MGH antimicrobial susceptibilities
advertisement

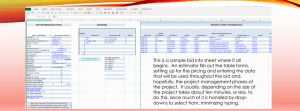
34. MGH antibiotic susceptibilities Chloramphenicol TMP - SMX Levofloxacin Rifampin Linezolid Nitrofurantoin* Tetracycline Erythromycin Clindamycin Vancomycin Cephalothin c Oxacillin Penicillin No of Strains Gram positive susceptibilities (Jan-Dec 2002) (% susceptible) 95 85 92 54 97 100 76 55 46 99 100 98 95 99 98 92 98 56 98 96 100 30 100 100 100 100 99 100 100 100 100 94 Staphylococcus aureus 2299 10 56 56 100 66 43 90 Coagulase-negative staphylococci 2855 12 35 35 100 61 35 85 Organism Staphylococcus saprophyticus 135 100 95 44 85 Staphylococcus lugdunensis 38 82 100 d 100 87 76 87 Streptococcus pneumoniae 245 75 100 92 78 82 63 100 100 100 94 87 86 -hemolytic streptococci (group A) 278 100 100 100 88 73 13 -hemolytic streptococci (group B) 44 100 100 100 93 73 31 -hemolytic streptococci (group C, G) 177 72 -hemolytic streptococcia 100 90 59 62 Streptococcus milleri groupb 42 98 100 83 86 81 Enterococci 2330 82 85 12 30 * Urine isolates only a Includes alpha-hemolytic streptococci, S. mitis, S. sanguis, S. salivarius, S. bovis, S. mutans b Includes alpha-hemolytic and beta-hemolytic S. milleri group c For Staph. spp. predicted from oxacillin; for streptococci predicted from penicillin d Determined by testing for the presence of mecA gene Piperacillin Cefazolin Cefpodoxime Ceftriaxone Ceftazidime Cefepime Aztreonam Meropenem Gentamicin Amikacin Levofloxacin TMP – SMX Chloramphenicol Tetracycline Nitrofurantoin* Acinetobacter calcoaceticusbaumanni complex Alcaligenes xylosoxidans Burkholderia cepacia complex Citrobacter freundii Citrobacter koseri (diversus) Enterobacter aerogenes Enterobacter cloacae Escherichia coli Haemophilus influenzae Klebsiella oxytoca Klebsiella pneumoniae Morganella morganii Proteus mirabilis Proteus vulgaris Pseudomonas aeruginosa Salmonella spp. Serratia marcescens Shigella spp. Stenotrophomonas maltophilia * Urine isolates only Ampicillin Organism No of Strains Gram negative susceptibilities (Jan-Dec 2002) (% susceptible) 283 0 0 0 0 0 21 24 9 35 34 72 29 44 24 0 50 43 152 96 177 378 5561 103 216 1199 79 476 32 1359 70 200 26 176 0 0 0 0 66 75 0 0 0 85 0 83 0 42 80 33 85 90 82 67 70 0 0 80 92 94 84 86 82 50 18 0 93 0 0 95 64 83 0 97 0 0 74 98 80 50 97 90 85 63 98 97 96 69 100 88 99 84 69 99 100 95 86 96 100 83 97 92 100 74 44 88 91 83 74 99 93 86 81 99 100 84 95 91 100 38 100 100 98 94 100 0 12 88 99 85 70 99 95 86 94 99 100 67 99 95 100 0 80 28 100 100 99 100 100 100 100 100 100 100 84 100 100 100 0 0 0 97 100 99 92 97 92 91 90 95 100 76 95 0 0 7 99 100 99 98 100 100 98 100 100 100 78 99 10 14 9 91 98 97 85 96 100 88 84 95 95 100 59 100 88 100 46 84 67 89 99 98 86 82 83 91 83 89 91 100 99 97 23 95 42 30 95 59 77 94 88 78 77 89 79 67 0 34 87 23 4 97 99 42 64 99 91 56 17 0 7 100 MGH Medical Housestaff Manual 96 86 100 89 100 84 100 100 100 86 35. HIV antiretroviral therapy Recommendations for initiation of antiretrovirals for HIV Asymptomatic infection: evaluate with history, physical exam, CBC, chemistry, LFTs, lipids, CD4+ count, Plasma HIV RNA (viral load), evaluation for opportunistic infections (OIs); CMV, syphilis, toxo, hepatitis serologies, PPD, CXR, Pap smear. Symptomatic disease (wasting, thrush, unexplained fever > 2 weeks) including AIDS: all patients should be offered treatment. Clinical Category Symptomatic (AIDS, severe symptoms) Asymptomatic AIDS Asymptomatic Asymptomatic CD4+ T cell count Any value Plasma HIV RNA Any value Recommendation Treat < 200/mm3 200-350/mm3 > 350/mm3 Any value Any value Treat Treatment offered; controversial Some would initiate therapy as 3-yr risk of AIDS >30%; others would monitor CD4+ count and RT-RNA frequently Many would defer therapy and observe as 3-yr risk of AIDS is <15% Treatment offered; ongoing clinical trials assess the benefit > 55,000 (bDNA or RT-PCR) Asymptomatic > 350/mm3 <55,000 (bDNA or RT-PCR) Acute HIV infection, within 6 months Any value Any value Goals of treatment Decrease in viral load (0.5-0.75 log10) within 4 weeks or VL (1 log) within 8 weeks Undetectable VL (<50 or <20 viral copies) at 4-6 months Restoration and/or preservation of immunological function Reduction of HIV-related morbidity and mortality Hospital management Assess patient's current regimen and adherence If adherent, and not admitted for adverse reactions (lactic acidosis, pancreatitis, hepatitis, rash, etc.) resume all antiretrovirals according to dosing schedule. Encourage patient to take own medicines to avoid delay from the pharmacy. When in ICU setting or if NGT is in place, Kaletra, ritonavir, and AZT can be given in liquid formulation. Other antiretroviral preparations may also be available in liquid formulation. Hold all antiretrovirals if patient had not been adherent or if suffering from an adverse reaction. May consider checking CD4+ count, RT-RNA VL, and resistance patterns prior to resuming current or prior to designing new regimen. MGH Medical Housestaff Manual 87 35. HIV antiretroviral therapy Generic drug Brand Typical dose Nucleoside Analog Reverse Transcriptase Inhibitors (NRTIs) 300 mg bid (available as Abacavir (ABC) Ziagen (Group A) 20 mg/mL oral solution) Zidovudine (AZT, ZDV) Retrovir (Group A) 200 mg tid 300 mg bid Stavudine (d4T) Zerit (Group A) Lamivudine (3TC) Epivir (Group B) >60 kg: 40 mg bid <60 kg: 30 mg bid 150 mg bid Didanosine (ddI) Videx (Group B) Zalcitabine (ddC) Hivid (Group B) AZT + 3TC Combivir (NRTI combo) Trizivir (NRTI combo) AZT + 3TC + ABC >60 kg: 200 mg bid, 250 mg bid, or 400 mg qd <60 kg: 125mg bid, 167 mg bid, or 250 mg qd 0.75 mg tid Adverse effects Hypersensitivity reaction: fever, rash, nausea, vomiting, malaise, fatigue, loss appetite; lactic acidosis; SOB Bone marrow suppression; anemia; neutropenia; GI intolerance; HA, insomnia; lactic acidosis Pancreatitis, peripheral neuropathy; lactic acidosis with hepatic steatosis Minimal toxicity; lactic acidosis with hepatic steatosis Pancreatitis; peripheral neuropathy; nausea, diarrhea; lactic acidosis with hepatic steatosis 1 tablet bid Peripheral neuropathy; stomatitis; lactic acidosis As per AZT and 3TC 1 tablet bid As above Non-nucleoside Reverse Transcriptase Inhibitors (NNRTIs) Delavirdine Rescriptor 400 mg tid Efavirenz Sustiva 600 mg qhs Nevirapine Viramune 200 mg bid Amprenavir Agenerase 1200 mg bid (cap) 1400 mg bid (soln) <50 kg: 20 mg/kg Indinavir Crixivan 800 mg q8h Nelfinavir Viracept Ritonavir Norvir 1250 mg bid (with meals) 750 mg tid (with meals) 600 mg q12h (with meals) Saquinavir Invirase 400 mg bid with ritonavir Rash; elevated LFTs; headache; inhibits P450 Rash; CNS effects; hepatitis; mixed incuder/inhibitor P450 Rash; elevated LFTs; hepatitis; induces P450 Protease inhibitors MGH Medical Housestaff Manual GI into, nausea, vomiting, diarrhea, rash, oral parathesias, inc. LFTs, diabetes, fat redistribution, lipid abnormalities Nephrolithiasis, GI intolerance, nausea, headache, increased LFTs, diabetes, fat redistribution Diarrhea, nausea, diabetes, fat redistribution, lipid abnormalities GI intol, nausea, vomiting, diarrhea, parathesias, hepatitis, pancreatitis, increased triglycerides; fat redistribution, diabetes GI intol, nausea, diarrhea, headache, 88 35. HIV antiretroviral therapy Lopinavir + Ritonavir Fortovase Kaletra (PI combo) 1200 mg tid (with meals) 400 mg lopinavir + 100 mg ritonavir bid elevated LFTs, fat redistribution, DM GI intol, nausea, vomiting, diarrhea, asthenia, inc. LFTs, diabetes, fat redistribution Nucleotide Reverse Transcriptase Inhibitors Tenofovir Same 300 mg qd No renal toxicity; limited expanded access Recommended regimens One choice from group A and one from group B Mono and dual therapies are not recommended. Hydroxyurea not currently recommended with NRTIs Class sparing regimens: NNRTI +2 NRTIs or triple NRTIs or PI + 2 NRTIs Consultation with HIV specialist recommended prior to initiating therapy Group A: efavirenz, indinavir, nelfinavir, ritonavir + indinavir, ritonavir + lopinavir (Kaletra), ritonavir + saquinavir (soft-gel capsule or hard-gel capsule) Group B: didanosine + lamivudine, stavudine + didanosine, stavudine + lamivudine, zidovudine + didanosine, zidovudine + lamivudine Recommended alternatives Group A: abacavir, amprenavir, delavirdine, nelfinavir + saquinavir-SGC, nevirapine, ritonavir, saquinavir-SGC Group B: zidovidine + zalcitabine Potentially fatal adverse reactions Lactic acidosis and steatohepatitis. Associated with “d” drugs, NRTIs, tenofovir, and abacavir. All antiretrovirals should be stopped and not restarted if this reaction is suspected. Fatal hypersensitivity reactions. Associated with abacavir. Symptoms include fever, skin rash, fatigue, GI symptoms, dyspnea, pharyngitis, cough. Should not be rechallenged with abacavir as death may ensue in hours. Propylene glycol toxicity. Found in the amprenavir oral solution. Contraindicated in children < 4 yo, pregnant women, patients with renal or hepatic failure, patients using metronidazole or disulfiram. Pancreatitis. Associated with didanosine, zalcitabine. Reference See also http://www.aidsinfo.nih.gov Meg Doherty, M.D., Ph.D. MGH Medical Housestaff Manual 89 36. Prophylaxis for opportunistic infections in HIV Prophylaxis to prevent first episode of OIs in adults and adolescents. Pathogen Pneumocystis carinii Preventive Regimens Indication First Choice Alternatives CD4+ count <200 or oropharyngeal candidiasis TMP-SMX 1 DS qd or Dapsone 50 mg bid or TMP-SMX 1 SS qd dapsone 100 mg qd or dapsone 50 mg + pyrimethamine 50 mg + leucovorin 25 mg qw or aero. pentamidine 300 mg qmonth. Mycobacterium tuberculosis (INH sensitive) PPD >5 mm or contact with active case INH 300 mg qd + B6 50 mg qd x 9 mo. or Rifampin 600 mg qd x 4 mo. or INH 900 mg + B6 100 mg biweek x 9 mo. rifabutin 300 mg qd x 4 mo. pyrazinamide 15-20 mg/kg qd + rifampin or rifabutin x 2 mos Mycobacterium tuberculosis (INH resistant) PPD > 5 mm or contact with active case with high prob INH resistance Rifampin 600 mg qd or rifabutin 300 mg qd x 4 mo. Pyrazinamide 15-20 mg/kg qd + rifampin or rifabutin x 2 mos Mycobacterium tuberculosis (multidrug resistant) PPD > 5 mm or contact with active case with high prob multidrug resistance Choice of drugs requires consultation with public health authorities; depends on resistance pattern Toxoplasma gondii IgG Ab + Toxo and CD4+ <100 TMP-SMX 1 DS qd TMP-SMX 1 SS qd or dapsone 50 mg qd + pyrimethamine 50 mg qw + leucovorin 25 mg qw or dapsone 200 mg + pyrimethamine 75 mg qw + leucovorin 25 mg qw or atovaquone 1500 mg qd + pyrimethamine 25 mg qd + leucovorin 10 mg qd Mycobacterium avium complex CD4+ <50 Varicella zoster (VZV) Exposure to VZV VZIG 5 vials (1.25 mL) IM within 48 hours CD4+ > 200 Strep. pneum vaccine Azithromycin 1200 mg qw or clarithromycin 500 mg bid Rifabutin 300 mg qd or azithromycin 1200 mg qw + rifabutin 300 mg qd Generally recommended Streptococcus MGH Medical Housestaff Manual 90 36. Prophylaxis for opportunistic infections in HIV pneumoniae Influenza virus All patients Influenza vaccine q year Oseltamivir 75 mg qd or rimantadine 100 mg bid or amantadine 100 mg bid Hepatitis B virus All susceptible Hepatitis B vaccine x 3 Hepatitis A virus All susceptible at increased risk HAV Hepatitis A vaccine x 2 Evidence for efficacy but not routinely indicated Bacteria Neutropenia G-CSF 5-10 mcg/kg sc qd or GM-CSF Cryptococcus neoformans CD4+ <50 Fluconazole 100-200 mg qd Histoplasma capsulatum CD4+ <100 Intraconazole 200 mg qd Cytomegalovirus (CMV) CD4+ <50 and CMV Ab+ Oral ganciclovir 1 g tid Itraconazole 200 mg qd References Refer to http://www.aidsinfo.nih.gov/ for text of 2001 USPHS/IDSA guidelines for the prevention of opportunistic infections in persons infected with HIV, November 2001. Meg Doherty, M.D., Ph.D. MGH Medical Housestaff Manual 91 37. Infection control Hand hygiene Essential for proper patient care. The hands Infection Control Resources: of health care workers are the most MGH Infection Control Unit: 6-2036 important vectors for nosocomial pathogens. For updates, see MGH ID Division website at Hand disinfection is essential before and http://www.mgh.harvard.edu/depts/id/index.html. after any patient contact (or contact with a Partners Handbook Clinical Topics patient’s immediate environment). Infectious Disease MGH Division of Proper procedure is the use of an alcoholInfectious Disease based hand rinse (CalStat at MGH); one pull of the dispenser lever (3mL) is generally sufficient. The rinse should be rubbed over the entire surface of the hands and allowed to dry. If hands are visibly soiled or have contacted blood or body fluids, they should be washed with soap and water and dried thoroughly prior to the application of CalStat. Standard precautions Hand hygiene, as described above, before and after any contact with patient or immediate patient environment. Gloves when anticipating contact with blood, body fluids, secretions, mucous membranes, non-intact skin and any contaminated items. Remove gloves promptly after use and disinfect hands. Gowns when clothes are likely to be contaminated with blood, body fluids, and secretions. Remove gown promptly after use and disinfect hands. Mask and goggles or a face shield during procedures that are likely to generate splashes or sprays of blood, body fluids or secretions. Remove the face protection promptly after use and disinfect hands. Transmission based isolation precautions Airborne precautions. For infections transmissible by droplet nuclei. Negative pressure isolation room, fit-tested N95 masks for all entering the room. Hand hygiene as above. Droplet precautions. For infections transmissible by droplets. Private room, surgical masks within 3 feet of patient. Hand hygiene as above. Contact precautions. For pathogens transmitted by direct or indirect contact. Private room (usually). Gowns and gloves to enter room. Hand hygiene as above. Application of transmission based isolation precautions: common examples Comprehensive list of diseases and applicable precautions available in the Infection Control Manual (print copies on patient care units) and on the Infection Control website Chickenpox or herpes zoster in an immunocompromised host: airborne and contact precautions, though persons with a history of chickenpox may enter the room without a mask. Persons without a history of chickenpox should not enter the room at all, if possible. Shingles (localized herpes zoster) in an immunocompetent host: standard precautions, though all who enter room must have a history of chickenpox. Clostridium difficile diarrhea: contact precautions Influenza: droplet precautions MRSA or VRE colonization: contact precautions Measles: airborne precautions MGH Medical Housestaff Manual 92 37. Infection control Meningitis secondary to Neisseria meningitis or Hemophilus influenzae: droplet precautions until patient has been on effective antibiotic therapy for >24 hours. Tuberculosis: airborne precautions (see algorithm below) Severe acute respiratory syndrome (SARS) (as of May 30, 2003): Airborne and contact precautions with eye protection (goggles or face shield). After leaving room, remove items in this order: 1. gloves, 2. N95 mask, 3. eye protection, 4. gown. Hand hygiene as above. TB ISOLATION One or more of respiratory signs and symptoms: chronic cough, sputum production, dyspnea, hemoptysis AND 1 risk factor: HIV; immigrant (except Canada, W. Europe, Australia, New Zealand); substance abuse; homeless status; recent incarceration; TB exposure; history of non-compliance with TB treatment No Yes Place surgical mask on patient until isolated in negative pressure isolation room on airborne precautions Regular admission Chest X-ray Normal Equivocal or abnormal HIV No Yes Obtain 3 sputums, >24 hours apart, for AFB smear and culture D/c precautions All negative Discontinue precautions unless clinical suspicion is high Any one positive Maintain precautions until no longer infectious by negative AFBs or clinical response (usually 14-21 d). Call Infection Control before discontinuing precautions. Kimon Zachary, M.D. MGH Medical Housestaff Manual 93