RRQcat - Leek High School
advertisement

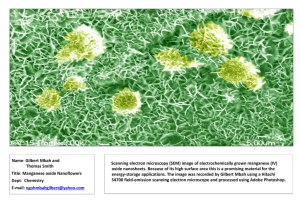
RATES OF REACTION - CATALYSTS Action of catalysts on hydrogen peroxide. 1. When manganese (IV) oxide is put into hydrogen peroxide solution there is a vigorous reaction and the gas oxygen is produced. The manganese(IV) oxide acts as a catalyst. Joe decides to investigate this reaction. He weighs out 1.0g of manganese(IV) oxide and he drops it into a flask containing 50 mls of hydrogen peroxide solution. The volume of the gas formed was recorded every 30 seconds for 5 minutes. The results are shown below. Time (secs) Volume (mls) of gas produced 0 0 30 16 60 28 90 40 120 50 150 58 180 64 210 68 240 70 270 72 300 72 a) What does a catalyst do ? b) Draw a diagram of the apparatus Joe could use. c) Plot a graph of the volume of gas produced (vertical axis) vs time taken (horizontal axis). d) During the reaction: How does the speed of the reaction change? What happens to the concentration of the hydrogen peroxide? What happens to the mass of the manganese(IV) oxide catalyst. e) If Joe used 1.0g of manganese(IV) oxide catalyst would the speed of reaction increase, remain the same or decrease. Explain your answer. 2. a) Hydrogen peroxide can be broken down into water and oxygen. This reaction is normally very slow (over weeks and months) but it can be speeded up if a catalyst is used. Write a word equation for the reaction. Jill decides to investigate this reaction by using a variety of substances which she thinks will act as catalysts. The substances she chooses are: copper oxide, lead oxide, zinc oxide, potassium iodide carrot juice. celery juice In each case she measures out 1.0g of each substance and then puts it into 100 mls of hydrogen peroxide. The volume of gas given off is recorded every 10 seconds over 2 minutes. The temperature of the hydrogen peroxide is 20oC. The results are shown below. Time (in secs) 0 10 20 30 Volume of oxygen given off (mls) 40 50 60 70 80 90 100 110 120 Substance copper oxide, 0 11 18 24 29 33 36 38 40 42 43 43 44 zinc oxide, 0 6 10 13 16 19 22 24 26 27 29 31 32 carrot juice 0 9 17 25 33 39 45 51 56 60 65 69 73 lead oxide 0 2 4 5 7 8 10 11 13 15 16 18 20 potassium iodide 0 8 15 22 29 35 41 47 54 60 66 71 76 celery juice 0 7 14 20 26 31 37 43 48 52 56 60 63 b) From these results i) Which is the best catalyst ? ii) Which is the worst catalyst ? iii) Carrot juice and celery contain enzymes which help breakdown the hydrogen peroxide. Explain what an enzyme is. c) Jill now decides to do the reaction with the hydrogen peroxide at 60oC. The results are shown below. Time (in secs) 0 10 20 Volume of oxygen given off (mls) 30 40 50 60 70 80 90 100 110 120 Substance copper oxide, 0 18 34 47 58 67 75 81 85 87 89 90 90 zinc oxide, 0 12 22 31 40 48 55 62 68 74 79 83 86 carrot juice 0 2 3 3 4 4 4 5 5 6 6 6 6 lead oxide 0 12 18 25 31 36 41 46 51 55 59 63 66 potassium iodide 0 18 34 48 59 68 76 82 86 89 89 90 90 celery juice 0 1 1 2 2 2 3 3 3 4 4 4 4 From these results: i) which is the best catalyst now ? ii) which is the worst catalyst now ? iii) why are the celery juice and the carrot juice so bad as catalysts now? iv) which reactions seem to have finished ? How do you know ? v) in 120 seconds lead oxide produces 66 mls of gas. How much gas would be produced with lead oxide when the reaction is complete ? 3. In this experiment 0.5g of manganese dioxide are added to 75 mls of hydrogen peroxide solution in a conical flask. The volume of gas produced (which is collected in a syringe) is measured every 30 seconds. The apparatus used is shown below. The results are shown in the table below. Time (secs) 0 30 60 90 120 150 180 210 240 270 300 Volume (mls) of gas 0 25 38 50 61 68 75 79 80 80 80 produced a. At what time does the reaction stop ? b. Draw a graph of volume of gas produced (vertical axis) against time (horizontal axis) c. How long does it take for: (i) the first 25 mls of gas to be produced ? (ii) the second 25 mls of gas to be produced? (iii) the third 25 mls of gas to be produced ? d. How does the speed of the reaction change in the first 180 seconds. e. Explain why the speed of the reaction changes. - RATES OF REACTION - CATALYSTS 4. Potassium chlorate decomposes when heated, like this: 2KClO3(s) 2KCl (s) + 302(g) a. Write a word equation for the reaction. b. What gas is given off when potassium chlorate decomposes? c. Manganese (IV) oxide acts as a catalyst for this reaction. What would you expect if two test tubes, one of potassium chlorate and the other a mixture of potassium chlorate and manganese (IV) oxide, were heated? d. Potassium chloride is soluble in water, and manganese(IV) oxide is insoluble. How could you show that the manganese (IV) oxide is not used up during the reaction? e. Will there be more or less oxygen produced, when the catalyst is used? Explain your answer.