PLEASE TYPE - Northern Arizona University
advertisement

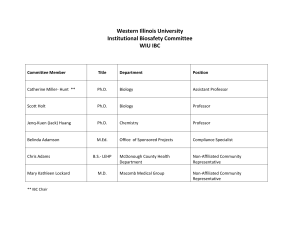
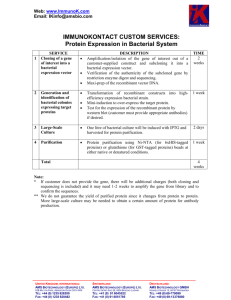
PLEASE TYPE Hand-written forms will be returned R-DNA Northern Arizona University Institutional Biosafety Committee Biological Safety Office Document of Registration RD - Principal Investigator: PI’s Title: Department: Address/Box: Phone: Email: (Added by BSO) Project Title: 1. Location of project 2. Animal Use: Yes No Project bldg: IACUC#: Project room: Animal housing and procedure rooms: ________________________________________ 3. Biological Agent Use: Yes No 4. Bio-Safety Office approval date: Isotope Use: Yes No RCC Approval date: 5. Will you attempt deliberate expression of a gene? 6. Will you use or produce any toxins? 7. Is your project purpose to introduce any drug / immunological resistance gene(s)? 8. Will you be working with 10 or more liters of recombinant material? 9. Purpose of the Project (one sentence): Yes Yes No No Yes Yes No No 10. Project goals/intent: 11. Expected outcome: 12. What will be analyzed or measured? 1/4 RDNA-Reg. ver. 02/16 R-DNA Document of Registration RD - 13. Use the table below to detail recombinants used in this project, one column per construct (attach sheets as necessary). Construct 1 Construct 2 Construct 3 Construct 4 Construct 5 Example DNA/Gene Source Example: Green fluorescent protein, GFP Gene function Example: marker Vector Name Example. pTR-UF12 provide map(s) Vector Type / Origin Example: Viral / Adeno-associated virus (AAV) Expression control elements (promoter, Example: CMV enhancer, enhancer, Chicken regulatory B-actin promoter elements, etc) Conc/titer of rDNA (i.p./ml) Example: 1 X 108 to 1 X1012 infectious particles/ml Example: E. coli, Host and Strain SureTM, Mouse heart cells, in vivo Host Range (including any genetic alterations to Example: Human, other mammalian cells host range) Is recombinant made in your lab? If not, where? Example: UF Powell Gene Therapy Center What (approx.) % of original vector genome has been deleted or substituted? Example: two thirds 2/4 RDNA-Reg. ver. 02/16 R-DNA Document of Registration RD 14. Full Project Description in Lay Terminology with Methods and procedures (add additional pages as needed): 15. What effect would transgene expression have in an accidental host? 16. Other possible safety hazards associated with this project and how you will address them: 17. Biosafety level and NIH guideline citation (IBC use) ________________________________________________ 18. Health surveillance (IBC use) ____________________________________________________________________ Please Attach Vector/Construct Maps. Electronic versions preferred (i.e. .bmp, .tif, .jpg, .pdf), but hard copies acceptable. The next page can be signed then faxed, mailed, or scanned and e-mailed. 3/4 RDNA-Reg. ver. 02/16 R-DNA Document of Registration RD - This page can be signed then faxed, mailed, or scanned and e-mailed separately if needed. 19.The undersigned individual(s) will be involved in the experimentation described above. They are familiar with and agree to abide by the current NIH Guidelines. ALL PARTICIPANT SIGNATURES REQUIRED. Name (Please Type or Print) Signatures Date 13. I attest to the fact that these individuals are properly trained in the area of recombinant DNA experimentation. Furthermore, I agree to comply with the NIH requirements pertaining to shipment and transfer of recombinant DNA materials. I am familiar with and agree to abide by the provisions of the current NIH Guidelines and other specific NIH instructions pertaining to the proposed project. The information above is accurate and complete. Principal Investigator Date 14. The indicated sites have been inspected and are in compliance with the NIH Guidelines. Biological Safety Officer Date Please return this completed form to: Shelley Jones Biological Safety Office Bldg. 56 P.O. Box 4073 Fax – 928-523-0050 Shelley.Jones@nau.edu Phone – 928-523-7268 4/4 RDNA-Reg. ver. 02/16