CO2-Soluble GRAS Solvents for Extracting Nutraceuticals By a Gas
advertisement

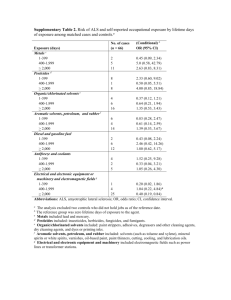
CO2-Soluble GRAS Solvents for Extracting Nutraceuticals By a Gas Anti-Solvent (GAS) Process Peter K. Kilpatrick, Ruben G. Carbonell Graduate Student Mentors: Xenia Tombokan and Remil Aguda Department of Chemical and Biomolecular Engineering North Carolina State University, Raleigh, NC 27695 This project involves a novel extraction process for obtaining high value nutraceuticals from plants. It utilizes CO2-soluble GRAS (generally recognized as safe) ester solvents to extract the nutraceuticals from plant tissues. CO2 is then used to recover the product via a gas anti-solvent (GAS) process1,2. This is followed by an increase in pressure to dissolve and recycle the liquid solvent. Since the liquid extraction step, the GAS process, and the dissolution of the liquid solvent can all operate at ambient temperatures, this process can reduce product degradation and thus increase yields. Current work focuses on screening for the GRAS solvents that give the best solubility for nutraceuticals (yield) and on finding the best conditions to optimize product yields and purity (selectivity). Nutraceuticals are polar organic molecules extracted from plants which have associated health benefits. As model systems, we are studying the recovery of anthocyanins isolated from purple sweet potato, solanesol and sclareol isolated from clary sage, and lutein as well as zeaxanthin derived from marigold flowers. The first approach to this project is to screen for GRAS solvents that can effectively remove these materials from plant tissues. Alkyl aryl esters are promising for this purpose since their substituent groups can be chosen to achieve desired polarity and hydrophobicity to target a specific product. In addition, complete miscibility of these esters with dense CO2 makes them suitable for GAS precipitation. Screening of GRAS solvents is done by measuring the solubility of each solute of interest in various ester solvents. Measurements can be done using UV visible spectroscopy for anthocyanins and marigold derivatives. However, it is difficult to analyze sclareol and solanesol using this method because they have very low absorbance at visible ranges of wavelengths. Hence, gas chromatography is used to measure the solubility of these components. We plan to extend our work in this project to optimize the extraction and fractionation of lutein and zeaxanthin using alkyl alkyl esters. These important are critical for controlling the health and vitality of important tissues in the eye for sight. References 1. Liu, Z.; Li, D.; Yang, G.; Han, B. (2000). Solubility of hydroxybenzoic acid isomers in ethyl acetate expanded with CO2. Journal of Supercritical Fluids 18:111-119. 2. Cocero, M.J.; Ferrero, S. (2002). Crystallization of β-Carotene by a GAS process in batch: Effect of operating conditions. Journal of Supercritical Fluids 22: 237-245. 3. Chang, C.J.; Randolph, A.D.; Craft, N.E. (1991). Separation of β-Carotene Mixtures Precipitated from Liquid Solvents with High-Pressure CO2. Biotechnology Progress 7: 275-278. 4. Catchpole, O.J.; Hochmann, S.; Anderson, S.R.J. (1996). Gas Anti-Solvent Fractionation of Natural Products. High Pressure Chemical Engineering, Industrial Research Ltd.