NiSix-Si Core-Shell Nanowires with Contact Engineering as Anodes
advertisement

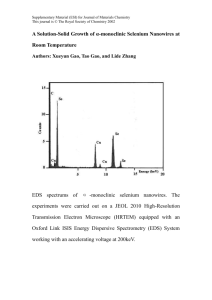
NiSix-Si Core-Shell Nanowires with Contact Engineering as Anodes for High-Rate Lithium-Ion Batteries Xiang Han, version 2, 29/03/2015 Abstract Introduction Silicon can store Li+at a capacity 10 times that of graphite anodes [1-3]. Core-shell silicon nanowire is a promising candidate for the anode material in high-rate lithium-ion batteries due to the core nanowires provide concrete mechanical supports and fast electron conductive pathways [4-6]. However, to harness this remarkable potential for high power density energy storage, one has to address the contact problem between the core and silicon core. Here, we present that… Batteries containing…exhibited improved performance… The excellent electrochemical performance is directly correlated with Experimental Fabrication of NiSix-Si core-shell nanowires To fabricate NiSi-Ge+Si core-shell NWs on Ni foam current collector, a simple two-step method was performed as following: (1) CVD growth of self-catalytic NiSix NWs on Ni foam. (2) Sputtering of Ge+Si on the NiSix NWs. Firstly, the nickel foam with a diameter of 14 mm was ultrasonically cleaned and rinsed with diluted oxalic acid (0.1 wt%) and acetone. The cleaned nickel foam was then transfeferred into a CVD tube furnace. After that, the tube was heated to 570 ℃ under the atmosphere of 30 sccm H2 with a rate of 5 /min, then 5 sccm SiH4 was introduced into the tube and kept for 60 min. After naturally colling the tube, the NiSi NWs on Ni foam was transfeferred into a radio frequency magnetron sputtering system using Ge and Si target with high purity of 99.99 %. Sputtering was performed at 200C with an Ar working pressure of 3 Pa and power of 80 W for 1h. The weight of the as-produced Si coating was calculated by measuring the supporting substrate on a microbalance (TB-25, 1 ug resolution) before and after. Characterization Electrochemical test Results and discussions Description of the synthesized NiSix-Si core-shell nanowires including morphology, structure, and elemental information… NiSix Si Ge Fig. 1 Schematic illustration of fabricating NiSix-Si core-shell nanowires. Figure 1 inllustrates the schematic of the fabrication process for network-like NiSi-Ge+Si core-shell nanowires. The synthesis process is detailed within the Experimental Section. The NiSix core nanowires was grown on Ni foam substrate under a SiH4 flow diluted in H in a chemical vapor deposition (CVD) reactors. Note that NiSix here is easy to synthesis, particularly on Ni foam without lithography []. The morphology of NiSix can be controlled by varing the SiH4 flow and growth time (Figure S1). In addition, CVD is a highly mature and industrial process, which is compatible with large area substrates []. Then, aGe and aSi were deposited by a RF-sputtering system. (a) (b) (c) 20 um (d) (e) Ge 100 nm Ni Si Fig. 2 Morphology characterization of NiSix-Si nanowires. Figure 2 shows the morphology and elemental composition of the nanowires. As shown in Figure 2a, the NiSi nanowires have random orientation and identical morphology on Ni foam substrate. Interestingly, the long NiSi nanowires twined from one to another Ni backbone and selfly cross-linked to construct free-standing network structure. Thus, the interconnect joints may provide a fast electron pathway. Figure 2b and Figure 2c show low magnification transmittance electron microscope (TEM) images of NiSix-Si and NiSix-Ge+Si core-shell nanowires, respectively. Both of the nanowires possess a dense black core a diameter of 20-30 nm, coated with and a gray shell (30-40 nm). With the same sputtering condition, brush-like Si grew sparsely on NiSix cores, but a condense uniform coating on the NiSix with Ge interlayer. A high resolution TEM image (HRTEM) of NiSix-Ge+Si NW was taken at the interface between the NiSi core and Si shell (Figure 2d), implying crystalline plane of NiSi core with a d-spacing of 0.34 nm. This value indicates a Ni3Si2 (002) plane. However, no crystalline plane was observed at the Ge/Si shell, revealing an amorphous material nature. A Ge layer with thickness of ~ 5 nm is inserted between NiSi and Si, however, there is no clear separation between the NiSi core and Si shell, confirming a condense interface. To accurately map the elemental composition of the NiSi-Ge+Si, energy dispersive X-ray (EDX) was taken across the whole nanowire (Figure 2e). The Ni image shows a signal in the middle of the nanowire, showing that the black core in the NW is composed of Ni. The Si image shows uniform Si signal in the middle core area and a stronger signal across the shell area. The Ni and Si images suggest the core is composed of Ni and Si and a uniform Si coating layer across the whole nanowire. The Ge image shows a continuous distribution across the core area a stronger signal at the interface of core and shell, suggesting an ultra-thin uniform Ge interlayer between the NiSi core and Si shell. The composition of the core–shell nanowires was further confirmed by the X-ray diffraction (XRD) patterns (Figure S2). All the diffraction peaks are attributed to Ni3Si2 and Ni2Si (PDF NO. ). No diffraction peaks of Ge/Si were observed. It is known that the aSi possesses of smaller volume changes than crystalline Si during Li+ insertion and extraction []. In addition, both theNiSix-Ge+Si and NiSix-Si nanowires were embedded in the Ni foam, which serves as electron conduction, as well as mechanical backbone. However, solely by inserting a Ge layer, the structure of Si shell changed from brush-like morphology to a uniform coating layer, which is expected to stabilize the electrochemical performance. On the other hand, the Ge interlayer may help to enhance the rate performance due to its excellent lithium-ion diffusivity (400 times faster than in Si), high electrical conductivity (104 times higher than Si) Description and explanation of the electrochemical performance… Fig. 3 Cyclic voltammetry (CV) of the core-shell nanowires. Fig. 4 Cycling performance of the core-shell nanowires. Fig. 5 Mechanism analysis… Fig. 6 Fig. 7 Conclusions References 1. M. Armand, J. M. Tarascon, Nature 2008, 451, 652. 2. B. Kang, G. Ceder, Nature 2009, 458, 190. 3. Kang, B., & Ceder, G. Battery materials for ultrafast charging and discharging. Nature 2009, 458, 190-193. 4. Fan, Y., Zhang, Q., Lu, C., Xiao, Q., Wang, X., & kang Tay, B. High performance carbon nanotube–Si core–shell wires with a rationally structured core for lithium ion battery anodes. Nanoscale, 2013, 5, 1503-1506. 5. Nguyen, H. T., Zamfir, M. R., Duong, L. D., Lee, Y. H., Bondavalli, P., Pribat, D. Alumina-coated silicon-based nanowire arrays for high quality Li-Ion battery anodes. J. Mater. Chem. 2012, 22, 24618-24626. 6. Cao, F. F., Deng, J. W., Xin, S., Ji, H. X., Schmidt, O. G., Wan, L. J., & Guo, Y. G. (2011). Cu‐Si Nanocable Arrays as High‐Rate Anode Materials for Lithium‐Ion Batteries. Adv. Mater. 2011, 23, 4415-4420. Acknowledgments