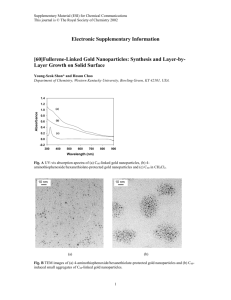
Electronic Supplementary Information
advertisement

Electronic Supplementary Information Selective recognition and discrimination of H2PO4 and F based on a cleft-shaped anion receptor incorporating bisamide and bispyrrole groups Yu-hui Zhou, Peng-cheng Zheng and Xiao-ping Bao* Figure S1 The selectivity receptor 1 for F in the presence of other anions Figure S2 The selectivity receptor 1 for H2PO4 in the presence of other anions Figure S3 The Absorbance at 354 nm of receptor 1 in the presence of both F and H2PO4 as well as three tested anions of H2PO4 , F and AcO Figure S4 UV-vis spectra of receptor 1 in DMSO upon addition of increasing amount of H2PO4 Figure S5 UV-vis spectra of receptor 1 in DMSO upon addition of increasing amount of AcO 1 1.0 _ 1+F _ _ 1+F +AcO - 1+F +Cl _ _ _ 1+F +Br _ _ 1+F +I _ 1+F +NO3 _ _ _ 1+F +HSO4 Absorbance at 354 nm 0.8 0.6 0.4 0.2 0.0 Figure S1 Absorbance at 354 nm of receptor 1 (2.70 105 M) upon addition of 10 equiv. of F in the presence of an equimolar amount of other anions in dry CH3CN. 2 Absorbance at 316 nm 0.8 1+H2PO4 _ _ _ _ 1+H2PO4 +AcO 1+H2PO4_+Cl_ 1+H2PO4 +Br _ _ 1+H2PO4 +I _ _ _ 1+H2PO4 +NO3 1+H PO _+HSO _ 2 4 4 0.6 0.4 0.2 0.0 Figure S2 Absorbance at 316 nm of receptor 1 (2.70 105 M) upon addition of 10 equiv. of H2PO4 in the presence of an equimolar amount of other anions in dry CH3CN. 3 Absorbance at 354 nm 1.0 only 1 _ 1+F +H2PO4 _ _ _ 1+F +H2PO4 +AcO _ 0.8 0.6 0.4 0.2 0.0 Figure S3 Absorbance at 354 nm of receptor 1 (2.7105 M) in the presence of both F and H2PO4 as well as three tested anions of H2PO4 , F and AcO (10 equiv. for every kind of the added anion). 4 1.0 Absorbance 0.8 0.6 0.4 0.2 0.0 250 275 300 325 350 375 400 425 450 Wavelength/nm Figure S4 UV-vis spectra of receptor 1 (2.0 105 M) in DMSO upon addition of increasing amount of H2PO4 (0~52 equiv.). 5 1.0 Absorbance 0.8 0.6 0.4 0.2 0.0 250 275 300 325 350 375 400 425 450 Wavelength/nm Figure S5 UV-vis spectra of receptor 1 (2.0 105 M) in DMSO upon addition of increasing amount of AcO (0~56 equiv.). 6