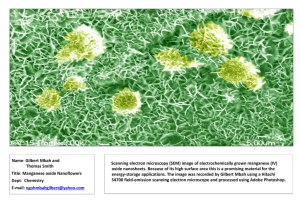
Electronic Supplementary Materal (ESM)
advertisement

Electronic Supplementary Materal (ESM) Preparation of protonated, two-dimensional graphitic carbon nitride nanosheets by exfoliation, and their application as a fluorescent probe for trace analysis of Cu (II) Xinrong Guo 1, Yong Wang 1, Fangying Wu 1, Yongnian Ni 1*, Serge Kokot 2 1 College of Chemistry, Nanchang University, Nanchang 330031, China 2 School of Chemistry, Physics and Mechanical Engineering, Science and Engineering Faculty, Queensland University of technology, Brisbane 4001, Australia Experimental Apparatus X-ray diffraction (XRD) measurements were recorded with the use of a Bede D1 high-resolution X-ray diffractometer (Bede Co., UK); the voltage was set to 40 kV and the current was 30 mA, and the Cu sources were K1=1.544398 and K2=1.544398. Transmission electron microscopy (TEM) images were obtained with the use of a JEM-2010 instrument (JEOL Co., Japan), and the accelerating voltage was 200 kV. Other operational settings for point and linear resolutions were set at 0.23 nm and 0.14 nm, respectively. X-ray photoelectron spectroscopy (XPS) measurements were made on a VG ESCALAB-MKII spectrometer, (VG Scientific Ltd., UK), which utilised Al Kα radiation. Fourier transform infrared spectra were collected with the aid of a Thermo Nicolet FT-IR fluorescent 380 spectrometer (Thermo Electron Scientific Instruments Co., Madison, USA). UV-vis absorption spectra were collected with an Agilent 8453 Spectrophotometer (Agilent Technologies, Santa Clara, CA, USA). The fluorescence measurements were obtained from an LS-55 Luminescence spectrometer (Perkin Elmer Co., MA, USA). The blue fluorescence of g-C3N4 nano-sheets solution was observed with the use of a ZF-2 UV lamp (Qilinbeier Instrumental Co., Haimen, China) set at 320 nm. Results and discussion Characterization of the g-C3N4 nanosheets The TEM images at high-magnification (Fig. S1A) supported the presence of sheet-like material and its layer structures were quite apparent. The wide-scan XPS spectra of bulk g-C3N4 and g-C3N4 nanosheets 1 displayed three peaks corresponding to elemental C, N and O (Fig. S1B). From the O1s spectrum of the bulk g-C3N4 sample (Fig. S1C), the two resolved peaks at approximately 532.0 and 533.3 eV were assigned to the N-C-O groups and the adsorbed O2 [1]. However, for g-C3N4 nanosheets, two peaks from the g-C3N4 nanosheets spectrum were associated with the binding energy shift at 531.6 and 532.8 eV. This observation was attributed to the N-C-O groups and the adsorbed oxygen, respectively [2]. This shift resulted from the presence of stronger covalent C-N bonds, and the higher frequencies were the result of an electrophilic effect of the O atoms adjacent to the C-N group. Figure S1 (A) Low–resolution TEM images of the dispersed g-C3N4 nanosheets. (B) XPS survey spectrum of bulk g-C3N4 and g-C3N4 nanosheets. (C) High–resolution peak–fitted XPS of the O1s regions of bulk g-C3N4 and g-C3N4 nanosheet samples. 2 Optical properties of g-C3N4 nanosheets From the UV-vis spectra (Fig. S2A), it was clearly evident that the absorption peak at 301 nm was associated with the g-C3N4 nanosheets. This band appeared to have blue shifted about 14 nm as compared to a similar band (315 nm) from the bulk g-C3N4 sample. Furthermore, the fluorescence spectrum (Fig. S2B) indicated that the emission peaks of the bulk g-C3N4 sample and the g-C3N4 nanosheets were located at 450 and 434 nm, respectively. Compared with the emission peak of bulk g-C3N4, it was also found that the band associated with the g-C3N4 nanosheets blue shifted about 16 nm when the excitation wavelength was 300 nm. This shift was probably associated with the O atoms on the surface of the g-C3N4 nanosheets [3]. Such UV-vis and fluorescence spectra from the g-C3N4 nanosheets could arise from the shifting, in opposite directions, of the CV and VB because they were affected by quantum confinement [3] (Fig. S2C). The fluorescence spectra of the g-C3N4 nanosheets had maxima at the excitation and emission wavelengths of 300 nm and 434 nm, respectively. The inset displayed a photo of the g-C3N4 nanosheets in solution, which were exposed to the visible light at 320 nm. The emission spectra of g-C3N4 nanosheets were also recorded progressively over the 280 to 350 nm excitation range at intervals of 10 nm (Fig. S2D), and it was found that the emission wavelengths were almost the same. This work indicated that the g-C3N4 nanosheets had an excitation-independent fluorescence behavior. The quantum yields (QY) of the bulk g-C3N4 and the g-C3N4 nanosheet samples were calculated with the use of the integrated fluorescence intensity (Fig. S2E) [4]. The quinine sulfate solution (0.1 M H2SO4) was chosen as the standard, which had a well - established value of QY, i.e. 54% [5]; the equation used for calculations was: QYtest = QYQS×(Stest/SQS)×(test2/QS2) where the subscripts “QS” and “test” denoted quinine sulphate and test material g-C3N4 ,respectively; S was the slope of the fluorescence intensity versus the absorbance plot, and was the refractive index of the solvent. As shown in Table 1 (see the paper), the QY of the bulk g-C3N4 samples, and the g-C3N4 nanosheets were calculated to be 5.99% and 10.3% respectively, at an excitation wavelength of 300 nm. 3 Figure S2 (A) UV-vis absorption spectra of bulk g-C3N4 and g-C3N4 nanosheets sampls. (B) Fluorescence spectra of bulk g-C3N4 and g-C3N4 nanosheets; excitation wavelength = 300 nm. (C) Excitation and emission spectra of g-C3N4 nanosheet samples. Inset: photos of UV irradiated (320 nm) g-C3N4 nanosheets solution. (D) Fluorescence spectra of g-C3N4 nanosheets excited at different wavelengths. (E) Linear plots of integrated FL intensity versus absorbance for three standard samples of quinine sulphate, bulk g-C3N4 and g-C3N4 nanosheets, respectively. 4 Optimization of the experimental conditions In this work, the g-C3N4 nanosheets were used as sensors for the detection of Cu2+. The experimental conditions, reaction time and pH of the medium, were investigated and optimized. A sample of g-C3N4 nanosheets together with 0.4 nmol L-1 Cu2+ was dissolved in a pH 7.0 B-R buffer (see section of “Detection of Cu2+ with the use of the g-C3N4 nanosheets”), and its fluorescence intensity was measured at 434 nm at 1 min intervals (Fig. S3A). It was found that after adding the Cu2+ solution, the fluorescence intensity was gradually quenched over a period of 0-10 min; thereafter, the fluorescence remained constant. Thus, a period of 10 min was chosen as the reaction time, and this was similar to a previously suggested time [6]. Also, these observations indicated that the quenching effect of Cu2+ on the fluorescence intensity emitted from g-C3N4, was quite rapid, and this then implied that a rapid and stable sensor was constructed. In this work, a series of B-R buffers with different pHs in the range of 2.0 - 12.0 (final concentration: 50 μmol L-1) were used as the medium to analyse Cu2+. It was shown (Fig S3 B), that, in general, the fluorescence intensity of the g-C3N4 nanosheets gradually decreased from pH 2.0 to 12.0, and it was also noted that the fluorescence was relatively stable from pH 7.0 to 12.0. Such results may be attributed to the presence of the terminal amino groups on the g-C3N4 nanosheets, and this facilitated an increase in the fluorescence intensity of the g-C3N4 nanosheets under acidic condition [7]. Figure S3 (A) Time-dependent (0-11 min) fluorescence intensity of the mixed solution containing 0.40 nmol L–1 Cu2+ and 0.1 mg mL−1 g-C3N4 nanosheets at 434 nm. (B) The fluorescence response of g-C3N4 nanosheets at different pH (B-R buffer, 0.1 M). 5 Calibration of g-C3N4 nanosheets-based sensor for Cu2+ Figure S4 (A) Fluorescence emission spectra of g-C3N4 nanosheet samples (0.10 mg mL−1) with different concentrations of Cu2+ (from top down: 0.00, 0.01, 0.02, 0.04, 0.06, 0.08, 0.10, 0.12, 0.14, 0.16, 0.18, 0.20, 0.22, 0.24, 0.26, 0.28, 0.30, 0.32, 0.36 and 0.40 nmol L–1). (B) calibration plot of F0 / F versus concentration of Cu2+. Selectivity study of the g-C3N4 nanosheets for detecting Cu2+ Selectivity of the method for the analysis Cu2+ with the use of g-C3N4 nano-sheets was investigated with the inclusion of potentially interfering metal ions: Al3+, Co2+, Ag+, Cd2+, Mg2+, Zn2+, Ba2+, Ca2+, Ni2+, Li+, Fe3+, Pb2+, and Hg2+. The concentration of each interfering ion was 0.4 nmol L–1, i.e. the same as that of Cu2+. The fluorescence intensity was quenched only by the added Cu2+ in the g-C3N4 solution (Fig. S5A); practically no fluorescence intensity change was detected in the presence of any one of the 13 different metal ions. Selectivity of Cu2+ (0.4 nmol L–1) was also investigated in the presence of above 13 metal ions (Fig. S5B) but at a higher concentration of the ions (8.0 nmol L–1). Only a very small interference effect was found in the presence of other metal ions in the mixture containing g-C3N4 nanosheets and Cu2+. The relative error was less than 5% in all cases and it was obtained for the concentration level specified. These results indicated that the detection of Cu2+ with the use of the method was practically unaffected by other metals. 6 Figure S5. (A) Results of a selectivity study: spectra of the g-C3N4 nano-sheet sensor (0.10 mg mL−1) in the presence of Cu2+ and other metal ions (pH = 7 B-R (0.1 mol L–1)); metal ion concentrations - all 0.4 nmol L–1. (B) An interference study of the g-C3N4 nano-sheets sensor (0.10 mg mL−1) for Cu2+ (0.4 nmol L–1) premixed with other metal ions (8.0 nmol L–1). References [1] Martin DJ, Qiu K, Shevlin SA, Handoko AD, Chen XW, Guo ZX, Tang JW (2014) Highly Efficient Photocatalytic H2 evolution from water using visible light and structure-controlled graphitic carbon nitride. Angew Chem Int Ed 53:1-7 [2] Li JH, Shen B, Hong ZH, Lin BZ, Gao BF, Chen YL (2012) A facile approach to synthesize novel oxygen-doped g-C3N4 with superior visible-light photoreactivity. Chem Commun 48:12017-12019 [3] Zhang SW, Li JX, Zeng MY, Xu JZ, Wang XK, Hu WP (2014) Polymer nanodots of graphitic carbon nitride as effective fluorescent probes for the detection of Fe 3+ and Cu2+ ions. Nanoscale 6:4157-4162 [4] Zhang K, Guo JK, Nie JJ, Du BY, Xu DJ (2014) Ultrasensitive and selective detection of Cu 2+ in aqueous solution with fluorescence enhanced CdSe quantum dots. Sens Actuat B: Chem 190:279-287 [5] Mitra S, Chandra S, Pathan SH, Sikdar N, Pramanik P, Goswami A (2013) Room temperature and solvothermal green synthesis of self passivated carbon quantum dots. RSC Adv 3:3189-3193 [6] Cheng NY, Jiang P, Liu Q, Tian JQ, Asiri AM, Sun XP (2014) Graphitic carbon nitride nanosheets: one-step, high-yield synthesis and application for Cu2+ detection. Analyst 139:5065-5068 [7] Rong MC, Lin LP, Song XH, Zhao TT, Zhao YX, Yan JW, Wang YR, Chen X (2015) A lable-free fluorescence sensing approach for selective and sensitive detection of 2,4,6-trinitrophenol (TNP) in aqueous solution using graphitic carbon nitride nanosheets. Anal Chem 87:1288-1296 7