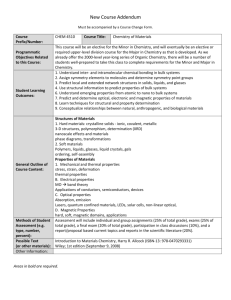
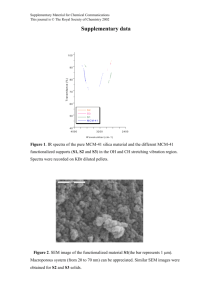
here - Hadi Nur
advertisement

COURSE OUTLINE Department & Faculty: Department of Chemistry, Faculty of Science Page : 1 of 4 Course Code: Solid State Chemistry (SSC 3473) Total Lecture Hours: 42 hours Semester: I Academic Session: 2009/2010 Lecturer Prof. Madya Dr. Hadi Nur Room No. Telephone No. IIS – 206-09 07-5534319 E-mail hadi@kimia.fs.utm.my Synopsis : What is solid state chemistry? An introduction to simple crystals structures: Close packing, Body-centred and primitive structures, Symmetry, Lattices and unit cells, Crystalline solids, Lattice energy. X-ray Diffraction: Generation of X-rays, Diffraction of X-rays, Powder diffraction, Solving single crystal structures. Preparative methods: Ceramic methods, Microwave synthesis, The sol-gel methods, Precursor method, Hydrothermal methods, Chemical vapour deposition. Bonding in solids and electronic properties: Bonding in solids – band model, Electronic conductivity – simple metals, Semiconductors – silicon and germanium, Doped semiconductors, Bands in compounds. Defects and non-stoichiometry: Defects and their concentration, Ionic conductivity in solids, Solid electrolytes, Non-stoichiometric compounds, Electronic properties of non-stochiometric oxides. Characterization of Inorganic Solids: Application of Physical Techniques. Optical properties of solids. Magnetic and dielectric properties of materials. Phase diagram and its interpretation. Relationship between structure, physicochemical and mechanical properties of materials. Zeolites and related structures. LEARNING OUTCOMES By the end of the course, students should be able to: Programme Learning Outcome(s) Addressed Assessment Methods LO1. describe the fundamental principles and concepts in the field solid state chemistry. PO1 Q, T, F LO2. demonstrate the ability to carry out calculations involved in solving single crystal structure and phase composition in phase diagram. PO2 Q, T, F LO3. recognize the physicochemical and mechanical properties based on structure of the materials PO1, PO2 Q, T, F LO4. apply the fundamental principles of structure of materials to explain the physicochemical and mechanical properties of materials. PO3, PO4 Q, T, F, A Course Learning Outcome Prepared by: Name: Prof. Madya Dr. Hadi Nur Signature: Date: 24 November 2008 T – Test ; Q – Quiz; F – Final Exam, A - Certified by: (Course Panel Head) Name: Dr. Che Rozid Mamat Signature: Date: 24 November 2008 COURSE OUTLINE Department & Faculty: Department of Chemistry, Faculty of Science Page : 2 of 4 Course Code: Solid State Chemistry (SSC 3473) Total Lecture Hours: 42 hours Semester: I Academic Session: 2008/2009 Assignment) STUDENT LEARNING TIME Teaching and Learning Activities Student Learning Time (hours) 1. Lecture 2. Independent Study - self learning - information search - library search - reading - group discussion 3. Assignment (2X) - self learning - group discussion 4. Test (1X) 5. Quiz (4X) 6. Exam 42 62 Total 2 1 3 120 TEACHING METHODOLOGY Lecture and Discussion, Independent Study, Group Project Assignment. WEEKLY SCHEDULE Week 1 10 : - What is solid state chemistry? An introduction to simple crystals structures o Close packing. o Body-centred and primitive structures. o Symmetry. o Lattices and unit cells o Crystalline solids o Lattice energy COURSE OUTLINE Department & Faculty: Department of Chemistry, Faculty of Science Page : 3 of 4 Course Code: Solid State Chemistry (SSC 3473) Total Lecture Hours: 42 hours Semester: I Academic Session: 2008/2009 Week 2 Quiz 1 : - X-ray Diffraction. o Generation of X-rays o Diffraction of X-rays o Powder diffraction o Solving single crystal structures Week 3 : - Preparative methods. o Ceramic methods. o Microwave sysnthesis o The sol-gel methods o Precursor method o Hydrothermal methods o Chemical vapour deposition Week 4 Quiz 2 : - Bonding in solids and electronic properties o Bonding in solids – band model o Electronic conductivity – simple metals o Semiconductors – silicon and germanium o Doped semiconductors o Bands in compounds Week 5 Assignment 1 : - Defects and non-stoichiometry o Defects and their concentration o Ionic conductivity in solids o Solid electrolytes. o Non-stoichiometric compounds o Electronic properties of non-stochiometric oxides Week 6 Test 1 : - Characterization of Inorganic Solids: Application of Physical Techniques – part 1 Week 7 : Week 8 : - Characterization of Inorganic Solids: Application of Physical Techniques – part 2 Week 9 Quiz 3 : - Optical properties of solids Week 10 : - Magnetic and dielectric properties of materials Mid-Semester Break COURSE OUTLINE Department & Faculty: Department of Chemistry, Faculty of Science Page : 4 of 4 Course Code: Solid State Chemistry (SSC 3473) Total Lecture Hours: 42 hours Semester: I Academic Session: 2008/2009 Assignment 2 Week 11 Quiz 4 : - Phase diagram and its interpretation – part 1 Week 12 : - Phase diagram and its interpretation – part 2 Week 13 Test 2 : - Relationship between structure, physicochemical and mechanical properties of materials – part 1. Week 14 - Relationship between structure, physicochemical and mechanical properties of materials – part 2. Week 15 : Week 16 : Week 17 : Study week. Week 18 : Final Examination. - Structure and properties of nanomaterials Zeolites and related structures. MAIN REFERENCES : 1. Solid State Chemistry An introduction, Lesley Smart and Elaine Moore, 1995, Chapman & Hall. 2. Solid State Chemistry and It’s Applications, Anthony R. West, 1984, John Wiley & Sons 3. Introduction to ceramics, W. D. Kingery, H. K. Bowen, D. R. Uhlmann. 1976, John Wiley & Sons. GRADING: No 1 2 3 4 Assessment Assignments Quizzes Test Final Exam Overall Total Number 2 4 1 1 % each 5 5 30 40 % total 10 20 30 40 100 Date Week 5, 10 Week 2, 4, 9, 11 Week 6, 14 Week 18