honchap8notes
advertisement

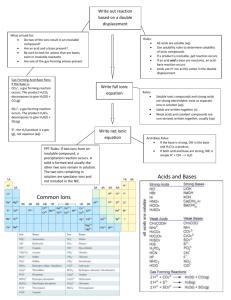
Honors Chemistry Mr. Fedell Chapter 8 Note Packet (teacher edition) Chapter 8 Problems: see handout! good figures in the book to look at.... The word on the street is that looking at illustrations, tables, and figures in the book is a good idea. 8.1 Describing Chemical Reactions chemical equations describe chemical reactions reactants chemical equation: word equation: say H2 + “Hydrogen plus Cl2 Chlorine yields products 2 HCl Hydrogen Chloride” other example - creation of sodium chloride Showing Phases in Chemical Reactions: gas = (g) or liquid = (l) solid = (s) aqueous (aq) ex: NaCl (s) = solid sodium chloride NaCl (aq) = solution of sodium chloride (in water) H2O (l) - liquid water (not aqueous - redundant) other symbols: = heat one arrow = one way reaction cat = catalyst* 2 arrows ↔ = reversible reaction = precipitate (solid - only found on products side) * a substance that speeds up a reaction without being used up in the reaction Balancing Chemical Equations: truly a trial and error process if there ever was one Helpful hints: 1. 1 atom at a time 2. Balance atoms that appear only 1X per side first 3. Balance polyatomic ions as whole units 4. Balance diatomic elements last 5. Save H + O for last if this doesn’t succeed, try doubling everything (particularly with combustion) Showing Energy Changes in Equations: 1 A + B C + heat ( - ∆H ) A + B + heat C ( + ∆H ) OR… exothermic - releases heat – 1 way to represent this: endothermic - put in heat – 2 ways to represent this: Interpreting Chemical Equation - what do chemical equations really mean anyway? H2O H2 + O2 (not balanced) 2 H2O 2 H2 + O2 (equal) 6 atoms = 4 atoms + 2 atoms (equal) 36.0 g = 4.0 g + 32.0 g (equal) 2 molecules ≠ 2 molecules + 1 molecules (not equal - doesn’t have to) 2 moles ≠ 2 moles + 1 mole (not equal - doesn’t have to) 8.2 Types of Chemical Reactions 5 types - synthesis, decomposition, combustion, single replacement, double replacement remember – “First you’ve got to find the right products, then you gotta balance!” 1. Synthesis (direct combination) - needs energy to happen general formula (element + element): A + B Ba + S Mg + Cl2 Al + Cl2 Na + O2 AB 2. Decomposition (analysis) - needs energy to happen (usually heat or electricity) general formula (molecule + element): FeCl3 HgO MgSO4 . 7 H2O AB A + B (hydrate) 3. Combustion - the reaction of hydrocarbons and oxygen to yield... “when you just say CHO,...” 2 general formula: ex. CxHy + O2 CO2 + H2O combustion of CH4, C3H8, C4H10, C2H5OH 4. double replacement – take place in aqueous solution - little energy involved - usually forms one soluble ionic product (aka - aqueous) and either a ppt, water, or a gas that bubbles out of water. general formula AB + CD AD + CB . FeCl3 + NaOH H2SO4 + NaOH NH4Cl + NaOH NH3 (g) + H2O(l) + NaCl(aq) 3. Single Replacement - take place in aqueous solutions and need very little energy Type 1: positive ions switch general formula: AB + M HI + Mg AlCl3 + Ca Ca + HOH MB + A (compound + metal) Type 2: negative ions switch general formula: AB + X NaCl + F2 BaS + O2 AX + B (compound + nonmetal) Hey, some reactions happen and some don’t… 3 For synthesis, combustion, and decomposition, we will assume they all happen given sufficient activation energy (Ea). For single replacement, use the activity series! Ca + H2O Al + H2O Al + HI Cu + HI Rules for the single replacement activity series: 1. 2. 3. 4. Any single element above an element in a compound will replace it. The top 5 elements react with water. Metals above H react with acids (molecules that start with H – not water). The nonmetal reactivity series is FClBr... For double replacement reactions, use a solubility table and the following rules: 1. If one of the products formed is water, the reaction happens. 2. If a gas is formed, the reaction happens. 3. If an insoluble product forms (I or Ss), the reaction happens (actually a reaction may happen when two soluble products form, but it doesn’t go to completion and is not directly observable). Na2CrO4 + KCl FeCl3 + KOH Yes! The reaction happens! NR (both products soluble) Fe(OH)3(ppt) + KCl(aq) Not balanced yet… “first you gotta find the right products,…” Note: precipitate symbols include (ppt), (s), and . NIB - Reactions in Aqueous Solution Ions in water solutions form aqueous solutions. 4 Ionic compounds are solids. When you put them in water, they dissociate: Ex1: NaBr(s) + Ex2: MgCl2(s) H2O(l) + Na+1(aq) + Br-1(aq) 2Cl-1(aq) Mg+2(aq) + H2O(l) Also happens with some molecular substances - particularly acids: we call it ionization Ex3: HCl(g) + Ex4: H2CrO4(s) H+1(aq) H2O(l) + H2O(l) 2H+1(aq) + Cl-1(aq) + CrO4-2(aq) Writing Ionic Equations so far we’ve looked at molecular equations now it’s time for ionic equations - show what happens to the ions in a reaction. first find products, balance, look up solubilities - remember water is a liquid, write net ionic, then total ionic, identify spectators..... Fe(s) (“molecular” equation) + (total ionic) Fe(s) + 2H+1(aq) + (net ionic) Fe(s) + 2H+1(aq) spectator ion = F- (“molecular” equation) (total ionic) (net ionic) 2HF(aq) 2F-1(aq) Fe+2(aq) FeF2(aq) + + Fe+2(aq) H2(g) + 2F-1(aq) + H2(g) H2(g) Spectator ions are ions that undergo no change in a chemical reaction. H2SO4(aq) + 2NaOH(aq) Na2SO4(aq) + 2H(OH)(l) 2H+1(aq) + SO4-2(aq) + 2Na+1(aq) + 2OH-1(aq) 2Na+1(aq) + SO4-2(aq) + 2H(OH)(l) 2H+1(aq) + 2OH-1(aq) spectator ions = SO4-2(aq) 2H(OH)(l) and Na+1(aq) Conservation of Charge - charges must balance in a chemical reaction. NIB - Solubility Trends 5 Cations - very soluble very insoluble- Na, K, ammonium Ag, Pb, Hg, transitions Anions - very soluble for monotomics very insoluble sulfides nitrate FClBr .... carbonate, hydroxide, phosphate, sulfate decompose general trend: As size decreases, solubility increases. Some helpful notes on writing phases in chemical reactions: 1. Metals are solids (except ) 2. In single and double replacement reactions, reactants that are compounds are always aqueous. 3. In single and double replacement reactions, products that are compounds should have their phases identified using a solubility chart (aqueous vs. precipitate) 4. In synthesis and decomposition reactions, ionic compounds are solids. 5. In combustion reactions, the water, CO2, and O2 are gases. The hydrocarbon is hard to tell, but is usually a liquid after C=5 or higher. 6. Most other covalent compounds are gases. 7. Acids (chemicals starting with hydrogen) are always aqueous. 6