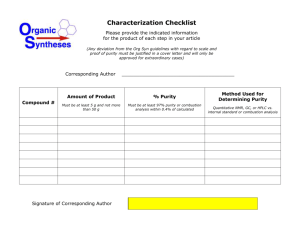
Project Summary - Wofford College
advertisement

An Investigation into the Purity of Commercially Available Creatine Monohydrate James Paul Atkinson Wofford College Total Requested: $150 12 March 2002 Project Summary This project aims to determine the purity of commercially available creatine monohydrate, which is currently taken by many athletes to improve performance. Creatine (Cr) supplements from name brands and generic brands will be analyzed using high-pressure liquid chromatography (HPLC) and infrared spectrometry (IR spec) to determine if the company label matches what is actually contained in the product. Other basic measurements such as mass and melting points will also be conducted. Creatine supplements that are found to be the purest will be stored under different environmental conditions and prepared for consumption in a variety of ways (see methods) to determine if either has an effect on the chemical properties. The control will also be stored under these conditions and analyzed. The expectations are that the company labels will be precise and that the storage and mixing will not have any negligible effect on the supplement. Justification Over the past fifteen years Cr supplementation among athletes has increased dramatically.5 Many companies have entered this new and expanding marketplace. Cr is considered a dietary supplement under the Dietary Supplement Health and Education Act of 1994. The Food and Drug Administration (FDA) regulates dietary supplements under a different set of regulations than conventional foods and drugs.9 This includes conventional foods and both prescription and over-the-counter drugs. The responsibility of ensuring that a supplement is safe before it is marketed is the sole responsibility of the manufacturer. 9 The FDA is only responsible for taking action against any manufacturer that markets an unsafe product. Manufacturers do not need to get FDA approval before selling their product. However, the FDA is responsible for adverse event reporting and product information, such as labeling. The Federal Trade Commission is responsible for regulating dietary supplement advertising.9 All of this leads to the potentially dangerous situation in which the manufacturer who is selling the product is also ensuring its purity and directing its proper use. Dr. Clark of Arizona State University found that several distributors of the dietary supplement dehydroepiandrosterone (DHEA) were selling products that did not contain what their labels read.10 This project aims to verify the manufacturer’s labels because there is a serious conflict of interest when the seller is the only one guaranteeing their product for purity. Background Chevreul (cited by 5) discovered Cr in 1835 and phosphorylcreatine (PCr), the phosphorylated and energy storing molecule was described in 1927.1,3,6 Creatine kinase (CK) was described a few afterward in 1934 along with its enzymatic reaction.1,3,6 PCr is used as a source of phosphate by CK to convert adenosine diphosphate (ADP) to adenosine triphosphate (ATP).6 (Fig. 1) This acts to buffer ATP levels in cells that are expending energy. Figure 1 Cr/PCr reaction catalyzed by CK.5 PCr can be hydrolyzed to generate ATP quickly compared to the synthesis of ATP by glycolysis and mitochondrial synthesis. The logic behind Cr supplementation is that if the level of PCr in muscle can be raised then the muscle cells would be able to attain higher tensions and maintain the tension longer over a short term. Cr supplementation of 20 – 30 g/day has been shown to increase muscle Cr and PCr levels in human muscles.7 Cr degrades nonenzymatically and irreversibly into creatinine (Crn) in vitro. 5 This is relevant to both storage and the mixing of Cr when used as a supplement. The Cr might be degrading to Crn in the bottle. Cr metabolism has been linked to certain neurological and muscular diseases and new evidence is showing that Cr may act as a precursor to some food mutagens. Cr supplementation may be used one day in treating some muscular diseases. 5 Athletes normally take 20-30 g/day of Cr for about a week to increase their muscle Cr levels and then 2-5 g/day to maintain their muscle Cr levels.5 Cr supplementation also significantly increases PCr levels.5 Methods Samples will be obtained from three commercial vendors and from a reputable chemical supply company. Creatine monohydrate from GNC, LifeLink, Nature’s Bounty and Wholesale-Creatine.com will be used. The control sample of Cr for this experiment will come from Sigma, a chemical supply company. Creatine monohydrate and creatinine will be purchased from Sigma. Cr degrades to Crn in vitro so Cr and Crn need to be obtained to test the commercial products for degradation. The melting points and masses of all the samples will be measured. Any deviation from accepted values could be an indication that the sample is impure. The melting point of Cr is 273o C The mass of the Cr tablet or powder will be compared with the label to evaluate the precision of the manufacturer’s measurement. It will be taken into account that some companies add inert ingredients such as magnesium stearate. Other companies enclose the Cr into gelatin capsules. These capsules will be removed for HPLC and IR spec analysis. An infrared spectrometer (IR spec analysis) will be used to determine the infrared signature of the samples of Cr and controls. IR spec analysis uses the fact that molecules and bonds vibrate and when an infrared light is shined on the compound, some of it is absorbed. The amount of light transmitted through the compound is detected and this produces a series of peaks, which correspond to particular wavelengths. This spectrum will be observed from each compound and compared to the control for Cr and Crn. At least 3 analyses of each compound will be performed to ensure accuracy. A Perkin Elmer 2000 FT-IR will be used to measure the infrared signature of the samples. A HPLC test will be performed as well. Chromatography is the process of differential migration of sample components where some fragments are selectively retained by the stationary phase.8 The stationary phase can be anything that does not move or dissolve in the solvent. A column will be filled with silica gel. Then, a mixture of methanol and water that has the sample dissolved in it will be slowly passed through the column. Certain components in the sample will have different affinities for the silica gel. This will cause them to move through the column at different speeds. Upon exiting the bottom of the column, a ultraviolet light is projected though the stream. Different components of the sample exiting the column at different times will produce peaks in absorption, which can be used to determine the relative proportion of the components in the sample. A Perkin Elmer 1022 with a diode ray detector 135c will be used to analyze the samples. About 0.5 g of sample will be analyzed in the HPLC. At least three analyses will be done of each sample. Commercial samples of Cr that have the highest purity of Cr will be stored at 0o C, 25o C, and 37o C for 2 weeks in the dark within a plastic container. This will simulate a short storage of the sample in different conditions that the consumer might store the sample in. The samples will also be mixed with different juices with differing pHs to simulate ways in which an athletes consume Cr . Orange juice, grape juice, and a shaved ice, fruit based mix known as a ‘smoothie’ will be used. Each sample of Cr will be mixed with the vehicle beverage and allowed to sit for either 10 minutes or 1 hour or 2 days. This will simulate an athlete preparing the mix and drinking it right away or waiting. The different juices will have there pH measured. These samples will then be analyzed with an IR spec and HPLC to determine if there is any degradation of Cr into Crn. Possible Complications The HPLC has not been used in some time. Starting the apparatus again could cause time delays. If the equipment fails to function alternate equipment will be searched for. Inert ingredients might complicate the HPLC or IR spec. In the event that the mixing of Cr with juice causes too much background to appear in the analysis, Cr will be mixed with water solution with HCL to simulate different pHs. Goals To precisely determine the purity of different Cr supplements To determine the effects of storage on Cr supplement composition and degradation. To determine the effects of different preparation methods on Cr supplements Budget $21.50 Cr from Sigma C3630 (100g) $9.50 Crn from Sigma C4255 (25g) $25.00 Cr from GNC (200g) $25.00 Cr from wholesale-creatine.com (500g) $25.00 Cr from LifeLink (500g) $25.00 Cr from Weider via Eckerd (200g) $19.00 Shipping and handling Total Requested- $150 *Shipping and handling has been estimated in because an order must be place before vendor can calculate the price. Equipment on hand Spatulas Glassware (beakers, flasks, etc.) Perkin Elmer 2000 FT-IR Perkin Elmer 1022 with a diode ray detector 135c (also included solvents and silica) Balance Refrigerators Incubators Thermometers Bibliography *Note-1&3 were cited in 5 1. Conway M, and Clark J. Creatine and Creatine Phosphate: Scientific and Clinical Perspectives. Orlando, FL: Academic, 1996. 2. Hadden N., Bauman F., MacDonald F., Munk M., Stevenson R., Gere D., Zamaroni F., Majors R. Basic Liquid Chromatography. USA: Varian Aerograph; 1971. 3. Saks V, Bobkov Y, and Strumia Eds E. Creatine Phosphate: Biochemistry, Pharmacology and Clinical Efficiency. Torino, Italy: Edizioni Minerva Medica, 1987. 4. Steenge G.R., Lambourne J., Casey, A., Macdonald I.A., and Greenhaff P.L. 1998. Stimulatory effect of creatine accumulation in human skeletal muscle. AJP – Endocrinology and Metabolism, 275: E974-E979. 5. Wyss M. and Kaddurah-Daouk R. Creatine and Creatinine Metabolism. 2000. Physiological Reviews, 80: 1107 – 1213. 6. Vander, Sherman and Luciano. Human Physiology. 8th edition. New York: McGrawHill. 795 p. 7. Vanderberghe V., Goris M., Van Hecke P., Van Leemputte M., Vangerven L., and Hespel P. 1997 Long-term creatine intake is beneficial to muscle performance during resistance training. Journal of Applied Physiology, 83: 2055 – 2063. 8. Yeung, E. editor. Detectors for Liquid Chromatography. New York: John Wiley & Sons, Inc.; 1986. 362 p. 9. [FDA] Food and Drug Administration. 2002 Feb. 26. FDA homepage. <http://vm.cfsan.fda.gov/~dms/supplmnt.html>. Accessed 2002 March 4. 10. [HEALL] Health Education Alliance for Life and Longevity, Clark, W. Dennis. 2000 DHEA Laboratory Analysis. <http://www.heall.com/products/vitamins/dhea/analysis.html>. Accessed 2002 March 12.