Rates of Reaction - Garbally Chemistry
advertisement

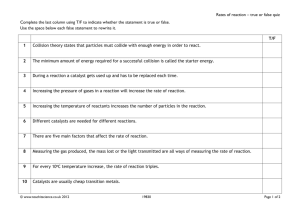
Rates of Reaction The rate of a reaction is defined as the change in concentration per unit time in any one reactant or product. The rate of reaction depends on 5 factors. Nature of reactants Particle size Concentration Temperature Catalysts. Nature of reactants The reaction between acidified sodium dichromate and Ammonium Iron (11) sulphate is instantaneous. (Ionic) The reaction between acidified sodium dichromate and ethanal occurs much more slowly. (Covalent) Particle Size. Large Marble chips Small marble chips Powdered Marble CaCO3 +2HCl CaCl2 +CO2 + H2O The Rate of the reaction increases as the particle size decreases. The smaller the particle size the greater the surface area. Therefore the greater the number of collisions, the greater the number of successful collisions. Concentration The greater the concentration the greater the rate of reaction This reaction is studied using the reaction between sodium thiosulfate and Hydrochloric acid. Na2S2O3 + 2HCl -> S + 2NaCl + SO2 + H2O If the concentration of the reactants is increased, the number of collisions will also be increased. If the number of collisions is increased then the number of effective collisions will be increased. Temperature The greater the temperature the greater the rate of reaction This reaction is studied using the reaction between sodium thiosulfate and Hydrochloric acid. Na2S2O3 + 2HCl -> S + 2NaCl + SO2 + H2O The rate of a reaction increases as the temperature increases because more of the colliding molecules have the minimum activation energy needed to react. Catalysts A catalyst increases the rate of a chemical reaction by lowering the activation energy of the reactants. A catalyst speeds up the rate of a chemical reaction but does not take part in the reaction itself. General properties of catalysts Catalysts are unchanged at the end of a reaction. Catalysts tend to be specific. I.e. Lipase breaks down fats Pepsin breaks down protein Amylase breaks down starch. Catalysts need only be present in small amounts. In the case of equilibrium reactions, catalysts increase the rate at which equilibrium is reached but have no effect on the position of equilibrium. Catalytic poisons can destroy catalysts. Types of Catalysis. Homogeneous catalysis. This is catalysis in which both the reactants and the catalyst are in the same phase. (Iodine snake experiment) Heterogeneous catalysis. This is catalysis in which the reactants and catalyst are in different phases. (Hydrogen peroxide (liquid) and Manganese dioxide (solid)) Auto catalysis. One of the products in the reaction catalyses the reaction. (Permanganate ions and Fe2+ ions.) Mechanisms of catalysis Intermediate compound theory. A + B AB SLOW A + C AC FAST AC + B AB + C FAST The decomposition of hydrogen peroxide catalysed by the presence of I ions (iodine snake reaction) illustrates the formation of an intermediate. Also Sodium Hydrogen tartrate +Hydrogen peroxide + Co2+ ions (pink) Pink Blue/Green Pink Cobalt Intermediate Cobalt Surface Adsorption Theory Platinum 2H2 + O2 2H2O A good example is the reaction of Hydrogen and Oxygen to form water using finely divided Platinum as the catalyst. The Hydrogen and oxygen molecules settle on the surface of the catalyst. The adsorbed atoms form weak bonds with the metal atoms. Transition metals can act as catalysts because they have vacant d orbitals. The hydrogen and oxygen molecules then react to form water. The products leave the surface of the catalyst. (desorption) Catalytic converters. Exhaust fumes contain carbon monoxide (CO), nitrogen monoxide (NO), nitrogen dioxide (NO2) and unburnt hydrocarbons. a catalytic converter converts these gases to environmentally friendly gases. The catalytic converter consists of a thin coating of platinum, palladium, and rhodium on a ceramic or metal honeycomb inside a stainless steel case. Temp= 300 C 2CO + 2NO Pt/Pd/Rh 2CO2 + N2 The unburnt hydrocarbons react with oxides of nitrogen to form carbon dioxide nitrogen and water.. Collision Theory For a reaction to occur, the reacting particles must collide with each other. For the formation of product a certain minimum energy is required in the collision. Such a collision is called an effective collision. The Activation energy is the minimum energy which colliding particles must have for a reaction to occur. Energy profile diagram. A catalyst works by reducing the activation energy.