Positional statement on Diclofenac/ ibuprofen switch
advertisement

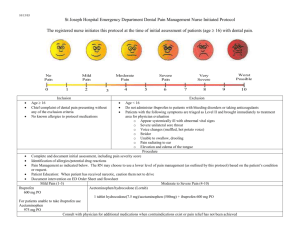
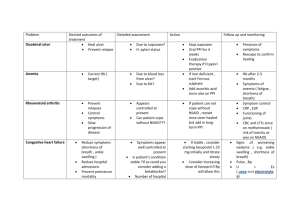
Policy Statement Oral NSAID (Non-Steroidal Anti-inflammatory Drug) of choice at ASPH NHS Foundation Trust Date of Issue 11th January 2012 Review Date: November 2013 (Unless new published evidence becomes available before this date OR there is new published national guidance e.g. NICE) Policy statement: ASPH Drugs & Therapeutics Committee recommends the following in relation to choice of oral NSAID and cardiovascular (CV) risk: Ibuprofen in doses of up to 1200mg/day in divided doses is NSAID of choice Naproxen up to 1g per day in divided doses is second line NSAID of choice. In cases where ibuprofen and naproxen are either not tolerated or ineffective, diclofenac may be considered as an option for the management of acute or chronic inflammatory conditions following discussion with the patient with regard to the risks and benefits of treatment. This should be documented in the patient’s notes, and used for the shortest possible course. Co-prescription of a gastroprotective agent should be considered according to individual risk factors Where oral diclofenac is prescribed first line for acute pain eg. post –op, A&E, trauma, this shall be substituted by ibuprofen or naproxen (if ibuprofen not suitable) in suitable doses as above. Prescriptions may be changed by pharmacists without contacting the prescriber, taking into account allergies, adverse drug reaction information etc. Where rectal or injectable diclofenac is indicated, this must be used only for the shortest possible time until conversion to oral ibuprofen/ naproxen is possible. There are no available rectal/ injectable dosage forms of ibuprofen or naproxen. Exceptions: In post caesearean section patients who are breast feeding and taking medication orally, ibuprofen is still first line choice for these patients, however diclofenac (short term and at the lowest possible dose) is second line as it is safer in breast feeding than naproxen. Patients who present with renal colic or renal stones and require admission may be treated with oral diclofenac for a short time whilst they are inpatient. Patients who are discharged after assessment in A&E, or after an inpatient stay must be prescribed ibuprofen first line, naproxen second line. This has been agreed with Urology. For paediatric patients who require short term oral NSAIDs, ibuprofen is first line, diclofenac will be second line (as this has more indications than naproxen in children). Chronic long term users of diclofenac (eg. rheumatology patients, palliative care patients) must be reviewed on an individual patient basis and a decision made accordingly. Any switching by pharmacists without contacting the prescriber does not apply in these patient groups. Key Considerations: NSAIDs are commonly prescribed drugs. All NSAIDs carry the risk of side effects, which can be serious and life-threatening. The gastrointestinal toxicity of NSAIDs is well documented and in recent years the risk of unwanted cardiovascular effects associated with NSAIDs has been publicised by the Medicines and Healthcare products Regulatory Agency (MHRA) and the National Prescribing Centre (NPC). In NHS Surrey, the proportion of diclofenac prescribing is relatively high- 48% of oral NSAID items compared to 37% nationally. This is despite the MHRA advice Oct 2006 on cardiovascular risk with diclofenac and subsequent NPC initiatives starting November 2007. Not all traditional NSAIDs carry the same CV risk: diclofenac 150mg/ day appears to be associated with a similar excess risk to coxibs, whereas low dose ibuprofen (<1200mg/day) and naproxen 1000mg/day appear to be associated with a lower risk. Subsequent epidemiological studies reviewed in Drug Safety update reinforce this and provide further evidence that thrombotic CV risk applies to all NSAID users irrespective of their baseline risk and not only to chronic users. All oral NSAIDs (including cox II inhibitors) have analgesic effects of similar magnitude. However about 60% of patients will respond to any NSAID, those who do not respond to a particular NSAID may well respond to another. Pain relief starts soon after the first dose and full analgesia is normally obtained within one week. Antiinflammatory effects may take 3 weeks to achieve. At ASPH, oral diclofenac stock (including prepacks) has been removed from most ward areas. Outpatients and A&E prescriptions will be monitored to assess compliance with prescribing recommendations above. Date taken to Drugs & Therapeutics Committee 24th November 2011