What is a mineral
advertisement

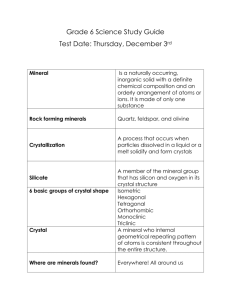
What is a mineral? When you see a glittering crystal in our Museum you would probably say it was a mineral - and you'd almost certainly be right. But can you say what a mineral really is? Defining a mineral A mineral is a naturally occurring inorganic solid, with a definite chemical composition, and an ordered atomic arrangement. This may seem a bit of a mouthful, but if you break it down it becomes simpler. Minerals are naturally occurring They are not made by humans Minerals are inorganic They have never been alive and are not made up from plants or animals Minerals are solids They are not liquids (like water), or gases (like the air around you) Minerals have a definite chemical composition Each one is made of a particular mix of chemical elements Minerals have an ordered atomic arrangement The chemical elements that make up each mineral are arranged in a particular way this is why minerals 'grow' as crystals The tricky bits ... atoms, molecules and the rest There are around four thousand different minerals in the world. They are all naturally occurring inorganic solids. Each mineral is defined by its particular chemical composition and crystal structure. To explain what a mineral is properly, we have to introduce you to a bit of chemistry. Minerals are made up of chemical elements. A chemical element is a substance that is made up of only one kind of atom. Have you heard of oxygen, hydrogen, iron, aluminium, gold and copper? These are all chemical elements. But what is an atom? An atom is the smallest unit of any chemical element. They are the building blocks that make up each chemical element, and are far too small to see with the naked eye. Imagine a small piece of copper, for example. Even the tiniest piece of copper is made up of billions and billions of copper atoms. There are 103 types of atoms, and because each chemical element is made up of only one kind of atom there are 103 chemical elements. Billions and billions of copper atoms stack together to form a piece of copper Each mineral has a fixed chemical composition. Some minerals are made up of just one chemical element - they contain only one type of atom. Native copper is made up of copper atoms only. Most minerals are chemical compounds - they contain atoms of more than one chemical element So what is a chemical compound? Put it this way. Just as chemical elements are made up of atoms, a chemical compound is made up of molecules. Each molecule in a chemical compound is made up of two or more different atoms connected together. Halite is a chemical compound. It is also called sodium chloride, but you would probably know halite better as rock salt - yes, the kind you put on your fish and chips. Each molecule of halite contains one atom of an element called sodium connected to one atom of an element called chlorine. Halite always contains just as many sodium atoms as it does chlorine atoms: this 'formula' does not vary - halite, like all other minerals has a definite chemical composition. Chlorine and sodium atoms will connect together to make halite molecules The atoms in a mineral are arranged in an organised 'atomic structure'. They connect together to form molecules, and the molecules stack together in a regular pattern to form a crystal. The shape of a crystal depends on the way the molecules are stacked up inside it. There is a set way in which the sodium atoms "connect" to the chlorine atoms to make a halite molecule, and these molecules can stack together to form a halite crystal. Halite molecules stack together to form halite crystals - this is the salt we eat everyday All the properties of a mineral - its crystal shape, hardness, colour, lustre - depend on which chemical elements it is made of and how the atoms of these elements are arranged inside it. Identifying Minerals Color - The color of a mineral is one of its most obvious attributes, and is one of the properties that is always given in any description. Color results from a mineral’s chemical composition, impurities that may be present, and flaws or damage in the internal structure. Unfortunately, even though color is the easiest physical property to determine, it is not the most useful in helping to characterize a particular mineral. Many minerals come in a rainbow of colors. Streak - The color of a mineral when it is powdered is called the streak of the mineral. Crushing and powdering a mineral eliminates some of the effects of impurities and structural flaws, and is therefore more diagnostic for some minerals than their color. Streak can be determined for any mineral by crushing it with a hammer, but it is more commonly (and less destructively) obtained by rubbing the mineral across the surface of a hard, unglazed porcelain material called a streak plate. The color of the powder left behind on the streak plate is the mineral's streak. The streak and color of some minerals are the same. For others, the streak may be quite different from the color. Luster - The luster of a mineral is the way its surface reflects light. Most terms used to describe luster are self-explanatory: metallic, earthy, waxy, greasy, vitreous (glassy), adamantine (or brilliant, as in a faceted diamond). It will be necessary, at least at first, only to distinguish between minerals with a metallic luster and those with one of the non-metallic lusters. A metallic luster is a shiny, opaque appearance similar to a bright chrome bumper on an automobile. Other shiny, but somewhat translucent or transparent lusters (glassy, adamantine), along with dull, earthy, waxy, and resinous lusters, are grouped as non-metallic. Hardness - Hardness is defined by how well a substance will resist scratching by another substance. For example, if mineral A scratches mineral B, and mineral B does not scratch mineral A, then mineral A is harder than mineral B. If mineral A and B both scratch each other, then their hardness is equal. A scale to measure hardness was devised by Austrian mineralogist Frederick (Friedrich) Mohs in 1822, and is the standard scale for measuring hardness. The scale consists of numbers one through ten; 1 being the softest and 10 being the hardest. Each number represents a different mineral - each harder than the previous. The 10 minerals are: 1. Talc 2. Gypsum 3. Calcite 4. Fluorite 5. Apatite 6. Feldspar 7. Quartz 8. Topaz 9. Corundum 10. Diamond All conceivable minerals fit in this scale, since Talc is the softest known mineral and Diamond the hardest. To demonstrate how to use the scale, understand the following example: Suppose a mineral scratches Fluorite, but not Apatite, then it has a hardness between 4 and 5. Several common household items have a fixed hardness, and can be used to test for hardness: Fingernail 2½ Penny 3 - 3½ Iron Nail 4½ Glass 5½ Steel file 6½ Streak plate (floor tile) 6½ - 7 Hardness is almost always rounded off to the nearest half number. Specific Gravity - The specific gravity of a substance is a comparison of its density to that of water. Imagine a gallon bottle filled with water, a second filled with feathers, a third filled with lead weights. There are equal volumes of material present, but the bottle with the feathers will weigh less than that containing water; the bottle with lead weights will weigh the most. In order of increasing specific gravity, these materials would be: feathers, water, lead. Specific gravity can be measured precisely, or estimated by a comparison, as above. To compare the specific gravity of any two minerals, simply hold a sample of one in your hand and "heft it," i.e., get a feeling for its weight. Then heft a sample of the other that is approximately the same size. If there is a great difference in specific gravity, you will detect it easily. It is often sufficient to note whether a mineral's specific gravity is significantly higher or lower than that of other minerals. Heft each of the specimens in your mineral set. Which ones have a high specific gravity? A low specific gravity? Crystal Shape - When minerals form in environments where they can grow without interference from neighboring grains, they commonly develop into regular geometric shapes, called crystals, bounded by smooth crystal faces. The crystal form for a given mineral is governed by the mineral's internal structure, and may be distinctive enough to help identify the mineral. For example, quartz forms elongated, six-sided prisms capped with pyramid-like faces; galena and halite occur as cubes; and garnets develop 12- or 24-sided equidimensional forms. Interference from other mineral grains during growth may prevent formation of well-formed crystals. The result is shapeless masses or specimens that developed only a few smooth crystal faces. This type of specimen is much more common than well-formed crystals. Cleavage & Fracture – Cleavage and Fracture are minerals properties that describe the way a mineral breaks when stressed. Cleavage describes the way a mineral breaks along its plane of weakness. These planes of weakness can be described as where the atomic bonding between atoms in the crystal structure is weak at those points causing the mineral break when stressed much like a stack of papers when drop will spread off in individual sheets. There are five basic types of cleavage. One Directional- One Directional Cleavage peels in layers much like taking individual cards off a deck. Mica is a good example of this type of cleavage. Two Directional - Two Directional Cleavage breaks along two directions but not at right angles. Students seem to have the hardest time describing or identifying this cleavage. A good rule of thumb to determine this type if your not sure is to eliminate the other types of cleavage which are easier sometimes to see and what is left is the two direction. Feldspar is a good example of this type of cleavage. Three Directional Not at Right Angles - Three Directional not right angles is often very easy to see. The mineral seems to have three dimensional boxes that would break if dropped. These boxes are leaning on their sides much like a Rhombus. Calcite is a good example of this type of cleavage. Cubic Cleavage - Cubic Cleavage is three directional cleavage at right angles. This cleavage can be easily recognize in that in breaks into perfect squares or cubes. A good example of this type of cleavage is the mineral Halite or "Salt" as it is commonly called. Octahedral Cleavage - Octahedral Cleavage is complex cleavage system where two foursided pyramids are stack on top of each other. This cleavage is often hard to see in some mineral samples containing it. A good example of this type of cleavage is the mineral Fluorite. Fracture is a mineral property where the atomic bonding between atoms in the crystal structure is perfect with no weakness. When these minerals are stressed they shatter making no two pieces truly the same. There are two basic types of fractures, Conchoidal and Non-Conchoidal. Conchoidal Fracture - Conchoidal Fractures are easy to spot. They have a circular pattern in their break are much like broken bottle glass. Obsidian is a good example of this mineral property. Non-Conchoidal Fracture - Non-Conchoidal Fracture minerals are also easy to identify. They have no directional cleavage planes or swirls in there structure. A good example of this is the mineral Quartz. Other Properties - There are a few other tests that can be used to differentiate one or more common minerals. Some of these should be used with great CAUTION! Magnetism - A few minerals are attracted to a magnet or are themselves capable of acting as magnets (the most common magnetic mineral is magnetite). Because these are so rare, this property helps narrow the possibilities drastically when trying to identify an unknown specimen. Feel - Some minerals, notably talc and graphite, feel greasy or slippery when you rub your fingers over them. The greasiness occurs because bonds are so weak in one direction that your finger pressure alone is enough to break them and to slide planes of atoms past neighboring atomic layers. Taste - Geologists use as many senses as possible in describing and identifying minerals. Taste is one of the last tests to be conducted, because some minerals are poisonous. Some minerals taste salty-most notably halite (salt). Sylvite, a mineral similar in all other properties to halite, tastes bitter. Taste is thus a diagnostic property because it distinguishes between these minerals. NEVER TASTE A MINERAL UNLESS INSTRUCTED TO! Reaction with Dilute Hydrochloric Acid - This is actually a chemical property rather than a physical attribute of a mineral. Minerals containing the carbonate anion (C03)2effervesce ("fizz") when a drop of dilute hydrochloric acid is placed on them. Carbon dioxide is liberated from the mineral and bubbles out through the acid, creating the fizz. This test is best performed on powdered minerals. Calcite (calcium carbonate) will effervesce readily in either massive or powdered form, but dolomite (calcium-magnesium carbonate) reacts best as a powder. Flourescence - Fluorescent minerals emit visible light when exposed to ultraviolet light. “Glow in the dark” References: http://www.oum.ox.ac.uk/thezone/minerals/define/index.htm http://geology.csupomona.edu/mineral/minerals.htm