Grade 9 Science – Unit 2
advertisement

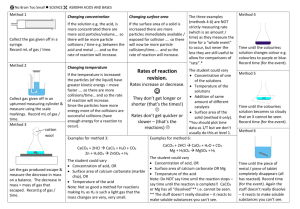
Grade 9 Academic Science – Chemistry Rate of a Reaction The rate of a reaction is the speed of the reaction. It is not “how much” of a product is made; instead, it is “how quickly” a reaction takes place. How can we measure the rate? Consider a reaction zinc + hydrochloric acid —> zinc chloride + hydrogen There are two possible ways of measuring the rate. 1. Measure how quickly one of the products (e.g. the hydrogen) is made. In this case, we could measure the volume of hydrogen (in milliliters) made during a time period (e.g., every 10 seconds). 2. Measure how quickly one of the reactants (e.g. the zinc) disappears. So, we could measure the loss in mass of the zinc and hydrochloric acid (in grams) as they change into hydrogen gas escaping from a beaker every time period (e.g., every 10 seconds). Collision Theory How different variables change the rate of reaction is called the collision theory. For a reaction to take place, the particles of the substances that are reacting have to collide. If they collide with enough energy then they will react. The minimum amount of kinetic (movement) energy that two particles need if they are going to react when they collide is called the Activation Energy. There are two main ways of increasing the rate of a reaction: 1. Increase the number of collisions, and 2. Increase the amount of movement (kinetic) energy so that more collisions lead to a reaction. What can change the rate of a reaction? The four main variables are: 1. Concentration 2. Temperature 3. Size of the particles (i.e., surface area) 4. Catalyst being present The Effect of Concentration The concentration of a solution is the solution “strength”. Calcium carbonate + Hydrochloric acid —> Calcium chloride + Water + Carbon dioxide A stronger acid contains more acid particles and less water particles than a weaker acid. By increasing the concentration of a solution, more collisions will occur, and thus, the rate of the reaction goes up. NOTE: Changing the concentration of the acid does not change how quickly the particles are moving. That is, it does not increase the amount of energy they have. The Effect of Temperature When the temperature at which a reaction is taking place is increased, the particles move more quickly. This has two effects. More collisions take place, and when a collision occurs, there is more chance that the collision will lead to a reaction because the amount of energy is more likely to be greater than the minimum amount of energy needed (i.e., activation energy) The Effect of Particle Size Solids with a smaller particle size (e.g., powders or small chips) react more quickly than solids with a larger particle size (e.g., large chips). If the size of the particles is reduced, the rate of a reaction will increase because the surface area available for collisions to take place has increased. The increased surface area increases the number of collisions. NOTE: Surface area has no effect on the energy of the particles. Adding a Catalyst A catalyst is a substance that speeds up a reaction without being used up itself. Hydrogen peroxide decomposes to form water and oxygen gas. Hydrogen peroxide —> Water + Oxygen This reaction only occurs very slowly. If the compound manganese oxide is added, it acts as a catalyst for the reaction. When the catalyst is added the reaction speeds up greatly but the manganese oxide never runs out. How does a catalyst work? 1. A catalyst provides a surface on which the reaction can take place. This increases the number of collisions between the particles of the substances that are reacting. 2. A catalyst lowers the activation energy (the minimum amount of energy needed for a reaction to take place). Thus, the particles can react with less energy. If the amount of energy needed for particles to react is lowered, more particles can react. Chemical Reactions - Demonstrations Reactants Products Observations Iron Sulfur Iron sulfide Black, solid mass Magnesium Hydrochloric acid Magnesium chloride Hydrogen Vigorous bubbling Magnesium disappears Calcium carbonate Hydrochloric acid Calcium chloride Water Carbon dioxide Bubbling Calcium carbonate disappears Sodium Water Sodium hydroxide Hydrogen Sodium floats and moves on water Sodium fizzes Iron Copper sulfate Iron sulfate Copper A red-brown colour appears around the iron The blue solution turns colourless Sulfur Oxygen Sulfur dioxide In a gas jar, fumes or pungent gas appear Magnesium Oxygen Magnesium oxide Blinding white flame White ash left