Quantitative Analysis (CHM 235)
advertisement

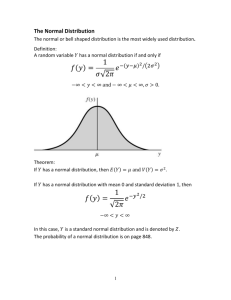
Quantitative Analysis (CHM 235) Dr. S.A. Skrabal Exam 1—8 February 2005 Name:_____________________________________ Instructions: Read each question carefully. Show calculations when required to receive full credit. It is not necessary to show work for the mean and standard deviation when your calculator functions are used. Partial credit is given when warranted. Please box in your final answer. Points will be taken off for incorrect significant figures. Value of each question is given in parentheses after the question. Total points = 100. Note that important information is given on the back pages of the exam. Good luck! 1. A solution containing vanadium was prepared by carefully weighing out 0.1005 g of high-purity solid V2O5 (vanadium pentoxide), transferring it quantitatively to a 200.0 mL volumetric flask, dissolving the solid V2O5 completely in HCl, then bringing the flask to volume with deionized H2O and mixing. (A) What is the concentration of vanadium (V) in the solution in units of ppm? (8) Remember 1 ppm = 1 mg/L (0.1005 g V2O5/200.0 x 10-3 L)(1 mol V2O5/181.88 g V2O5)(2 mol V/1 mol V2O5)(50.94 g V/1 mol V)(1000 mg/g) = 281.47 or 281.5 mg/L or ppm (B) What is the concentration of V in the solution on a %(w:v) basis? (7) %(w:v) = [mass solute (g)/vol. soln. (mL)] 100 (281.47 mg V/L)(g/1000 mg)(1 L/1000 mL)100 = 0.028147% or 2.815 x 10-2 % (w:v) 2. List three methods that can be used to detect and correct for systematic error. (9) Analyze blanks Analyze certified (or standard) reference materials (CRM, SRM) Perform interlaboratory comparison Calibrate instruments Analyze for same analyte using different methods 3. In preparation for a clinical determination of lead (Pb) in human blood, a standard reference material (SRM) was analyzed using a common laboratory method. The SRM is certified to contain 13.9 μg dL-1 (micrograms per deciliter) of Pb. A clinical analyst obtained the following values for Pb in the SRM: 15.7, 16.2, 13.2, 14.8, and 15.1 μg dL-1. (A) Determine whether or not any values should be rejected as questionable values using the Q test at the 90% confidence level. (7) 13.2, 14.8, 15.1, 15.7, 16.2 13.2 appears to be the outlier. Qcalc = gap/range = (14.8 – 13.2)/(16.2 – 13.2) = 0.533 Qtable(n = 5, CL = 90%) = 0.642. Since Qcalc < Qtable, do not reject the data (B) What are the mean, standard deviation, and relative standard deviation of the acceptable results? (10) Using calculator and including all data Mean = 15.0 or 15 μg/dL Std. dev. = 1.1 or 1 μg/dL RSD = 1.1/15.0)100 = 7.3 or 7% Note rounding using “real rule” on sig. figs.---first uncertain digit is last sig. fig. (C) Using the appropriate t-test, determine whether or not the analyst obtained a Pb concentration in the SRM that was significantly different from the certified value at the 95% confidence level. (10) t calc t calc known value x s n 13.9 g / dL 15.0 g / dL 1.1 g / dL 5 2.236 df = n -1 = 5 -1 = 4. ttable(df = 4, 95% CL) = 2.776 Since tcalc < ttable, value is not significantly different from the certified value at the 95% CL. 4. A 10-year-old child began treatment for lead poisoning on 15 February 2000. On that day, the child’s blood was analyzed (4 measurements) and contained 32.7 ± 0.9 μg dL-1, where the uncertainty is the standard deviation. After 30 days of treatment, the child’s blood was reanalyzed (5 measurements) and found to contain 27.5 ± 0.8 μg dL-1. (A) Use the F-test to verify that the standard deviations of the two sets of data are not significantly different at the 95% confidence level. (6) Fcalc = s12/s22 where s1 > s2 Fcalc = (0.9 μg/dL)2/(0.8 μg/dL)2 = 1.226 df = 4 -1 = 3 and 5 -1 = 4. Ftable(df = 3,4; 95% CL) = 6.59 Since Fcalc < Ftable, std. devs. are not significantly different at the 95% CL. 2 4. (B) Use the appropriate t-test to determine whether or not the lead level in the child’s blood was significantly different at the 95% confidence level after the 30 days of treatment. (10) t-test for comparison of means s pooled t calc s12 (n1 1) s 22 (n2 1) n1 n2 2 x1 x 2 n1 n2 s pooled n1 n2 32.7 27.5 0.8443 (0.9) 2 (4 1) (0.8) 2 (5 1) 452 (4)(5) 45 0.8443 9.181 df = n1 + n2 – 2 = 4 + 5 - 2 = 7 ttable(df = 7, 95% CL) = 2.365 Since tcalc > ttable, the mean values on the two days are significantly different at the 95% CL. 5. Twenty-six measurements of the oxygen partial pressure in an experimental chamber were made over a period of 60 days. The mean and standard deviation of these measurements was 0.2095 ± 0.0008 atm (atm = atmosphere). Calculate the 99% confidence interval of these measurements. (10) x ts n df = n -1 – 26 – 1 = 25 ttable(df = 25, 99% CL) = 2.787 0.2095 (2.787)(0.0008) 26 0.2095 0.0004 3 atm 3 6. A student prepared a set of vanadium standards (0-50 μM) which she analyzed using atomic absorption spectroscopy. Using EXCEL, the student plotted her results, then used the LINEST function to obtain the parameters of the regression equation, including the slope (m), the uncertainty or standard deviation of the slope (sm), the y-intercept (b), the standard deviation of the y-intercept (sb), and the standard deviation of the y values (sy). These results are shown in the spreadsheet depicted below. [V] (μM) Abs. Corr. Abs. 0 5.00 10.0 20.0 50.0 0.001 0.023 0.040 0.079 0.192 0 0.022 0.039 0.078 0.191 m= sd of m= R^2 = 0.003797468 3.30894E-05 0.999772274 13170.73171 0.02278481 0.001443038 0.000813892 0.001315279 3 5.18987E-06 =b = sd of b = sd of y (A) Write the linear regression equation in the form y (± sy) = m (± sm) x + b (± sb) using the correct number of significant figures. (6) Apply “real rule” on sig. figs. y (± 0.0013) = 0.003797 (± 0.000033) x + 0.00144 (± 0.00081) or y (± 0.001) = 0.00380 (± 0.00003) x + 0.0014 (± 0.0008) (B) Suppose an unknown sample was analyzed for vanadium in the same manner as the standards, giving an absorbance of 0.112. Determine the vanadium concentration (in μM) of the unknown sample. (8) Blank-corrected absorbance = 0.112 – 0.001 = 0.111 x = (y – b) / m = (0.111 – 0.00144) / 0.003797 = 28.85 μM Keeping 3 sig. figs. so far until uncertainty analysis is performed 4 6. (C) Using the LINEST results, determine the absolute and relative uncertainties of the concentration of the unknown sample. Be sure to express your final answers with the correct number of significant figures. (9) Rewrite equation in (B) including uncertainties: x ( s x ) y ( s y ) b ( sb ) m ( s m ) 0.111( 0.0013 ) 0.0014 4 ( 0.00081 ) 0.00379 7 ( 0.000033 ) 28.85 s x Propogation of uncertainty for mixed operations: enum (0.0013 ) 2 (0.00081 ) 2 %enum 0.0015 0.0015 100 1.3 % (0.111 0.0014 4 ) %edenom 0.000033 100 0.8 6 % 0.00379 7 %eoverall (1.3 %) 2 (0.8 6 %) 2 1. 5 % Convert to absolute uncertainty and express the answer with the correct number of sig. figs. (1.5%) (28.85 μM) = (0.015) (28.85 μM) = 0.43 μM Answers: 28.85 ± 0.43 μM or 28.8 ± 0.4 μM and 28.85 μM ± 1.5% or 28.8 μM ± 2% 5 Useful information Standard deviation: s ( xi x ) 2 s 100 x %RSD = i Confidence interval: x n 1 ts where df = n – 1. n Comparison of means (when standard deviations are not significantly different): t calc x1 x 2 n1 n2 s pooled n1 n2 s pooled s12 (n1 1) s 22 (n2 1) n1 n2 2 where degrees of freedom = n1 + n2 - 2 Comparison of means (when standard deviations are significantly different): t calc x1 x 2 s12 / n1 s 22 / n2 DF ( s 2 / n s 22 / n2 ) 2 2 1 21 ( s 22 / n2 ) 2 ( s1 / n1 ) n1 1 n2 1 Comparing measured result to known result: t calc 2 known value x s n where degrees of freedom = n – 1. Fcalc s12 s 22 where s1 > s2 and degrees of freedom is n1 – 1 and n2 -1 en e12 e22 e32 ... en21 %en %e12 %e22 %e32 ... %en21 y = mx + b Q = gap/range 6