Molecular Biology and Biotechnology Test III
advertisement

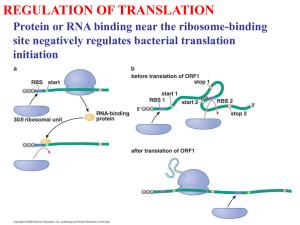
Molecular Biology and Biotechnology Test III Spring 2006 Woo-hoo!!!!!!!!!! Last one!!!!!!!!!!!! (Except for the comprehensive final…..) Translation Definition: The process of making proteins from mRNA. The mRNA is identical to the DNA coding strand except that uracil replaces thiamine. The mRNA transcription codes for the amino acids that make up the peptide. The peptide undergoes post-translational modifications to become active protein. ***Biotech note*** Keep in mind that this process is much more complicated in Biotech than in natural biology. The cell is designed to make proteins; it is difficult to do in a lab. The goal is to produce products: - proteins (i.e., insulin): requires a gene - small non-protein molecules (i.e., antibiotics) – may require a gene - Other products (i.e., DNA or RNA) – may require a gene We produce them in organisms: - bacteria - fungi - plants - animals - animal cells in culture For successful recombination, must have: - correct peptide ( successful transcription; translation) - correct post-translational modifications DNA_____________________GAT_____________________ (coding strand) _____________________CTA_____________________ (template) RNA_____________________GAU_____________________ (transcription) Peptide NH2_______________Asp______________________COOH Genetic code: A codon is a sequence of three base pairs that code for a specific amino acid. For example, above, “GAU” codes for Aspartate (Asp). The genetic code is redundant, which means that several codons can code for the same amino acid (for example, AAA and AAG both code for lysine). This helps prevent point mutations: If an AAA underwent a point mutation to an AAG, this mutation would not be apparent in the phenotype because the mutant codon codes for the same amino acid as the normal codon. All aminos except Trp and Met have two or more codons. Start codon: AUG (also codes for Met; this means that all peptides start with a methionine – but it can be removed during post translational modifications. Only the first AUG in the mRNA is a start codon: AUG later in the strand simply codes for Met) Stop codons: UAA, UGA, UAG (Do not code for any amino acids) mRNA 5’__AUG_____________________UAA_____________________AAAUAA 3’ start coding region stop un-translated region poly A tail The 5’ end is fed into ribosome first. Translation begins at the start and ends at the stop. The un-translated region can be 10 – 100’s of BP’s. It is an additional protection against exonucleases. Mutations: 1. Start mutation: Point mutation at the start codon means that the protein won’t start at the right place; instead it may start later when the ribosome encounters an AUG. This results in a truncated protein. 2. Stop mutation: A mutation in the stop codon means the protein is too long; this useless protein is usually degraded in the cell. 3. Coding mutation: Change in the protein’s primary structure can have a variety of huge consequences: altered folding, altered biologic activity; altered affinity for receptor/ligand. Frameshift mutation is, of course, the most serious mutation. Finishing the protein After translation, the protein may undergo several modifications in order to be biologically active. 1. Chaperones: Help fold the protein and translocate the protein to the correct membrane/cell. Specific to the type of cell that produces the protein that the chaperone assists in folding. If the peptide is a mutant, the chaperone may not be able to fold it. If the chaperone folds the protein wrong, the cell usually degrades the protein. Not all proteins require chaperones; some fold spontaneously. *** Biotech Note*** If we produce a recombinant protein that requires a chaperone, we must have that chaperone available to fold the protein. A big problem occurs when producing a Eukaryote protein in a prokaryote cell. ~~~Clinical Note~~~ Mad-Cow Disease (Cruetzfeld-Jakob’s) is caused by an infectious protein (these are known as “prions”). PrPc is a normal protein found on the surface of neurons (their function is unknown). PrPc’s 4’ structure consists mainly of a-helix. PrPsc is an abnormal infectious protein consisting mostly of B-sheet. When the PrPsc contacts PrPc, it has after-the-fact chaperone activity and changes the conformation of PrPc to B-sheet. This initiates a cascade effect and soon all PrPc’s are converted to PrPsc. 2. Secretion: A produced protein may need to be secreted by the cell. For example, activated B cells secrete Ab (Aaaagh! Immunology flashbacks!!!), or a cell secretes hormones into the blood stream. Proteins that must be secreted follow this general process: a. Peptide contains a hydrophobic signaling sequence on the N-terminus b. This sequence is recognized by a signal recognition particle, a protein that directs the growing peptide (still attached to the ribosome) to the ER. c. The ribosome attaches to the rough ER because the signal sequence binds to a receptor (called sec61 translocation complex) on the ER membrane. The peptide enters the lumen of the ER through a channel that opens to allow entry. d. Signal sequence is removed by enzymes e. Folding, glycosylation, etc may occur (chaperones and glycosyl transferase are present in the ER) f. Transport vesicles bring the peptide to the cis face of the golgi g. The protein is transolcated by the golgi apparatus; it is secreted by secretory vesicles. There are several secretory pathways. Secretion depends on several factors: secreted proteins are usually soluble in the RER; various chaperones or translocation / export apparatus may be involved. This process requires a secretory signal. This link demonstrates secretion: http://www.sumanasinc.com/webcontent/anisamples/molecularbiology/proteinsecretion_f la.html 3. Glycosylation: Addition of sugar(s) to create a glycoprotein. Glycosyl Transferase adds sugar to a peptide. This process is cell and tissue dependent – the protein must be produced in a cell that is able to glycosylte it, or it will remain inactive. Why add a sugar? - mono/polysaccharides have increased water solubility - change the 3-D structure (and therefore the function) - Add a recognition signal (most antigens are recognized by their sugars) ***Biotech Note*** Again, when making a biotech protein, must be able to glycosylate your protein if necessary; if your bacterial host cannot glyocosylate the human protein you are producing, your protein will be inactive. 4. Protein targeting: Getting the protein to the right location in the cell. This depends on correct folding and glycosylation. Proteins will contain a targeting sequence, a sequence of amino acids that that signal that the protein must be translocated. There are various translocation mechanisms and sequences. For example, the nuclear localization sequence is recognized by nuclear transport receptors in the nuclear cytosol. They bind to the localization sequence and transport the peptide into the nucleus through nuclear pores. ***Biotech Note*** To successfully create a recombinant protein, have to identify the signaling sequence when identifying the gene. 5. Other protein structure modifications: a. Proteolysis: cutting the peptide. Some proteins must have a piece removed after folding in order to be active. Ex: Insulin ***Biotech note*** When insulin was discovered, at first scientists thought it was two separate peptides bound by disulfide linkages. Actually it is one protein that has been modified by proteolysis. That is why it is not always a good idea to work backwards from the protein to the gene sequence: may give wrong sequence. For example, if we did that for insulin we would leave out a huge chunk of sequence because the connecting peptide is not present in mature insulin. b. Modification of amino acids: Allows protein to function (alter amino acids in ligand binding sites; change affinity for certain molecules, etc) Examples: - add acetyl group - add disulfide bonds (requires correct folding to allow necessary proximity required for bond to form) - methylation - 4-OH added to proline Collagen is an example of a protein that undergoes extensive post-translational modifications necessary for it to be functional. This research from Raili Myllyla, PhD, discusses collagen modifications if you are interested: Collagen molecules are composed of three polypeptide chains, which fold to form a characteristic triple-helical structure. The biosynthesis of collagen requires a series of post-translational modifications and many of these are unique to collagens and other proteins with a collagen-like amino acid structure. The modifications, which occur within the rough endoplasmic reticulum (ER) of the cell, include hydroxylation of lysyl residues and glycosylation of hydroxylysyl residues. This glycosylation is unique to collagenous structures and the carbohydrate group can be either the monosaccharide galactose or the disaccharide glucosylgalactose. The side chains of the modified lysyl residues extend outward from the helix and have a high potential for lateral interactions between collagen molecules and other extracellular proteins. Modifications of lysyl residues are essential for the proper function of collagen, although the structural and regulatory roles of hydroxylysyl residues and glycosylated hydroxylysyl residues are not well understood. 6. Regulation of Translation: We can regulate a protein during several stages including transcription (most control here), translation, degradation, and allosteric regulation. Regulation during translation is rather rare and includes: a. Phosphorilation of the ribosome. Usually this decreases translation activity. b. Repressor proteins bound to mRNA “save” mRNA until translation is needed. For example, an ovum contains a lot of repressed mRNA that is released for transcription as soon as the egg is fertilized: this allows for the rapid translation required once egg is fertilized. This translation is not necessary if the egg is not fertilized, so it is repressed. More on Proteolysis: Protein Degradation: After the protein is made, it can be controlled by degradation. 1.Why degrade a protein? -. Protein is damaged by age, oxidation, proteolysis, or denaturation -. Protein contains an error from translation or during post-translational modifications (the cell has some “surveillance” system that finds proteins with errors, but little is known about this). -.Need to change activity in cell: -We do not want a protein to constantly act in the cell; protein needs change with metabolic needs of the cell. - Or, we may only want the transient effect of a hormone, so once the protein is used, it is shut off. FSH stimulates follicular growth, but once the follicle is mature it releases a hormone that shuts off FSH stimulation. The FSH that is already circulating must be degraded to prevent continued action. FSH is taken up by cells and degraded in lysosomes (or recycled). FSH is degraded quickly. - Other proteins, such as clotting factor, take a long time to be degraded. -The protein that makes up the lens of the eye is the longest lasting protein in the body. 2. Proteolysis in more depth: a. Protesosomes: Complexes designed to break down proteins inside a cell in a natural, controlled way.Made up of 28 peptides. The 2 componants of a proteosome are the: -Cap, which grabs a protein tagged by ubiquitin, unfold it, and feeds it into the core - Core, which slices proteins up Ubiquitin is a protein that binds to proteins that are targeted for break down. ***Biotech note*** Even if a protein is produced, the producing cell can target it as foreign, tag it with ubiquitin, and destroy it. b. Intracellular digestion: Cell endocytosis a peptide in a endocytotic vesicle. The vesicle fuses with a lysosomes full of lytic enzymes. The enzymes break down the peptide. ***Biotech note*** If a protein is not secreted, we may lyse the cells in order to harvest the protein produced by the cell. Unfortunately, we may also lyse the lysosomes, releasing harsh enzymes into the culture and destroying all the proteins. c. Other proteolytic systems: include caspases, fast paced proteins that cut up proteins inside cells during apoptosis. This protein is under investigation as a cancer drug.