Supplementary Information (doc 7792K)
advertisement

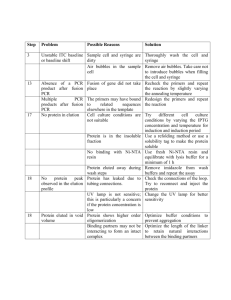
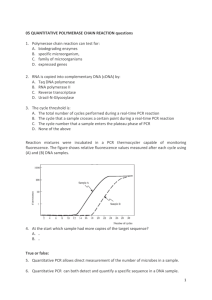
Supplementary Methods: PCR detection of fusion gene To confirm the suspected the BCR-FGFR1 fusion, genomic DNA and total RNA from the patient and a healthy donor were isolated using AllPrep DNA/RNA/Protein kit (Qiagen), and reverse-transcription on RNA was performed with SuperScript II Reverse Transcriptase (Life Technologies). For detection of the BCR-FGFR1 fusion gene from genomic DNA, forward primer on the intron between BCR exon 4 and exon 5 and reverse primer on the intron between exon 8 and exon 9 of FGFR1 were used, while the fusion transcript was detected with forward primer on BCR exon 3 and reverse primer on FGFR1 exon 9. PCR products verification and breakpoint mapping were performed with Sanger sequencing. The reciprocal FGFR1-BCR fusion gene from genomic DNA and cDNA was also detected by PCR with primer pairs flanking FGFR1 upstream of the breakpoint and BCR downstream of the breakpoint. Detection of BCR and FGFR1 with primers flanking within the genes was performed as controls. To measure fusion gene allele burden, nested quantitative real-time PCR on gDNA was performed with Power SYBR® Green PCR Master Mix (Life Technologies) on 7900HT System (Applied Biosystems) following manufacturer’s instruction. Fifteen nanograms of genomic DNA was initially amplified by PCR. This PCR product was then used in a quantitative realtime PCR assay with a second set of nested primers. All primers are listed in Supplementary Table 1. The level of RNase P, a target that exists as a single copy gene per haploid genome was used as internal control for the quantitative real-time PCR1. Supplementary Figure 1. Morphologic, flow cytometric, and cytogenetic findings at presentation. (A) The peripheral blood showed a marked leukocytosis with an absolute neutrophilia, monocytosis, and 42% blasts with a variety of morphologic forms including round to highly convoluted nuclei and delicate to deeply basophilic cytoplasm (60x). (B&C) The bone marrow was hypercellular with similar composition and morphology to that noted in the peripheral blood with occasional eosinophils and mast cells also present (60x). (D) Cytogenetic findings included a t(8;22) with two copies of the der(22). (E) BCR-ABL1 fusion probe analysis demonstrated an abnormal signal pattern with four BCR signals (green); arrows indicate one BCR signal localizing to the der(8)t(8;22). (F) FGFR1 break-apart probe analysis demonstrates a 5’/3’ signal separation consistent with an FGFR1 rearrangement. PCR analysis (not shown) confirmed the BCR-FGFR1 fusion product. (G-I) Flow cytometric findings demonstrate three distinct populations of MPO+ myeloblasts, CD19+/CD79a+/CD22+ lymphoblasts, and cytoplasmic CD3+ T-lymphoblasts. The bright CD117+ events are consistent with increased mast cells. Key: SSC: side scatter; TdT: terminal deoxynucleotidyl transferase; MPO: myeloperoxidase. Supplementary Figure 2. Quantitative nested PCR detection of BCR-FGFR1 fusion. (A) Quantitative nested PCR was performed with primer sets specific for the patient’s BCR-FGFR1 fusion breakpoint or RNase P. Threshold cycle (Ct) versus dilution is shown, demonstrating satisfactory efficiency of the PCR reaction. (B) Allele burden for each sample was detected in triplicate. Allele burden was normalized to the average value at diagnosis. Two replicate experiments are shown. Supplementary Figure 1. Morphologic, flow cytometric, and cytogenetic findings at presentation. A B C E D F p11.2 q11.2 der(22) der(22) 8 G 22 der(8) 262144 H 14.4% I 2.6% 105 15.2% 0.2% 105 SSC-A Blast 131072 104 cCD3 PE-A 79a PE-A 196608 103 65536 29.2% 102 103 104 105 -101 102 CD45 PerCP-Cy5-5-A 1.2% 3.3% K 103 51.6% 102 -101 -102 12.9% 103 104 CD19 APC-A 105 10.4% 103 102 102 74.6% -101 9.9% 102 103 104 105 TdT FITC-A 104 -101 -101 -102 105 105 CD34 PE-A CD10 PE-A 104 -102 104 MPO FITC-A 105 82.6% 103 L 3.2% 2.1% 105 CD117 PE-A J -101 53.8% -103 -102 -101 103 102 -102 0 104 104 103 102 35.3% -101 102 103 104 CD22 FITC-A 2.7% 36.3% 105 -101 58.4% 102 103 104 CD15 FITC-A 105 Supplementary Figure 2. Supplementary Table 1. Primers for detection and quantification of BCR-FGFR1 fusion gene BCRFGFR1 FGFR1BCR gDNA cDNA gDNA cDNA gDNA BCR cDNA gDNA FGFR1 cDNA First round of nested PCR RNase P Second round of nested PCR RNase P BCRFGFR1 BCRFGFR1 Forward Reverse Forward Reverse Forward Reverse Forward Reverse Forward Reverse Forward Reverse Forward Reverse Forward Reverse Forward Reverse Forward Reverse 5’-CACTTGCTTGCCACAGGTTC-3’ 5’-GGGGTGACTCCTTCCCATTC-3’ 5’- GCTGGACTTGGAAAAGGGCT-3’ 5’- CGGGAAGCTCATACTCAGAGAC-3’ 5’-TGTCAAGCTGGAAGAGGTGC-3 5’-ACTGCACCACTGTACTCCAGA-3’ 5’-GACGCAGGGGAGTATACGTG-3’ 5’-ACTCCGTAGTTGTCCACGAA-3’ 5’- GCTGGACTTGGAAAAGGGCT-3’ 5’-AGAGCAGAAAACTCAGCCCC-3’ 5’- GCTGGACTTGGAAAAGGGCT-3’ 5’-ACTCCGTAGTTGTCCACGAA-3’ 5’- TGTCAAGCTGGAAGAGGTGC-3’ 5’-GGGGTGACTCCTTCCCATTC-3’ 5’- GACGCAGGGGAGTATACGTG-3’ 5’-CGGGAAGCTCATACTCAGAGAC-3’ 5'-TAGGATGTTTTTCGCATCTGGC-3' 5'-GGATACAGATTTGCCACGTCAT-3' 5'-CACTTGCTTGCCACAGGTTC-3' 5'-CGGGAAGCTCATACTCAGAGAC-3' Forward 5'-CCCACTTTCTTTAGGCTTCAACTT-3' Reverse Forward Reverse 5'-GGCCCTCTTATTTCTAAAGGCTGA-3' 5'-GACCAGCTCAGTAGGGTTGG-3' 5'-GGGGTGACTCCTTCCCATTC-3' 1. Szantai E, Elek Z, Guttman A, et al: Candidate gene copy number analysis by PCR and multicapillary electrophoresis. Electrophoresis 30:1098-101, 2009