Matter Unit Vocabulary
advertisement

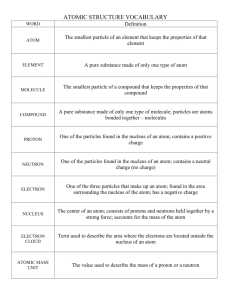
Matter Unit Vocabulary With Definitions 1. Matter – is basically anything that takes up space. Put it another way, it is anything made of stuff. The dictionary says: a. Something that occupies space and can be perceived by one or more senses; a physical body, a physical substance, or the universe as a whole. b. Physics. Something that has mass and exists as a solid, liquid, gas, or plasma… 2. State or Phase of Matter: the forms in which matter (stuff) can exist. All of the stuff in the universe that takes up space can be divided into one of the states of matter. For now, we will only look at three of them: 1. Solids, like wood, steel and glass 2. Liquids, like water, syrup and gasoline, and 3. Gases like air (which is really many gases including: oxygen, carbon dioxide, and nitrogen.) All matter can be found in any of the phases depending on the temperature and pressure, so steel can be found as a solid, liquid and under very unusual conditions gas. 3. Phase change or Change in State: when matter changes from solid to liquid to gas in any way. Add heat to go from S to L to G, take away heat to go from G to L to S. 4. Mass: to simplify, the mass of a thing is how much matter or stuff is in it. Physicists say it is “the measure of an object's resistance to changes in either the speed or direction of its motion.” If you think about it something that has more stuff or matter in it will be harder to move than something with only a little stuff in it. Compare moving your little brother (a little bit of stuff) and moving me (a lot of stuff.) Mass is measured in pounds, ounces, grams and kilograms. 5. Weight: a measurement of the force of gravity on an object. The larger the mass, the more gravity pulls it, so the greater the weight. The smaller the mass, the less gravity pulls it, so the smaller the weight. Weight IS NOT the same as mass. Your weight is much different on the moon than on earth, yet your mass remains the same. We use different tools to measure mass and weight. Weight uses the same units of measure as Mass. 6. Volume: the amount of space an object takes up. Volume is measured in pints, cups, quarts, and gallons in the customary English system and in Liters and Milliliters om the metric system. 7. Density: the amount of matter that is contained in a unit of volume. Something that has 5 grams of matter per liter is much less dense than one with 500 grams per liter. 8. Solid: the phase of matter that has a definite shape and volume; not liquid or gaseous 9. Liquid: the phase of matter that has a definite volume, but not a definite shape, liquids are fluid (as water), meaning they flow from one place to another and take the shape of their container BUT do not expand to fill their container. Liquids can be thick like corn syrup or thin like water (scientists call this viscosity.) Even under a lot of pressure, liquids are only slightly compressible. 10. Gas: the phase of matter that has no definite shape or volume, gases expand to fill whatever container they are in. They are not viscous (thick) and can be compressed by forcing them into small rigid containers. 11. Evaporation: When a liquid is converted or changed into a gas (also called a “vapor.”) This is one of the changes in the phase or state of matter. 12. Condensation: When a gas is converted or changed into a liquid. This is one of the changes in the phase or state of matter. You see this when water vapor condenses on your mirror when you shower. The water gas (vapor) changes to liquid water when it is cooled by touching the cool mirror surface. 13. Freeze: When a liquid is converted or changed into a solid. This is one of the changes in the phase or state of matter. 14. Melt: When a solid is converted or changed into a liquid. This is one of the changes in the phase or state of matter. 15. Freezing point: the temperature at which a liquid changes to solid at a particular pressure. 16. Melting point: the temperature at which a solid changes to liquid at a particular pressure. 17. Temperature: is a measurement of the hotness or coldness of a piece of matter. Scientists say it is average amount of kinetic (movement) energy that something has. 18. Atom: a Greek word that was first used by a Greek scientist named Democritus. It means that which can not be cut. Democritus figured if you cut something into smaller and smaller pieces, you would finally get to a piece so small it could not be cut any more. This is the atom. After about 2000 years, scientists discovered that even atoms can be cut. They are made of much smaller particles: the Proton, Neutron, and electron (which are made of Quarks, which some think are made of even smaller things called “strings.” 19. Nucleus: the protons and neutrons in the center of an atom. 20. Proton: a small sub-atomic (smaller than an atom) that is in the nucleus and has a POSITIVE electrical charge. 21. Sub-atomic particles: particles that are smaller than an atom. Particles used to make atoms. 22. Electron: a small sub-atomic that is in the nucleus and has a NEGATIVE electrical charge. 23. Neutron: a small sub-atomic (smaller than an atom) that is in the nucleus and has a NEUTRAL electrical charge. 24. Molecule: the smallest piece of a compound that still has the physical properties of that compound. 25. Meter 26. Centimeter 27. Liter 28. Milliliter 29. Gram 30. Kilogram These are Metric system units of measurement that you must know how to use from practice. 31. Solution: 32. Solute 33. Homogenous – uniform in nature. It is the same throughout without and clearly different material in the mixture. Example- ice tea – it is all the same throughout. 34. Heterogeneous – made of a number of different things. Clearly distinguishable parts like salad or mixed cereal. 35. Suspension - heterogeneous fluid containing solid particles that are sufficiently large for sedimentation an example is silt mixed in water. When stirred it will look fairly homogenous (but it is heterogeneous on close inspection), but the silt will settle and form sediment after a time. 36. Colloid – a type of chemical mixture where one substance is dispersed evenly throughout another. Milk is a perfect example. The particles of the dispersed substance are only suspended in the mixture, unlike a true solution where they are completely dissolved within. The true solution is generally transparent – for example sugar dissolved in water, The particles in a colloid are larger than in a solution - small enough to be dispersed evenly and maintain a homogenous appearance - but large enough to scatter light and not dissolve. This makes colloids cloudy in appearance. 37. 38. 39. 40. 41. 42. 43. 44. 45. 46. 47. 48. 49. 50.