds that regulate antimicrobial gene expression range from dietary histone deacetylase inhibitors like sulforaphane and butyrate to ligands for steroid/hormone nuclear receptor family members like retinoic acid and vitamin D. The regulation of the cathelicidin antimicrobial peptide (CAMP) gene by vitamin D is of particular interest to our lab. Vitamin D-mediated induction is a human/primate specific adaptation and is not conserved in other mammals. To study the biological importance of this regulatory mechanism, our group has developed a humanized, transgenic mouse model. The vitamin D receptor (VDR) is a member of the steroid/hormone nuclear receptor superfamily comprised of 48 ligand-activated transcription factors. These factors bind to conserved elements in the promoters of genes through a zinc-finger motif and the adjacent ligand-binding domain activates or represses transcription upon binding to ligand. The active form of vitamin D (1,25-dihydroxyvitamin D) binds to the VDR to modulate the expression of thousands of genes. The nuclear receptor superfamily members function as intracellular detectors for endocrine hormones, dietary lipids and xenobiotic metabolites or compounds. Interestingly, the vitamin D receptor acts as a high affinity receptor for vitamin D and a low affinity receptor for secondary bile acids and potentially other novel nutritional compounds. This suggests that the evolutionary selection to place the cathelicidin gene under the vitamin D receptor allows for its regulation under both endocrine and xenobiotic response systems. Accumulating evidence suggests that in the intestinal tract, the byproducts of gut microbes are important for communicating with the epithelial cells and modulating the expression of the cathelicidin gene. Short chain fatty acids like sodium butyrate are produced by fermentation of fiber by microbes in the colon to induce expression of the cathelicidin and the vitamin D pathway has been implicated in this induction. Both primary and secondary bile acids (the latter produced by gut microbes) modulate cathelicidin expression in the colon and biliary tract via the VDR and the xenobiotic receptor, farnesoid X receptor (FXR). These regulatory pathways may be important for establishing a mucosal barrier to prevent contact of microbes and pathogens with the intestinal epithelium. Several recent studies suggest that the VDR may act as a receptor for additional nutritional ligands including curcumin and polyunsaturated fats like -linolenic acid, docosahexaenoic acid, eicosapentaenoic acid and arachidonic acid. The in vivo relevance of these findings remains to be elucidated, but it is intriguing to consider that numerous nutritional compounds may modulate the expression of VDR target genes like antimicrobial peptides. Future studies in both humans and our “humanized” mouse model will elucidate the importance of this regulation and provide additional support for adequate nutrition to fight infection and possibly lead to the development of potential therapies to treat infection.