Lab #6 - Cloudfront.net
advertisement

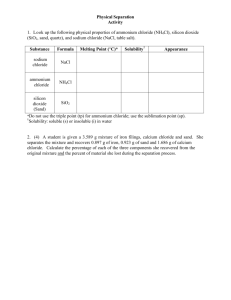
Lab #6 Gravimetric Determination of Chloride Ion Concentration Introduction The chloride ion concentration in a sample of an unknown solid or solution can be determined by precipitating the chloride as silver chloride. This is done by addition of an excess of silver nitrate, forming a precipitate that is easy to filter, wash, and dry in micro- or semi-micro quantities. The mass of the precipitate is used to calculate stoichiometrically the amount of chloride present in the unknown. This experiment is an example of gravimetric analysis. The mass percent of chloride is determined gravimetrically. In this investigation, you will start with a mixture of barium chloride and potassium chloride; your goal is to determine the mass percent of each compound in the mixture. Modified Instructions 1. Weigh out approximately 0.5 g of the unknown mixture. Record the mass of the mixture in the data table. 2. Place the solid in the test tube. 3. Add about 10 mL of distilled water to the solid to dissolve it. 4. Add 2 drops of nitric acid to the solution. 5. Add about 1 mL of 1.0 M AgNO3 dropwise to the solution with gentle shaking. 6. Allow the precipitate to settle. 7. Add a few drops of 1.0 M AgNO3 to the solution. If more white precipitate forms on the addition of AgNO3, repeat steps 5-7. 8. Obtain a piece of filter paper and determine its mass. Record the mass in the data table. 9. Using a funnel and filter paper, filter the precipitate. Rinse three times with water. 10. Put filtrate in the waste container, not down the sink. 11. Allow the precipitate to dry of night. 12. Mass the filter paper and precipitate. Prelab Questions 1. Write the balanced net-ionic equation for the precipitation of chloride ion by silver ion in aqueous solution. 2. If 100 mg of BaCl2∙2H2O is dissolved in water and then an excess of 1.00 M AgNO3 (aq) is added, calculate the mass of silver chloride that will form, assuming complete precipitation. 3. What volume of 1.00 M silver nitrate solution would be needed to completely precipitate the chloride from the barium chloride in Question #2? 4. Why is it important to use an excess of silver nitrate? Calculations 1. The number of moles of silver chloride must equal the number of moles of chloride ion in your sample. Why? 2. Further, the moles of chloride ion must equal the sum of the moles of potassium chloride in the sample, plus twice the number of moles of barium chloride. Why? Represent this total by the letter t: Mol Cl- = mol KCl + 2 (mol BaCl2) = t 3. Further, if we let n represent the number of moles of chloride that originated with the potassium chloride, then t-n give the number of moles of chloride originally in the barium chloride. From this, we can deduce that the mass of the original mixture sample, m, is given by n(MW of KCl) + (0.5)(t-n)(MW of BaCl2) = m Carry out the necessary calculations to determine the composition of the original mixture. Show your work. Postlab Questions 1. What would be the effect on your chloride percentage if: a. Insufficient silver nitrate solution were used? Explain. b. Drying of the filter and precipitate was not complete? Explain c. In the original instructions, the precipitate is dried in an oven and then cooled and weighed. What would the effect be if the precipitate is weighed before it is cooled? 2. Conclude your report with a discussion of experimental errors and identify those portions of the procedure where extra care is needed to ensure satisfactory results. Include, but do not limit your analysis to, the answrs you gave in the preceding question. Consider only those factors that might apply to your own experiment. 3. The purpose of adding the nitric acid during sample preparation is to remove traces of carbonate ion, thus preventing coprecipitation of silver carbonate along with the silver chloride. Why is hydrochloric acid not suitable to use in place of the nitric acid? 4. A sample of a pure unknown chloride (0.0212 g) gave 0.0716 g of silver chloride. Identify the unknown chloride.