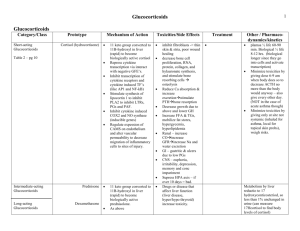
Cortisol in Psychiatry

Cortisol in Psychiatry
5HTP raises serum cortisol, more in manic and depressed persons (Meltzer 1984)
Amos T, Stein DJ, Ipser JC. Pharmacological interventions for preventing post-traumatic stress disorder
(PTSD). Cochrane Database Syst Rev. 2014 Jul 8;7:CD006239.
AUTHORS' CONCLUSIONS: There is moderate quality evidence for the efficacy of hydrocortisone for the
prevention of PTSD development in adults. We found no evidence to support the efficacy of propranolol, escitalopram, temazepam and gabapentin in preventing PTSD onset. The findings, however, are based on a few small studies with multiple limitations. Further research is necessary in order to determine the efficacy of pharmacotherapy in preventing PTSD and to identify potential moderators of treatment effect.
Aurilio C, Ceccarelli I, Pota V, Sansone P, Massafra C, Barbarisi M, Pace MC, Passavanti MB, Bravi F,
Aloisi AM. Endocrine and behavioural effects of transdermal buprenorphine in pain-suffering women of different reproductive ages.Endocr J. 2011 Dec 27;58(12):1071-8.
Chronic pain is a common problem in clinical practice and women are affected more often than men. Morphine is often used for long-term pain relief, but it induces side effects including endocrine alterations. The aim of the present study was to assess the behavioural and hormonal effects of transdermal buprenorphine in women suffering from persistent non-malignant pain. Hormones (LH, FSH, total and free testosterone, estradiol, cortisol) and pain measures (visual analogue scale, McGill Pain questionnaire, present pain intensity test) were evaluated at baseline and after 1, 3 and 6 months. Subjects were recruited in the Second University of Naples Pain Research Centre.
Eighteen chronic pain women were included in the study, divided into pre- and post-menopausal groups. A transdermal buprenorphine patch (Buprenorphine TDS, 35 µg/h) was administered every 72 h. As expected, buprenorphine administration led to a decrease in pain intensity and no side effects suggestive of hypogonadism were recorded. Pain measures decreased at the first control visit (T1) in both groups. Total and free testosterone were not reduced by treatment (they tended to increase in both groups) while cortisol progressively recovered from
the quite low levels detected at the beginning of treatment. These data confirm that buprenorphine is a safe and effective drug for pain relief in women. It is free from the adverse effects on gonadal hormones frequently associated with other opioid treatments. The lack of opioid-induced effects on gonadal hormones (i.e. hypogonadism) is important to guarantee safe long-term pain treatment. PMID: 21937837
Berardelli R, Margarito E, Ghiggia F, Picu A, Balbo M, Bonelli L, Giordano R, Karamouzis I, Bo M,
Ghigo E, Arvat E. Neuroendocrine effects of citalopram, a selective serotonin re-uptake inhibitor, during lifespan in humans. J Endocrinol Invest. 2010 Oct;33(9):657-62.
OBJECTIVE: Serotonergic system contributes to the regulation of hypothalamus-pituitary-adrenal axis. In humans, serotonergic agonists increase PRL, ACTH, and cortisol, while serotonin (5HT) influence on GH is controversial.
Central 5HT activity and neuroendocrine function change during lifespan. DESIGN: To clarify the neuroendocrine response to 5HT across lifespan, we assessed ACTH, cortisol, DHEA, PRL, and GH responses to citalopram (CT) in young adults (YA) (no.=12, 29.2±1.7 yr mean±SEM), middle aged (MA) (no.=12, 54.3±0.9 yr), and elderly (ES)
(no.=12, 69.3±0.9 yr) males. All the subjects received placebo (saline iv over 120 min) or CT (20 mg iv over 120 min). Blood samples were taken every 15 min up to 240 min. RESULTS: During placebo, ACTH, cortisol, GH, and
PRL were similar in all groups while DHEA showed an age-dependent reduction from middle age (p<0.001).
During CT, ACTH, and cortisol were higher than during placebo in YA (p<0.05) and even more in MA (p<0.01 vs placebo, p<0.05 vs YA); in ES, the increase of both ACTH and cortisol (p<0.05 vs placebo) was lower than in
MA (p<0.05) and higher than in YA (p<0.05 for cortisol only). No changes were observed for DHEA, GH, and PRL in any group. CONCLUSIONS: Corticotrope response to CT is age-dependent in normal men, being amplified starting from middle age, suggesting precocious changes in the serotonergic neuroendocrine control during lifespan. CT is a useful tool to evaluate the age-dependent serotonergic function in humans. PMID: 20414043
Cone EJ, Johnson RE, Moore JD, Roache JD. Acute effects of smoking marijuana on hormones, subjective effects and performance in male human subjects. Pharmacol Biochem Behav. 1986
Jun;24(6):1749-54.
Four healthy male subjects smoked two marijuana cigarettes or one marijuana cigarette and one placebo cigarette, or two placebo cigarettes on separate days in a random order crossover design. Each marijuana cigarette contained
2.8% delta-9-tetrahydrocannabinol (THC). Plasma hormones and THC were measured before and after each smoking session. Plasma LH was significantly depressed and cortisol was significantly elevated after smoking marijuana. Nonsignificant depressions of prolactin, FSH, testosterone and free testosterone and elevation of GH also occurred. Concurrent measures of subjective effects via subscales of the Addiction Research Center Inventory,
Single Dose Questionnaire and a Visual Analog Scale were generally elevated. Significant impairment on a psychomotor performance task paralleled elevations in subjective effects, hormone effects and peak THC determinations. Although all the hormone effects were within normal basal ranges, interactions between these systems, and their effects on behavior cannot be discounted. PMID:3016764
Fehm HL, Holl R, Steiner K, Klein E, Voigt KH. Evidence for ACTH-unrelated mechanisms in the regulation of cortisol secretion in man. Klin Wochenschr. 1984 Jan 2;62(1):19-24.
In an attempt to elucidate the significance of ACTH independent mechanisms in the regulation of cortisol secretion in man, the dynamics of plasma ACTH and cortisol levels were studied in response to different stimuli. The cortisol response to small amounts of exogenous ACTH and to insulin induced hypoglycemia was preceded by an increase in
ACTH levels appropriate to explain the increase in cortisol. In contrast, after administration of methamphetamine, there was an increase in cortisol levels in the absence of any changes in ACTH concentrations. Apparently, the methamphetamine induced cortisol secretion was not mediated by radioimmunoassayable ACTH. A diurnal rhythm was observed for the responses to hypoglycemia and to methamphetamine with larger cortisol responses in the evening as compared to the forenoon. These changes were not accompanied by parallel changes in the ACTH responses. From these differences, additional evidence is provided for the importance of ACTH independent mechanisms in the regulation of cortisol secretion. PMID: 6321849
Girdler SS, Pedersen CA, Straneva PA, Leserman J, Stanwyck CL, Benjamin S, Light KC. Dysregulation of cardiovascular and neuroendocrine responses to stress in premenstrual dysphoric disorder. Psychiatry
Res. 1998 Nov 16;81(2):163-78.
Twelve women with prospectively confirmed premenstrual dysphoric disorder (PMDD or PDD) were compared with 12 healthy control subjects for cardiovascular and neuroendocrine responses to speech and mental arithmetic
(Paced Auditory Serial Addition Task) stressors during both the follicular and luteal phases of the menstrual cycle.
Structured clinical interview was used to assess psychiatric and abuse histories, and standardized questionnaires were administered to assess current life stress. Results revealed that PMDD women had significantly lower stroke
volume, cardiac output and cortisol levels but significantly elevated norepinephrine and total peripheral resistance at rest and also during mental stressors compared with control subjects. These effects were evident in both cycle phases. Significantly more women with PMDD had histories of sexual abuse, and they also reported greater current life stress than control subjects. Consistent with a history of trauma, the PMDD women exhibited significantly greater ratios of norepinephrine to cortisol at rest and during stress. These results are interpreted as reflecting dysregulation of the stress response and may be related to histories of severe and/or chronic exposure to stress for a subgroup of PMDD women. PMID: 9858034
Girdler SS, Leserman J, Bunevicius R, Klatzkin R, Pedersen CA, Light KC. Persistent alterations in biological profiles in women with abuse histories: influence of premenstrual dysphoric disorder.Health
Psychol. 2007 Mar;26(2):201-13.
OBJECTIVE: To examine dysregulation in biological measures associated with histories of abuse in women and whether women with premenstrual dysphoric disorder (PMDD) differ in their dysregulation. DESIGN: Twenty-five women meeting prospective criteria for PMDD and 42 non-PMDD controls underwent structured interview to
determine abuse histories and lifetime Axis I diagnoses, excluding those with current Axis I disorders or using medications. MAJOR OUTCOME MEASURES: Plasma cortisol and norepinephrine (NE), heart rate (HR), blood pressure (BP), and vascular resistance index (VRI) were assessed at rest and in response to mental stress.
RESULTS: A greater proportion of PMDD women had prior abuse compared with non-PMDD women. Regardless of PMDD status, all abused women had lower plasma NE and higher HRs and tended to have lower plasma
cortisol at rest and during stress. Abused women also reported more severe daily emotional and physical symptoms.
Greater VRI and BP at rest and during stress were seen only in PMDD women with abuse. CONCLUSION: There is persistent dysregulation in stress-responsive systems in all abused women that cannot be accounted for by current psychiatric illness or medications, and PMDD women may be differentially more vulnerable to the impact of abuse on measures reflecting alpha-adrenergic receptor function. PMID: 17385972
Harris DS, Baggott M, Mendelson JH, Mendelson JE, Jones RT.Subjective and hormonal effects of 3,4methylenedioxymethamphetamine (MDMA) in humans.Psychopharmacology (Berl). 2002
Aug;162(4):396-405.
RATIONALE: 3,4-Methylenedioxymethamphetamine (MDMA) is a widely used phenethylamine. Reports have described the effects of MDMA in a controlled laboratory setting, but the full range of effects of MDMA in humans is still not completely characterized. OBJECTIVES: To describe the physiological, subjective, and hormonal changes after single doses of MDMA in a laboratory setting and examine relationships between these effects.
METHODS: Eight MDMA-experienced volunteers each received placebo, 0.5 mg/kg, and 1.5 mg/kg oral doses of
MDMA in a double-blind crossover study. RESULTS: The 1.5 mg/kg dose (comparable to that typically used by most participants) produced significant subjective effects, peaking at about 2 h after dosing, including some effects commonly associated with stimulant drugs, hallucinogens, and entactogens. MDMA significantly increased plasma cortisol, prolactin, and dehydroepiandrosterone (DHEA) levels. Increase in plasma cortisol after the 1.5 mg/kg dose correlated with increased heart rate, rate-pressure product, and drug liking. Rise in DHEA correlated with euphoria. CONCLUSIONS: A typically used dose of MDMA produced effects commonly associated with stimulants and hallucinogens. Subjects liked MDMA. Correlations between cortisol and DHEA levels and some physiological and psychological effects are consistent with animal data suggesting that hormones modulate some responses to drugs of abuse. PMID: 12172693
Jacobs D, Silverstone T, Rees L. The neuroendocrine response to oral dextroamphetamine in normal subjects.Int Clin Psychopharmacol. 1989 Apr;4(2):135-47.
In 24 male subjects oral dextroamphetamine 20 mg caused a statistically significant rise in cortisol, prolactin, TSH,
FSH, and LH compared to placebo. Dextroamphetamine caused a short-lived rise in growth hormone and attenuated the later rise which occurred after placebo. The findings are discussed in the light of previous reports of the neuroendocrine response to dextroamphetamine in normal subjects and in psychiatric patients. PMID: 2501383
Meltzer HY, Umberkoman-Wiita B, Robertson A, Tricou BJ, Lowy M, Perline R. Effect of 5hydroxytryptophan on serum cortisol levels in major affective disorders. I. Enhanced response in depression and mania. Arch Gen Psychiatry. 1984 Apr;41(4):366-74.
The serum cortisol concentration following administration of 5-hydroxytryptophan (5-HTP), 200 mg orally, a precursor of serotonin (5-HT), was significantly greater in unmedicated depressed and manic patients than in normal controls. Increases in serum cortisol levels greater than 5 micrograms/dL were significantly more frequent in both unmedicated depressed and manic patients than in the normal controls. There was significant test-retest reliability. Baseline serum cortisol concentration correlated negatively with the cortisol response to 5-HTP in normal controls. These results suggest increased 5-HT receptor sensitivity may be present, possibly in the hypothalamus or pituitary, in some patients with affective disorders. These results are consistent with the hypothesis that decreased serotonergic activity, which would be expected to produce increased 5-HT receptor sensitivity, may be present in both depression and mania. PMID: 6608335
Meltzer HY, Lowy M, Robertson A, Goodnick P, Perline R. Effect of 5-hydroxytryptophan on serum cortisol levels in major affective disorders. III. Effect of antidepressants and lithium carbonate. Arch Gen
Psychiatry. 1984 Apr;41(4):391-7.
Serum cortisol levels were significantly increased following administration of 5-hydroxytryptophan (5-HTP), 200 mg orally, to patients with affective disorders. A three- to five-week period of treatment with lithium carbonate or monoamine oxidase (MAO) inhibitor augmented the mean 5-HTP-induced increase in serum cortisol
concentration in manic or depressed patients, respectively: tricyclic antidepressant (TCA) and second-generation antidepressant treatment diminished the mean serum cortisol response in patients with major depression. These results are consistent with the hypothesis that lithium carbonate may enhance serotonin (5-HT) receptor
sensitivity, whereas TCA and second-generation antidepressants diminish 5-HT receptor sensitivity. The enhancement of the cortisol response to 5-HTP by MAO inhibitors may be due to decreased metabolism of 5-HT. It is important to assess the effect of thymoleptic drug treatment on various responses to biogenic amine precursors or agonists in patients rather than laboratory animals. PMID: 6422900
Morris MC, Compas BE, Garber J., Relations among posttraumatic stress disorder, comorbid major depression, and HPA function: a systematic review and meta-analysis. Clin Psychol Rev. 2012
Jun;32(4):301-15.
Exposure to traumatic stress is associated with increased risk for posttraumatic stress disorder (PTSD) and alterations of hypothalamic-pituitary-adrenocortical (HPA) function. Research linking traumatic stress with HPA function in PTSD has been inconsistent, however, in part due to (a) the inclusion of trauma-exposed individuals without PTSD (TE) in control groups and (b) a failure to consider comorbid major depressive disorder (MDD) and moderating variables. This meta-analysis of 47 studies (123 effect sizes, N=6008 individuals) revealed that daily cortisol output was lower for PTSD (d=-.36, SE=.15, p=.008) and PTSD+MDD (d=-.65, SE=.25, p=.008) groups relative to no trauma controls (NTC); TE and NTC groups did not differ significantly from each other.
Afternoon/evening cortisol was lower in TE (d=-.25, SE=.09, p=.007) and PTSD (d=-.27, SE=.12, p=.021) groups and higher in PTSD+MDD groups (d=.49, SE=.24, p=.041) relative to NTC. Post-DST cortisol levels were lower in
PTSD (d=-.40, SE=.12, p<.001), PTSD+MDD (d=-.65, SE=.14, p<.001), and TE groups (d=-.53, SE=.14, p<.001) relative to NTC. HPA effect sizes were moderated by age, sex, time since index event, and developmental timing of trauma exposure. These findings suggest that enhanced HPA feedback function may be a marker of traumaexposure rather than a specific mechanism of vulnerability for PTSD, whereas lower daily cortisol output may be
associated with PTSD in particular. PMID: 22459791
Murphy LL, Muñoz RM, Adrian BA, Villanúa MA. Function of cannabinoid receptors in the neuroendocrine regulation of hormone secretion. Neurobiol Dis. 1998 Dec;5(6 Pt B):432-46.
Marijuana and its cannabinoid constituents have profound effects on anterior pituitary hormone secretion.
Exposure to delta 9-tetrahydrocannabinol inhibits gonadotropin, prolactin, growth hormone, and thyroidstimulating hormone release and stimulates the release of corticotropin. Consequently, cannabinoid exposure could have profound effects on the function of the reproductive system, lactation, metabolism, and on the endocrine stress axis. The acute effects of cannabinoids on the endocrine system are consistent with its actions on brain neurotransmitter systems involved in the regulation of neuropeptides that modulate anterior pituitary hormone secretion. Although cannabinoid receptors appear to play a major role in the ability of cannabinoids to influence hormone release, much remains to be learned concerning their function in the neuroendocrine regulation of hormone secretion. PMID: 9974176
Pariante CM. Risk factors for development of depression and psychosis. Glucocorticoid receptors and pituitary implications for treatment with antidepressant and glucocorticoids. Ann N Y Acad Sci. 2009
Oct;1179:144-52.
Increased levels of glucocorticoid hormones-the main product of the hypothalamic-pituitary-adrenal (HPA) axishave been considered to be "depressogenic," but this notion has largely derived from studies in patients with endocrine conditions, such as Cushing's syndrome or exogenous treatment with synthetic glucocorticoids. In these
conditions, it is likely that the full impact of the high glucocorticoid levels is felt on the brain, through overstimulation of the glucocorticoid receptors (GRs); indeed, normalizing these high levels leads to an improvement of mood in these patients. However, a completely different mechanism may be operating in major depression, where the increased levels of glucocorticoid hormones are conceptualized as driven by an impairment in GR function
(glucocorticoid resistance), and therefore as a "compensatory" mechanism. Moreover, clinical and experimental studies have shown that antidepressants increase GR function, thus leading to resolution of glucocorticoid resistance. Interestingly, a number of studies have also demonstrated that manipulating GR function with both agonists and antagonists has an antidepressant effect, and indeed that other drugs targeting the HPA axis and cortisol secretion-even drugs with opposite effects on the HPA axis-have antidepressant effects. These studies do not support the notion that "high levels of glucocorticoids" always have a depressogenic effect, nor that decreasing the effects of these hormones always has an antidepressant effects. PMID: 19906237
Rabin DS, Schmidt PJ, Campbell G, Gold PW, Jensvold M, Rubinow DR, Chrousos GP. Hypothalamicpituitary-adrenal function in patients with the premenstrual syndrome. J Clin Endocrinol Metab. 1990
Nov;71(5):1158-62.
Patients with primary affective disorders, such as melancholic depression and anorexia nervosa, frequently have a hyperactive hypothalamic-pituitary-adrenal (HPA) axis, characterized by hypersecretion of CRH and a blunted
ACTH response to exogenous CRH. Premenstrual syndrome (PMS) is a luteal phase dysphoric disorder characterized by primarily affective and behavioral disturbances. HPA axis function was compared in PMS patients and control women, respectively, diagnosed by DSM3-R criteria or found to have no current psychiatric disorders, determined by the Schedule for Affective Disorders and Schizophrenia-Lifetime Interview. Urinary free cortisol excretion was the same in PMS and normal women, and no differences in urinary free cortisol excretion between the follicular and luteal phases occurred in either group. Two HPA axis abnormalities, however, were noted when PMS patients were compared to normal women. First, basal evening cortisol concentrations in plasma were significantly decreased, while the time-integrated response of plasma cortisol to ovine (o) CRH was significantly increased.
Second, the negative correlation between time-integrated plasma ACTH and cortisol responses to oCRH and basal luteal progesterone concentrations present in normal control women was not seen in the PMS patients. These changes in basal and oCRH-stimulated plasma cortisol levels in association with normal urinary free cortisol excretion suggest that women with PMS might have transient or episodic disturbances of their HPA axis, which appear adequately corrected by this system's servomechanisms. This probably explains the maintenance of regular menstrual cycles in PMS patients, which contrasts with the irregular menses observed in patients with depression, anorexia nervosa, or women who participate in chronic strenuous exercise. PMID: 2172272
Ranganathan M, Braley G, Pittman B, Cooper T, Perry E, Krystal J, D'Souza DC. The effects of cannabinoids on serum cortisol and prolactin in humans. Psychopharmacology (Berl). 2009
May;203(4):737-44.
BACKGROUND: Cannabis is one of the most widely used illicit substances, and there is growing interest in the therapeutic applications of cannabinoids. While known to modulate neuroendocrine function, the precise acute and chronic dose-related effects of cannabinoids in humans are not well-known. Furthermore, the existing literature on the neuroendocrine effects of cannabinoids is limited by small sample sizes (n = 6-22), heterogeneous samples with regard to cannabis exposure (lumping users and nonusers), lack of controlling for chronic cannabis exposure, differing methodologies, and limited dose-response data. Delta-9-tetrahydrocannabinol (Delta-9-THC) was hypothesized to produce dose-related increases in plasma cortisol levels and decreases in plasma prolactin levels.
Furthermore, relative to controls, frequent users of cannabis were hypothesized to show altered baseline levels of these hormones and blunted Delta-9-THC-induced changes of these hormones. MATERIALS AND METHODS:
Pooled data from a series of laboratory studies with multiple doses of intravenous Delta-9-THC in healthy control subjects (n = 36) and frequent users of cannabis (n = 40) was examined to characterize the acute, chronic, and acute on chronic effects of cannabinoids on plasma cortisol and prolactin levels. Hormone levels were measured before (baseline) and 70 min after administration of each dose of Delta-9-THC. Data were analyzed using linear mixed models with +70 min hormonal levels as the dependant variable and baseline hormonal level as the covariate. RESULTS: At socially relevant doses, Delta-9-THC raised plasma cortisol levels in a dose-dependent manner but frequent users showed blunted increases relative to healthy controls. Frequent users also had lower
baseline plasma prolactin levels relative to healthy controls. CONCLUSIONS: These group differences may be related to the development of tolerance to the neuroendocrine effects of cannabinoids. Alternatively, these results may reflect inherent differences in neuroendocrine function in frequent users of cannabis and not a consequence of cannabis use. PMID: 19083209
Seyler LE Jr, Fertig J, Pomerleau O, Hunt D, Parker K. The effects of smoking on ACTH and cortisol secretion. Life Sci. 1984 Jan 2;34(1):57-65.
The relationship among changes in plasma nicotine, ACTH, and cortisol secretion after smoking were investigated.
Ten male subjects smoked cigarettes containing 2.87 mg nicotine and 0.48 mg nicotine. No rises in cortisol or
ACTH were detected after smoking 0.48 mg nicotine cigarettes. Cortisol rises were significant in 11 of 15 instances after smoking 2.87 mg nicotine cigarettes, but ACTH rose significantly in only 5 of the 11 instances where cortisol increased. Each ACTH rise occurred in a subject who reported nausea and was observed to be pale, sweaty, and tachycardic. Peak plasma nicotine concentrations were not significantly different in sessions when cortisol rose with or without ACTH increases, but cortisol increases were significantly greater in nauseated than in non-nauseated smokers. Our data suggest that smoking-induced nausea stimulates cortisol release by stimulating ACTH secretion and that cortisol secretion in non-nauseated smokers may occur through a non-ACTH mechanism. It is not clear whether nicotine or some other stimulus inherent in smoking is responsible for cortisol secretion without ACTH secretion. PMID: 6319934
Torpy DJ, Grice JE, Hockings GI, Crosbie GV, Walters MM, Jackson RV.
The effect of desipramine on basal and naloxone-stimulated cortisol secretion in humans: interaction of two drugs acting on noradrenergic control of adrenocorticotropin secretion. J Clin Endocrinol Metab.
1995 Mar;80(3):802-6.
Desipramine (DMI), a tricyclic antidepressant and norepinephrine (NE) reuptake blocker, is reported to induce
ACTH and cortisol release acutely in humans, probably by facilitating central NE neurotransmission. Tricyclic antidepressant therapy, including DMI, normalizes the ACTH and cortisol hypersecretion that often accompanies depression. The mechanism of hypothalamic-pituitary-adrenal (HPA) axis inhibition by DMI in humans is unknown.
In rats, DMI reduces the activity of the locus ceruleus, a major source of NE innervation of the hypothalamic paraventricular nucleus, the site of CRH neurons. Naloxone induces ACTH and cortisol release in humans through a noradrenergic-mediated mechanism and a probable consequent stimulation of hypothalamic CRH
release. To study the interaction of these drugs on NE neurotransmission and, hence, HPA axis activity in humans, we administered DMI alone and with naloxone in a randomized, double blind, placebo-controlled protocol in eight healthy male volunteers. DMI (75 mg, orally) was given 180 min before naloxone (125 micrograms/kg BW, i.v.).
Plasma ACTH and cortisol were measured at frequent intervals from 60 min before to 120 min after naloxone treatment. Plasma cortisol levels were 77% higher 180 min after DMI compared to those after placebo treatment
(287 +/- 17 vs. 162 +/- 14 nmol/L; P = 0.000005). DMI reduced the naloxone-induced rise in cortisol (P = 0.02), but there was no change in the integrated cortisol response. The increase in basal plasma ACTH levels after DMI treatment did not reach statistical significance. DMI significantly increased systolic blood pressure and heart rate consistent with an effect on the noradrenergic control of the cardiovascular system. In summary, DMI increased basal cortisol levels consistent with facilitation of NE neurotransmission and, hence, hypothalamic CRH release.
However, DMI had no enhancing effect on naloxone-induced cortisol release. This contrasts with the synergy observed when non-antidepressant agents that increase NE neurotransmission are given with naloxone to humans.
DMI increases glucocorticoid feedback sensitivity in the rat HPA axis after several weeks through up-regulation of central corticosteroid receptors. However, this slowly developing effect is unlikely to occur during these acute studies. The effect of DMI on naloxone-induced cortisol release is consistent with an inhibitory effect on central noradrenergic control of ACTH release, perhaps at the locus ceruleus. This is the first human study to suggest an inhibitory effect of DMI on central noradrenergic control of ACTH release. PMID: 7883833
Wilkins JN, Carlson HE, Van Vunakis H, Hill MA, Gritz E, Jarvik ME. Nicotine from cigarette smoking increases circulating levels of cortisol, growth hormone, and prolactin in male chronic smokers.
Psychopharmacology (Berl). 1982;78(4):305-8.
Results of this study indicate that nicotine from cigarette smoking increases circulating levels of cortisol, growth hormone, and prolactin in male chronic smokers. Previous studies have not addressed the question of whether the stimulus for smoking-related hormone release is the 'stress' of smoking or a pharmacologic action of nicotine and other tobacco substrates. Nicotine exposure is controlled in this study by allowing each subject to smoke only two
2.0 mg nicotine cigarettes during one experimental session and two 0.2 mg nicotine cigarettes in another session.
Plasma levels of cortisol, growth hormone, and prolactin for the higher nicotine session were found to be significantly elevated over those for the low-nicotine session, indicating that nicotine itself plays a predominate role in smoking-induced hormone increases. All hormone levels for the 2.0 mg nicotine session had not returned to baseline 60 min after smoking. PMID: 6818588