sm_file_BIP21010_1
advertisement

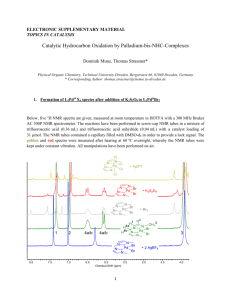
Supporting Information Development of Bifunctional Photoactivatable Benzophenone Probes and their Application to Glycoside Substrates Nir Qvit1,*, Galya Monderer-Rothkof2, Ayelet Ido2, Deborah E. Shalev3, Orna AmsterChoder2 and Chaim Gilon1,* 1 Institute of Chemistry, The Hebrew University of Jerusalem, Jerusalem, 91904, Israel and 2 Department of Molecular Biology, The Hebrew University Medical School, Jerusalem, 91120, Israel, and 3The Wolfson Centre for Applied Structural Biology, The Hebrew University of Jerusalem, Jerusalem, 91904, Israel. *To whom correspondence should be addressed. Institute of Chemistry, The Hebrew University of Jerusalem, Jerusalem 91904, Israel. Phone: +972-2-6586181. Fax: +972-26515731. Email: nir.qvit@gmail.com, gilon@vms.huji.ac.il. Contents Structure and characterization of 7 Pages S2 – S6 Structure and characterization of 4 S7 – S9 Structure and characterization of 5 S10 – S11 Structure and characterization of 6 S12 – S14 S1 Figure S1. Structure and characterization of 7. Structure (A) and MS (B) of 7. The peaks in the MS are 481.2 m/z, MW of 7 + H, and 319.1 m/z MW of the fragment of 7 (marked in Figure A, in the box) obtained from the MS analysis, (C) Analytical HPLC of product 7, (D) 1H NMR of 7 in ACN-d3, (E) 13C NMR of 7 in ACN-d3, (F) COSY spectrum of 7 in ultra-dry ACN-d3 showing aromatic (left) and aliphatic (right) regions of spectrum, (G) ROESY spectrum of 7 in ultra-dry ACN-d3 showing specific interactions designated in text, which give the connectivity of the molecule. S2 S3 F S4 G Proton Proton chemical shift (ppm), multiplicity (d-doublet, t-triplet, mDesignation multiplet), (integration), J-coupling (Hz), 600 MHz spectrometer. H1 H 4.98 ppm, d (1H) H2 H 3.47 ppm, unresolved OH2 H 3.75 ppm, d (1H), J=3.3 Hz H3 H 3.47 ppm, unresolved OH3 H 3.59 ppm, d (1H), J=2.6 Hz H4 H 3.44 ppm, m (1H) OH4 H 3.47 ppm, unresolved H5 H 3.51 ppm, m (1H) CH26 H 3.87, 3.70 ppm, m (2H) OH6 H 2.92 ppm, t (1H), J=6.2 Hz H12 H 7.74 ppm, s, broad (1H) H8 H 7.28 ppm, dd (1H) J=9.0, 3.0 H9 H 7.07 ppm, d (1H) J=9.0 Hz OH10 H 11.80 ppm, s (b, 1H) H14 H 8.11 ppm, dm (2H) J=8.6, 1.9 Hz S5 Proton Proton chemical shift (ppm), multiplicity (d-doublet, t-triplet, mDesignation multiplet), (integration), J-coupling (Hz), 600 MHz spectrometer. H15 H 8.01 ppm, dm (2H) J=8.6, 1.9 Hz H19 H 7.89 ppm, dm (2H) J=8.4 Hz H20 H 7.62 ppm, tm (2H) unresolved H21 H 7.75 ppm, tt (b, 1H) J=7.5, 1.3 Hz Assignment considerations: Chemical shift considerations determined that H1 was the low-field signal in the aliphatic region. The H1 peak showed a single interaction with H2, which showed a strong interaction with its hydroxyl, OH2 and a weak interaction with OH3. All five hydroxy groups in the molecule showed exchange peaks with water in the ROESY spectrum. The OH6 peak was the only OH peak with triplet multiplicity. It showed COSY interactions with both C6 methylene protons, which both interacted with H5 and each other. H5 interacted with H4. H3 and OH4 are assigned as part of the unresolved multiplet at H 3.47 ppm, which also shows an exchange peak with water in the ROESY spectrum. The aromatic spin systems comprising protons H14-H15 and H19-H20-H21 were assigned according to the number of spins in the system, their connectivity and multiplicity. H14 and H15 showed COSY interactions between them, and H14 showed a ROESY interaction with OH10 whereas H15 did not, thereby giving the directionality of this system. The H19-H20H12 protons were in a unique system with unambiguous coupling and integration values. The aromatic spin system comprising H8-H9-OH10-H12 showed COSY interactions between H8 and both H9 and H12. Coupling values gave the assignment; however the specific connectivity of the azo bond was not evident from the correlation spectrum, and is discussed in the text. S6 Figure S2. Structure and characterization of 4. (A) Structure of 4, (B) MS of 4. The peaks in the MS are: 538.2 m/z – MW of 4 + H, 560.2 m/z - MW of 4 + Na and 376.1 m/z The fragment of 4 (marked in Figure A, in the box) obtained from the MS analysis, (C) Analytical HPLC of 4, (D) 1H NMR of 4 in ACN-d3, (E) 1 H NMR of 4 in DMSO-d6. (F) 13C NMR of 4 in DMSO-d6. S7 S8 S9 Figure S3. Structure and characterization of 5. (A) Structure of 5, (B) MS of 5. The peaks in the MS are: 647.4 m/z – MW of 5 + H, 669.4 m/z - MW of 5 + Na and 485.3 m/z, the fragment of 5 (marked in Figure A, in the box) obtained from the MS analysis, (C) Analytical HPLC of 5, (D) 1H NMR of 5 in ACN-d3, and (E) 13C NMR of 5 in ACN-d3. S10 S11 Figure S4. Structure and characterization of 6. (A) Structure of 6, (B) MS of 6. The peaks in the MS are: 793.2 m/z – MW of 6 + H, and 630.2 m/z, the fragment of 6 (marked in Figure A, in the box) obtained from the MS analysis, (C) Analytical HPLC of 6, (D) 1H NMR of 6 in DMSO-d6 at 25 °C, (E) 1 H NMR of 6 in DMSO-d6 at 45 °C and (F) 13C NMR of 6 in DMSO-d6. S12 S13 S14 S15