Lecture 1 - Introduction
advertisement

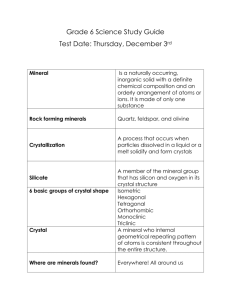
Introduction What’s the definition of a mineral ? Definition of a mineral (Geology 101) 1) Naturally occurring solid 2) Inorganic 3) Definite Chemical Composition 4) Systematic internal pattern - ordered Atomic Arrangement Mineralogy (Gel 2130) definition of a mineral A mineral is a naturally occurring homogenous solid with a definite (but not fixed) chemical composition and a highly ordered atomic arrangement. A mineral is usually formed by inorganic processes. 1) Naturally occurring - Formed by natural processes (incl. kidney stones, bones, siderite (FeCO3) that ppt in water pipes) - Synthetic diamonds, emeralds, rubies, etc. are not minerals - Chemical compounds are not minerals even if they have “mineral structures”. E.g. Calcium Carbonate (CaCO3) added to your Cheerios 2) Homogenous solid - cannot be subdivided into smaller components (excludes perthite – Na-rich lamella in a K-rich feldspar host) - fluids are not minerals – water, oil, gas, mercury (mineral water) - Ice in a glacier is a mineral 3) Definite Chemical Composition - Quartz - SiO2 – pure substance - Dolomite – Ca,Mg (CO3)2 - can also contain Mn, Fe, Sr that substitute for similar sized and charged atoms - Dolomite is better written Ca(Mg,Fe,Mn,Sr)(CO3)2 (within a range) – geothermometers and geobarometers - If concentration of an element exceeds a specific range we give mineral a new name 3) Definite Chemical Composition - If concentration of an element exceeds a specific range we give mineral a new name e.g. Siderite (FeCO3), Magnesite (MgCO3)… - Spinels XY2O4 where X = +2 , Y = +3 Spinel – MgAl2O4; magnetite Fe,Fe2O4 4) Systematic internal pattern (crystalline) - ordered Atomic Arrangement - internal framework of atoms (ions) that are arranged in a regular geometric pattern - if material has no structure is amorphous and called a mineraloid (e.g. opal, limonite) - some materials can start off as minerals but the atomic arrangement can be destroyed by radiation to produce a metamict material (e.g. allanite, zircon, many other radioactive minerals) 5) Inorganic processes – the usual case - Biogenic Organic: carbonates (aragonite, calcite, vaterite) phosphates silica sulfur oxides (magnetite in bird brains) History of Mineralogy Early Humans – used hematite and psilomelane to draw pictures in caves - (25,000 B.C.) Bronze Age metallurgists (5,000 B.C.) Egyptian Tomb paintings of smelting metals Theophratus – (372-287 B.C.) – Greeks Pliny (the younger) – (~100A.D.) 1556 – Georgius Agricola – De Re Metallica - translated by Hoover + Hoover 1912 1669 – Nicolas Steno – noted that angles between different faces were the same despite differences in crystal size, origin, or habit 1780 – Carangeot – invented contact goniometer to measure interfacial angles (external morphology reflects internal structure) 1783 – Rome de l’Isle measured interfacial angles and formulated law of constancy of interfacial angles. 1784 – Rene J. Hauy – developed the concept that minerals were made from small building blocks. - today = molecules forming unit cell. 1801 – He developed system indices for crystal faces 1809 – Wollaston - invented reflecting goniometer and made crystallography an exact science 1779 to 1848 – Berzelius – developed chemical classification system for minerals 1815 – Cordier – started using the microscope to study minerals and developed the immersion (water + oil) methods for crushed minerals. 1828 – William Nicol – Developed the polarizing tool (Nicol prism) for the optical microscope. 1912 – von Laue suggested that crystals would diffract X-rays - diffraction demonstrated by Friederich + Knipping 1914 – Bragg+Bragg – First determination of crystal structure (Nobel prize in Physics) 1960’s – Computerized X-ray diffractometers - Electron Microprobe - Scanning Electron Microscope 1970’s - Transmitting Electron Microscope 1990’s - Ion Microprobe What to learn from course 1) 2) 3) 4) 5) Symmetry – classes Miller Indices Crystal Chemistry – what else fits in Associations Identification