Activity Series of Metals Lab: High School Chemistry
advertisement
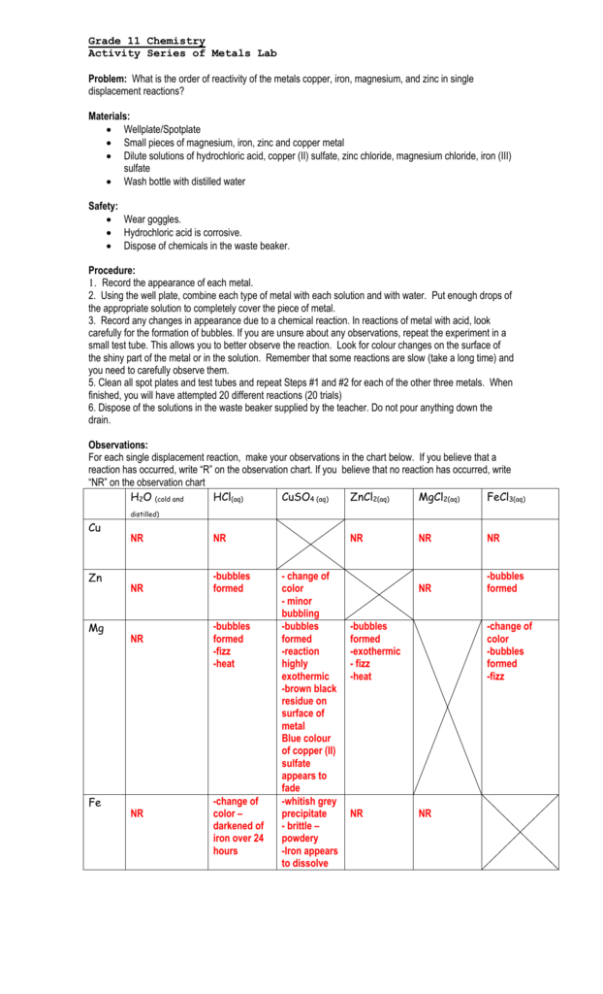
Grade 11 Chemistry Activity Series of Metals Lab Problem: What is the order of reactivity of the metals copper, iron, magnesium, and zinc in single displacement reactions? Materials: Wellplate/Spotplate Small pieces of magnesium, iron, zinc and copper metal Dilute solutions of hydrochloric acid, copper (II) sulfate, zinc chloride, magnesium chloride, iron (III) sulfate Wash bottle with distilled water Safety: Wear goggles. Hydrochloric acid is corrosive. Dispose of chemicals in the waste beaker. Procedure: 1. Record the appearance of each metal. 2. Using the well plate, combine each type of metal with each solution and with water. Put enough drops of the appropriate solution to completely cover the piece of metal. 3. Record any changes in appearance due to a chemical reaction. In reactions of metal with acid, look carefully for the formation of bubbles. If you are unsure about any observations, repeat the experiment in a small test tube. This allows you to better observe the reaction. Look for colour changes on the surface of the shiny part of the metal or in the solution. Remember that some reactions are slow (take a long time) and you need to carefully observe them. 5. Clean all spot plates and test tubes and repeat Steps #1 and #2 for each of the other three metals. When finished, you will have attempted 20 different reactions (20 trials) 6. Dispose of the solutions in the waste beaker supplied by the teacher. Do not pour anything down the drain. Observations: For each single displacement reaction, make your observations in the chart below. If you believe that a reaction has occurred, write “R” on the observation chart. If you believe that no reaction has occurred, write “NR” on the observation chart H2O (cold and HCl(aq) CuSO4 (aq) ZnCl2(aq) MgCl2(aq) FeCl3(aq) distilled) Cu Zn Mg Fe NR NR NR -bubbles formed NR NR -bubbles formed -fizz -heat -change of color – darkened of iron over 24 hours NR - change of color - minor bubbling -bubbles formed -reaction highly exothermic -brown black residue on surface of metal Blue colour of copper (II) sulfate appears to fade -whitish grey precipitate - brittle – powdery -Iron appears to dissolve NR NR NR -bubbles formed -bubbles formed -exothermic - fizz -heat NR -change of color -bubbles formed -fizz NR Analyze and Interpret 1. a) Write a chemical equation for each single displacement reaction recorded in your observation table. Include the predicted placement of lithium in your ranking. b) Rank the metals you tested in order of reactivity, beginning with the most reactive metal. Include lithium in your ranking. Keep in mind that hydrogen is a diatomic element. ***If no reaction occurs, write NR. Use the chart provided below or develop one of your own. Metals that displace other metals) Mg + ZnCl2 -> MgCl2 + Zn 3Mg + 2FeCl3 -> 3MgCl2 + 2Fe Mg + CuSO4 -> MgSO4 + Cu Zn + CuSO4 -> ZnSO4 + Cu 3Zn + 2FeCl3 -> 3ZnCl2 + 2Fe Fe + CuSO4 -> FeSO4 + Cu Or 2Fe + 3CuSO4 -> Fe2(SO4)3 + 3Cu Metals that Displace hydrogen from acids Metals that displace hydrogen from cold water Zn + 2HCl -> ZnCl2 + H2 n/a (lithium however should and to a small extent maybe magnesium) Mg + 2HCl -> MgCl2 + H2 Fe + 2HCl -> FeCl2 + H2 Or 2Fe + 6HCl -> 2FeCl3 + 3H2 Activity Series (list metals below) Most Reactive (rank) Li Mg Zn Fe Cu Least Reactive The reactivity of the metals in decreasing order would be lithium, magnesium, zinc, iron, and copper. Lithium would be the most reactive metal because it can react with water and magnesium can’t. Magnesium is second highest because it reacted with every compound except for water. Zinc is lower than magnesium because no reaction occurred between zinc and magnesium chloride. Zinc is higher than iron because zinc reacted with iron (lll) chloride, but iron did not react with zinc chloride. Copper is the least reactive metal because it did not react with any of the compounds. This means that copper would be below all other metals in the activity series. 1. Lithium metal reacts with water. a) Is lithium more or less reactive than magnesium? Yes, lithium is more reactive the magnesium because its above magnesium on the activity series. b) Write a balanced chemical equation to represent the reaction of lithium and water. 2Li(s) + H2O(l) -> Li2O(s) + H2(g) c) Would the resulting product be acid or basic? How can you hypothetically test if it was an acid or base? (suggest at least 2 different ways) The product is an acid because it is an aqueous solution containing hydrogen ions. Therefore, we would predict its PH to be less than 7 and can verify this using a pH meter or a pH natural or universal indicator. To test the acidity of the product, you can use litmus paper to test the pH level of the solution or add a known base and see if it reacts and neutralizes – creating a salt and water. if you use red litmus paper in a product if it turns blue then it is a base, if you use blue litmus paper in a product and it turns red, then it is an acid. You can also use a Phenolphthalein, if it is an acid it would remain colourless, if it is a base it would turn a pink or violet colour. A Bromothymol blue is also available, if it is tested in a base it’ll remain blue, if it is tested in an acid it’ll turn yellow. 2. Explain how the activity series can be used to predict single displacement reactions. Consult your textbook. The activity series can be used to predict the occurance of single displacement reactions by analyzing which elements are more reactive or less reactive than others. Morea reactive elements on the activity series would displace another element below of it. The activity series for metals as well as a noble gases have been explored. Understanding if a reaction would occur would allow for construction of safe building materials, building tools and operational machinery, just to name a few. 3. Given that lithium reacts with water and that magnesium does not, do you expect lithium to react with hydrochloric acid? If so, write a balanced chemical reaction. Yes, Lithium will react with hydrochloric acid. It will strip the electron e from hydrogen atom. 2Li + 2HCl (aq) → H2 + 2LiCl 4. Many people use copper piping to route water through their homes. Explain why magnesium or aluminum could not be used. People choose to use copper piping to route water through their homes because copper does not react with the water or acid residues contained in the pipes (i.e. resistant to corrosion). Copper is lower in the activity series than hydrogen, making it less reactive, therefore it wouldn’t be a single displacement reaction. If the pipes were made of Magnesium and aluminum (both more reactive metals) it would be significantly more dangerous. Magnesium burns in steam to produce white magnesium oxide and hydrogen gas. Pure magnesium can also react with water to produce magnesium hydroxide which is insoluble in water and would clog the pipes and potentially lead to high pressure area with hydrogen buildup, potentially a flood or fire hazard (since hydrogen gas is highly flammable). Magnesium will do this since its higher than hydrogen in the activity series, and would create a single displacement reaction. Aluminum would create Aluminum hydroxide as well as hydrogen gas, resulting in a similar scenario as would magnesium. This makes it unsafe to route water to homes, since aluminum and magnesium would react, where as it is safe to use copper since it is less reactive than hydrogen and below it on the activity series. Copper is also a very ductile metal and has very good heat and electrical conductivity. Also, copper is soft and easily bent. 5. From the following displacement reactions, arrange these hypothetical chemicals in order of decreasing activity (most to least reactive): A + B+ A+ + B (Since A displaces B, this indicates that A > B) C + B+ no reaction (Since there is no reaction, B >C) D + C+ D+ + C (Since D displaces C, D >C) F + G+ no reaction (Since there is no reaction, G >F) A + F+ no reaction (Since there is no reaction, F >A) F + D+ F+ + D (F displaces D, therefore F>D) D + B+ no reaction (no reaction means B>D) G + A+ G+ + A (G displaces A, therefore G>A) Most reactive to least reactive: G> F>A>B>D>C 6. Did you see bubbles produced during any of the reactions? From your balanced chemical equations, you can see that gas is not a product in all of these single displacement reactions. What would account for the presence of bubbles? Bubbles suggest that there may have been another reaction taking place within the solution. The bubbles were more visible and bigger in the reactions that took place with magnesium, the most reactive of the elements we used in the lab. The formation of bubbles is an indication of the chemical reaction taking place. When the bubbles stop, it is an indication that the chemical reaction has finished. The bubbles are accounted from the magnesium granules being forced to react with the solvent (water) causing hydrogen to be displaced. Since the magnesium granules were very small, it had a large available surface area which increased the virulence of the reaction with the metal salt as well as the solvent itself. Since the metal salt did not contain the hydrogen, the hydrogen gas produced must be a result of magnesium extracting the hydrogen from a water molecule. . Since magnesium primarily reacts with hot water and steam, which it was able to do by the already exothermic nature of the reaction with the metallic salt. This hot water (which was in small volume) was easily generated by the exothermic and vigorous reaction of magnesium and zinc chloride or iron (III) chloride. For example: Primary reaction: magnesium turnings are added to a solution of iron(III) chloride. Mg(s) + Fe3+(aq) => Mg2+(aq) + Fe2+(aq) Seocndary reaction: magnesium + water ==> magnesium hydroxide + hydrogen Mg(s) + 2H2O(l) ==> Mg(OH)2(aq/s) + H2(g) As more heat is generated in the reaction, the magnesium hydroxide and hydrogen gas are genererated a bit faster..