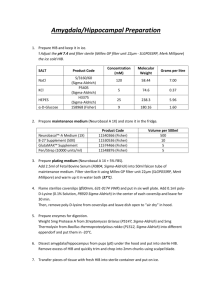
Supporting Figures
advertisement

SUPPLEMENTARY MATERIALS AND METHODS Animals For in vivo experiments, male Wistar Hannover (WH) rats and Spontaneuosly Hypertensive Rats (SHR) (Taconic Farms, Germantown, NY) were kept three per cage with free access to water and a standard diet at 22°C under a 12:12 hour dark-light cycle. For in vitro studies, three-week-old WH rat males were used for isolation of brain microvascular endothelial cells; eigth-day-old Sprague Dawley rat pups (Taconic Farms) were used for isolation of cerebellar granule cells; and one-day-old Sprague Dawley rat pups were used for isolation of cortical microglial cells. The National Institute of Mental Health Animal Care and Use Committee (Bethesda, MD, USA) approved all procedures. All efforts were made to minimize the number of animals used and their suffering (National Institutes of Health Guide for the Care and Use of Laboratory Animals, Publication No. 80-23, revised 1996). In vivo studies Short-term AT1 receptor blockade. To study the effects of short-term AT1 receptor blockade on LPS-induced immune response, 9-week-old WH rats received daily subcutaneous injection of vehicle or AT1 receptor blocker candesartan (CV-11974, Astra-Zeneca, Mölndal, Sweden) at the dose of 1 mg/kg/day for three consecutive days. Candesartan was initially dissolved in 0.1 N Na2CO3 and further diluted to working concentration with isotonic saline, at a final pH of 7.5–8.0. The animals were injected intraperitoneally with 50 µg/kg 1 lipopolysaccharide (LPS, Escherichia coli serotype 055:B5; Sigma-Aldrich, St Louis, MO) or sterile saline on day 3 and were euthanized three hours later by fast decapitation for plasma and tissue collection. Behavioral studies. To establish the effect of short-term AT1 receptor blockade on LPSinduced anorexia and body weight loss, 9-week-old WH rats were housed individually with free access to a standard diet and water. The rats were injected daily with either candesartan (1 mg/kg/day sc.) or vehicle until the end of experiment. On the third day, the rats were injected with LPS (50 µg/kg ip.) or saline and were euthanized 2 days later. Body weight and food consumption were monitored daily throughout the experiment. For determination of anxiety, 9-week-old SHR were treated subcutaneously with 1 mg/kg body weight candesartan or vehicle for three consecutive days followed by intraperitoneal injection of LPS, 50 µg/kg body weight. Three hours after LPS injection, the rats were tested for anxiety-like behavior on an elevated plus maze (Columbus Instruments, Columbus, OH). The elevated plus maze consisted of two opposite open arms (50x10 cm) and two opposite closed arms of the same dimensions with walls 40 cm high. The arms were connected through a central square (10x10 cm) forming the shape of cross and elevated 50 cm above the ground. Testing was conducted in a quiet room under dim illumination. To facilitate adaptation, the animals were placed in the testing room at least 1 hour prior to testing. The test began by placing the rat on the central square facing an open arm. The time spent and the number of entries into the open and closed arms were recorded during 5 min. The arm entry was defined by placing the all four limbs on the arm (Saavedra et al., 2006). 2 Sustained AT1 receptor blockade. To determine the effects of sustained AT1 receptor blockade, 9-week-old WH rats were anesthetized with 30 mg/kg pentobarbital, and were implanted subcutaneously with osmotic minipumps (model 2002, Alza Scientific Products, Palo Alto, CA). Half of the animals received minipumps containing candesartan diluted as above to be delivered at the rate of 1 mg/kg/day; the rest of animals received minipumps filled with vehicle. On day 14, the animals were injected with LPS (50 µg/kg ip.) or sterile saline and were euthanized three hours later by fast decapitation for plasma and tissue collection. For immunohistochemical studies, separate groups of animals treated as above were anesthetized with 50 mg/kg pentobarbital three hours after LPS or saline injection and were transcardially perfused with saline followed by a fixative solution containing 4% formaldehyde (Mallinckrodt Chemicals, Philipsburg, NJ) in phosphate-buffered saline (PBS: 5.6 mM Na2HPO4, 1.0 mM KH2PO4, 154 mM NaCl, pH 7.4). After perfusion, the brains were dissected, post-fixed in the same fixative overnight at 4°C and cryoprotected by consecutive immersions into 10, 20, and 30% sucrose in PBS at 4°C until saturation. Brains were frozen in isopentane on dry ice and stored at −80 °C until used. Plasma and tissue collection. Trunk blood was collected into pre-chilled tubes containing 1 mg/ml EDTA (Sigma-Aldrich), centrifuged at 1,800 g for 15 min at 4ºC and the plasma was stored at -80ºC until used. The brains were quickly dissected, frozen in isopentane on dry ice and stored at -80°C until used. In vitro studies 3 Cerebellar granule cell (CGC) culture. CGCs were isolated from eight-day-old Sprague Dawley rat pups as described previously (Gao, et al 1995). Briefly, 10 cerebella from rat pups were broken down into small pieces by repeated pipetting and incubated with 0.25 mg/ml trypsin (Sigma-Aldrich) in neurobasal medium (Invitrogen, Carlsbad, CA) for 12 min at 37°C. The digest was briefly treated with 0.52 mg/ml trypsin inhibitor (Sigma-Aldrich) and 0.08 mg/ml DNase II (Sigma-Aldrich) and pelleted by centrifugation. The cells were resuspended in B27/neurobasal medium (Invitrogen), plated at a density of 1.2 × 106 cells/ml onto poly-L-lysine coated 6-well plates (Becton Dickinson and Company, Franklin Lakes, NJ) and cultured for 6 to 14 days at 37°C under 5% CO2 atmosphere. We have confirmed that contaminating glial cells represented only 2-3% of the total cell population as initially described (Gao, et al 1995). Before the experiment, the cells were starved overnight in plain neurobasal medium and were subsequently treated for 4 h with 500 ng/ml LPS with or without pre-treatment with 5 μM candesartan for 2 h. At the end of the experiment the medium was saved and the cells were rinsed with PBS and lysed with TRIzol reagent (Invitrogen) for RNA isolation as described below. Candesartan was initially prepared as 10 mM stock in DMSO and further diluted in incubation medium to 0.05% final DMSO concentration. The control cells were treated with vehicle (0.05% DMSO). Brain microvascular endothelial cell culture. Primary endothelial cell cultures were prepared from 3-week old rats under aseptic conditions as described previously (Nakagawa, et al 2009) with a few modifications. Forebrains cleaned of meninges and white matter were minced into small pieces and dissociated by 25 up-down strokes with a 5 ml pipette in Dulbecco’s 4 modified Eagle’s medium (DMEM, Sigma-Aldrich) containing 1 mg/ml collagenase type 2 (Worthington Biochemical Corp., Lakewood, NJ), 15 µg/ml DNase I (Sigma-Aldrich), and 50 µg/ml gentamycin (Mediatech, Herndon, VA) followed by digestion for 90 min at 37°C under constant shaking. The cells were sedimented by centrifugation at 1000g for 10 min at 4°C, resuspended in DMEM containing 20% bovine serum albumin (BSA, Sigma-Aldrich) and recentrifuged at 1000g for 20 min at 8°C. The pellet was resuspended in DMEM containing 1 mg/ml collagenase-dispase (Roche Applied Sciences, Basel, Switzerland) and 6.7 µg/ml DNase I (Sigma-Aldrich) and further digested for 45 min at 37°C. The dissociated cells were sedimented at 800g for 8 min at 8°C, washed, recentrifuged and plated on 35 mm plastic dishes coated with collagen type IV and fibronectin (both 0.1 mg/ml, Sigma-Aldrich) at 4x 105 cells/ml. The cells were kept in DMEM/F12 (Sigma-Aldrich) supplemented with 10% plasma-derived serum (Animal Technologies Inc., Tyler, TX), 1.5 ng/ml basic fibroblast growth factor (Roche Applied Sciences), 100 µg/ml heparin (Sigma-Aldrich), 5 µg/ml insulin, 5 µg/ml transferrin, 5 ng/ml sodium selenite (insulin-transferrin-sodium selenite media supplement, Sigma-Aldrich), 50 µg/ml gentamycin and 4 µg/ml puromycin (Sigma-Aldrich) at 37°C in a humidified 5% CO2 atmosphere. On the third day the medium was replaced with puromycin-free medium supplemented with 500 nM hydrocortisone (Sigma-Aldrich). After the first passage, the cells were plated onto collagen IV- and fibronectin-coated 6-well plates at 4x105 cells/ml and used for experiments when they reached 80% confluence. Before the experiment, the cells were starved overnight in serum-free plain DMEM/F12 medium and were subsequently treated with 100 ng/ml LPS for 4 h with or without prior pre-incubation with 10 µM candesartan for 2 h. At the end of incubation, the medium was saved and the cells were washed in PBS and lysed in Trizol reagent for RNA isolation, as described below. 5 Cortical microglial cell culture. Microglia was prepared from one-day-old Sprague Dawley rat pups as initially described by Giulian and Baker (1986) with slight modifications. Pups were decapitated and the whole brain was placed into Hanks’ Balanced Salt Solution (Invitrogen) containing 10 mM HEPES and 1 mM sodium pyruvate. Cerebral cortices were detached and minced by scissors, followed by 0.25% trypsin (Invitrogen) digestion for 5 min at 37°C. Cortices were transferred to fresh tubes and dissociated by trituration in a complete culture medium DMEM/F12 (Invitrogen), supplemented with 10% FBS (Invitrogen), 100 U/ml penicillin-streptomycin (Invitrogen), 0.1 mg/ml DNase I (Sigma-Aldrich). After centrifugation at 1000 rpm for 5 min, mixed cells were resuspended in the complete medium and passed through a 70 μm cell restrainer (BD Falcon, Vernon Hills, IL). The cell suspension was plated at a density of 2 brains/185 cm2 flask in a humidified 5% CO2 atmosphere at 37°C. The medium was replaced with fresh one after 24 h and then twice per week. After 10 days of culture, flasks were shaken for 2 h at 200 rpm on a rotary shaker at 37°C to detach the microglia. Floating cells were collected by centrifugation at 1000 rpm for 5 min and then seeded onto 6-well plates in a fresh complete medium at a density of 2x106 cells/well. Microglia cultures were washed 30 min after seeding to remove non-adherent cells. Cells were allowed to grow for 24 h at 37°C in a humidified atmosphere enriched with 5% CO2 and then starved overnight in plain DMEM/F12 medium. Subsequently, the cells were treated for 4 h with 100 ng/ml LPS with or without 2 h pre-incubation with 10 μM candesartan. At the end of experiment the medium was saved and the cells were rinsed with PBS and lysed in TRIzol reagent (Invitrogen) for RNA isolation as described below. 6 Determination of cytokine levels Cytokines in plasma and cell culture medium were measured by commercial enzymelinked immunosorbent assay (ELISA) kits from BioSource (Camarillo, CA) for IL-6 and TNF-α and from R&D Systems (Minneapolis, MN) for IL-1β and IL-10. Microdissection of brain structures. Three hundred µm thick coronal sections from fresh-frozen brains were cut on a Leica CM3050S cryostat (Leica Microsystems, Wetzler, Germany) at -10°C, mounted on Superfrost Plus slides (Daigger, Vernon Hills, IL) and immediately frozen on dry ice and stored at -80°C until further processed. Sections were used for punch microdissection as follows: three sections between -1.3 and -2.2 mm from bregma for the PVN and the central nucleus of the amygdala; two sections between -0.7 and -1.3 mm from bregma for the SFO; three sections between -2.2 and -3.1 mm from bregma for the CA1 subdivision of the hippocampus; and three sections between 3.7 and 2.9 mm from bregma for the ventromedial prefrontal cortex (Paxinos and Watson, 1998). Punch microdissection was performed under stereomicroscope control using Harris Uni-Core microdissection needles (Electron Microscopy Sciences, Hatfield, PA), 0.75 mm diameter for the PVN and 1 mm for the other brain structures. The individual punches dissected from same nucleus were pooled and transferred into ice-cold micro-homogenizers (Kontes Glass Co., Vineland, NJ) and immediately homogenized in TRIzol reagent for RNA isolation as described below. Tissues from each rat were studied individually. 7 Determination of mRNA expression of pro-inflammatory factors by real-time PCR RNA from microdissected brain tissue or cell culture lysates was isolated by TRIzol reagent (Invitrogen) followed by purification using the RNeasy Mini kit (Qiagen, Valencia, CA) according to the manufacturer’s instructions. Total RNA (0.15 μg from brain tissue; 1 μg from cultured cells) was reversely transcribed into cDNA using Super-Script ΙΙΙ first-Strand Synthesis kit (Invitrogen) according to the manufacturer's protocol and quantified by real-time PCR using the DNA Engine Opticon system (Bio-Rad, Hercules, CA). The reaction consisted of 10 µl SYBR Green PCR Master Mix (Applied Biosystems, Foster City, CA), 2 µl of diluted cDNA and 0.3 µM primers in a total volume of 20 µl. Primers used in the study are listed in supplementary Table S1. Amplification was initiated by denaturation at 95°C for 10 min, followed by 40-50 cycles of 95°C for 15 sec and 60°C for 60 sec. At the end of amplification, the specificity of the PCR products was confirmed by melting point analysis. Serial dilutions of rat cDNA from the same source as the samples were used to obtain a standard curve. The target cDNA from individual samples was quantified by determining the cycle threshold and comparison with standard curves. The relative amount of the target cDNA was normalized to GAPDH mRNA and expressed as a fold change relative to vehicle-saline group. The expression of GAPDH mRNA was not affected by any of the treatments used. Angiotensin II receptor autoradiography Fresh-frozen brains were cut to 16 μm thick coronal sections containing PVN and SFO on a cryostat at -20°C. The sections were thaw-mounted on Superfrost Plus slides, vacuum-dried 8 overnight at 4°C and stored at −80°C until used. The receptor autoradiography was performed as described previously (Tsutsumi and Saavedra, 1991). Briefly, the sections were preincubated for 15 min at 22°C in a 10 mM sodium phosphate buffer, pH 7.4, containing 0.005% bacitracin (Sigma-Aldrich), 5 mM Na2EDTA, 120 mM NaCl and 0.2% proteinase-free BSA (SigmaAldrich). Sections were then incubated for 2 h at 22°C in fresh buffer containing 0.5 nM [125I]Sar1-Ang II. To characterize Ang II receptor subtypes, adjacent tissue sections were incubated as above in the presence of either 10 μM losartan (DuPont Merck, Wilmington, DE), a selective AT1 receptor antagonist, or 5 μM unlabeled Ang II (Peninsula Laboratories) to assess nonspecific binding. The number of AT1 receptors was determined as the specific binding displaced by losartan. After incubation, slides were washed four times for 1 min in Tris–HCl, pH 7.4 at 4°C, followed by a 30 s rinse in distilled water at 4°C. Slides were then dried under a stream of cold air and exposed to Biomax MR film (Eastman Kodak, Rochester, NY). Optical densities of autoradiograms were normalized after comparison with [14C]-microscale standards as described (Tsutsumi and Saavedra, 1991) and quantified by computerized densitometry using the Scion Image 4.0.2 software (Scion Corporation, Frederick, MD) based on the NIH Image Program of the National Institutes of Health. Binding signal was expressed as fmol/mg of protein. Localization and quantification of mRNA expression by in situ hybridization Fresh-frozen brains were cut on a cryostat at -20°C to 16 µm thick coronal sections containing PVN, SFO and choroid plexus. The sections were thaw-mounted on Superfrost Plus slides, dried for 5 min at 50°C and stored at -80°C until used. 9 In situ hybridization of c-Fos and IL-1β. In situ hybridization of c-Fos and IL-1β mRNA was performed using [35S]-labeled synthetic oligonucleotide probes. Antisense probes for the rat IL-1β 5’-CCACGGGCAAGACATAGGTAGCTGCCACAGCTTCTCCACAGCCAC-3’ (Kim, et al 2004) and c-fos 5’-GCAGCGGGAGGATGACGCCTCGTAGTCCGCGTTGAAACCC GAGAA-3’ (Vaisanen, et al 2004) and their corresponding sense probes were synthesized by Bioserve Biotechnologies (Beltsville, MD). Labeling was performed using the terminal deoxynucleotidyl transferase (New England Biolabs, Ipswich, MA) to a specific activity of 3–4 × 108 cpm/μg. Each reaction was performed with 10 pmol of oligonucleotide probe in the presence of 200 μCi of [α-35S] ATP (NEN/Perkin Elmer, Waltham, MA). The labeled oligonucleotides were separated from unincorporated nucleotides using ProbeQuant G-50 Micro columns (GE Healthcare, Buckinghamshire, UK). The labeling of oligoprobes was monitored with liquid scintillation counting. Prior to use, the sections on slides were warmed in a desiccator at room temperature and then fixed in 4% formaldehyde (Mallinckrodt Chemicals) in PBS for 10 min. After two washes in PBS, they were acetylated for 10 min in 0.1 M triethanolamine HCl, 0.9% NaCl, and 0.25% acetic anhydride (Sigma-Aldrich), delipidated in ethanol and chloroform and air-dried. The sections were covered with 150 µl hybridization buffer containing 50% formamide (Sigma-Aldrich), 1×Denhardt’s solution (Sigma-Aldrich), 10% dextran sulfate (Oncor Inc, Gaithersburg, MD), 100μg/ml PolyA (Sigma-Aldrich), 100μg/ml salmon testes DNA (SigmaAldrich), 100μg/ml yeast RNA (Sigma-Aldrich), 250μg/ml yeast tRNA (Invitrogen), 150mM DTT (MP Biomedicals), 0.2% sodium dodecyl sulfate (SDS) (Quality Biological Inc, Gaithersburg, MD), 0.2% sodium thiosulfate (Sigma-Aldrich) and 2 × 107cpm/ml sense or antisense probe and the slides were covered with glass cover slips. After hybridization for 18 h at 40°C in humidified chamber, the coverslips were removed by rinse in 4× standard saline citrate 10 (SSC) and the sections were washed 3 x 5min in 1x SSC at room temperature followed by 15 min high-stringency washings consecutively in 1x SSC, 0.5x SSC and 0.2x SSC at 55°C. The sections were dehydrated in graded ethanols containing 0.3 M ammonium acetate, air dried and exposed to Biomax MR film (Eastman Kodak). The optical densities of autoradiograms were measured using the Scion Image 4.0.2 software (Scion Corporation). The intensities of the hybridization signals were expressed as nCi/g tissue equivalent after calibration with [ 14C]microscales standards (Tsutsumi and Saavedra, 1991), and after subtraction of the non-specific hybridization signal obtained in the same areas of adjacent sections hybridized with sense (control) probes. In situ hybridization of AT1A receptor. In situ hybridization was performed using [35S]labeled antisense and sense (control) riboprobes as described previously (Jőhren, et al 1995). Fragments from the 3’-non-coding regions of the AT1A cDNA were subcloned into the polylinker site of the pBluescripts KS(+) vector (Stratagene, La Jolla, CA). A 368 bp PalI fragment of the rat AT1A cDNA was ligated into the EcoRV site of the pBluescript vector. The orientation of the insert was determined by restriction analysis with AvaII, which cuts asymmetrically within the subcloned AT1A cDNA fragment. The subclones containing plasmids were linearized with HindIII or EcoRI to generate [35S]-labeled sense and antisense probes. Radiolabeled probes were prepared by in vitro transcription in the presence of [35S]-UTP (MP Biomedicals, LLC, Solon, OH), 1μg of linearized subclone plasmid and T7 or T3 RNA polymerase, using a MAXI-Script® T7/T3 kit (Ambion, Austin, TX) according to the protocol of the manufacturer. After transcription, the template DNA was digested with DNase I for 15 min at 37 °C. Unincorporated nucleotides were removed by centrifugation through ProbeQuant™ G-50 Micro Columns (GE 11 Healthcare). The labeling of riboprobes was monitored with liquid scintillation counting and the specificity verified by a preliminary experiment with the PVN as a positive control (Jőhren, et al 1995). Prior to use, the sections prepared for hybridization were warmed in a desiccator at room temperature and then fixed in 4% formaldehyde in PBS for 10 min. After two washes in PBS, they were acetylated for 10 min in 0.1 M triethanolamine HCl, 0.9% NaCl, and 0.25% acetic anhydride, delipidated in ethanol and chloroform, and air-dried. The sections were covered with 150μl hybridization buffer containing 50% formamide, 0.3 M NaCl, 1 mM EDTA, 20 mM Tris, pH 7.5, 1x Denhardt's solution, 10% dextran sulfate, 100μg/ml salmon testes DNA, 250 μg/ml yeast RNA, 250 μg/ml yeast tRNA, 150 mM DTT, 0.2% SDS, 0.2% sodium tiosulfate, and 2×107 cpm/ml sense or antisense probe, and the slides were covered with glass cover slips. After hybridization for 18 h at 54 °C, coverslips were removed by rinse in 4x SSC and the sections were washed 3 x 5 min in 4x SSC at room temperature. Non-hybridized probes were digested by incubation with 40μg/ml RNase A (Sigma-Aldrich) for 30 min at 37 °C followed by high stringency wash in 0.1X SSC at 65 °C for 60 min. The sections were dehydrated in graded ethanols containing 0.3 M ammonium acetate, air dried and exposed to Biomax MR film (Kodak, Rochester, NY). The mRNA expression was analyzed by measuring optical film densities as described above. Two consecutive sections per rat were averaged and used to calculate group means. Each animal was quantified independently. Results are presented as means ± SEM. Tissue background was not different from the film background and no hybridization signal over background was observed using sense RNA probes. Immunohistochemical localization and quantification of c-Fos protein and microglia 12 Formaldehyde-fixed rat brains were cut on a cryostat at -20°C to 16 μm thick coronal sections containing PVN. The sections were thaw-mounted to Superfrost Plus slides, vacuumdried overnight at 4°C and stored at -80°C until used. Before the immunohistochemical staining, the slides were warmed in a desiccator at room temperature, immersed in ice-cold acetone for 15 min, and incubated 3 x 5 min in Tris-buffered saline (TBS: 20 mM Tris, 136 mM NaCl, pH 7.4) containing 0.2% Triton X-100 (Sigma-Aldrich). Intracellular peroxidase was quenched with 0.3% hydrogen peroxide containing 0.1% sodium azide in TBS for 30 min at room temperature. The sections were washed 3 x 5 min in TBS and blocked for 60 min with TBS containing 1% protease-free BSA, 10% normal goat serum (Vector Laboratories, Burlingame, CA), and 4 drops/ml of avidin solution (Avidin/Biotin Blocking Kit, Vector Laboratories). After blocking, the slides were rinsed briefly in TBS and incubated overnight at 4°C with a primary antibody diluted in TBS containing 1% BSA, 5% normal goat serum and 4 drops/ml biotin (Avidin/Biotin Blocking Kit, Vector Laboratories). Rabbit polyclonal antibody against rat c-Fos (Ab-5, 1:10000, Calbiochem, La Jolla, CA), or mouse monoclonal antibody against rat CD11b/c (Ox-42, 1:300, BD Pharmingen, San Jose, CA) were used as primary antibodies. The sections were washed 4 x 5 min with TBS containing 0.05% Tween-20 (TBS-T) and incubated for 2 h at room temperature with the goat biotin-conjugated secondary antibody against rabbit or mouse IgG (1:1000, Jackson ImmunoResearch, West Grove, PA) diluted in TBS containing 1% BSA and 5% normal goat serum. The sections were then washed 4 x 5 min with TBS-T and incubated 30 min with avidin and biotin-conjugated horseradish peroxidase H (Vectastain ABC kit, Vector Laboratories). The sections were exposed to DAB reagent (Vector Laboratories), counter-stained with hematoxylin, dehydrated with ethanol of increasing grade (from 50 to 100%), cleaned with 13 xylene and mounted with coverslips using xylene-based mounting medium (Permount, Fisher Scientific, Fair Lawn, NJ). Data acquisition and quantification. For data acquisition and quantification, sections were examined by an investigator blinded to the protocol used and the identity of the slides, using a Zeiss Axioscope light microscope (Zeiss, Jena, Germany) and the images were acquired by a Zeiss Axiocam digital camera under the constant conditions of light illumination. Induction of c-Fos protein in the PVN was evaluated by automated image analysis using Image J software (National Institutes of Health, Bethesda, MD). The images analyzed were converted into grey scale and the identical threshold was applied to all pictures to eliminate background. Positive cells were counted automatically by particle analysis and data were expressed as the number of c-Fos-positive cells per section of PVN. Five to eight animals per group were analyzed individually. Microglia activation was evaluated as an area of PVN positive for the microglia marker Ox-42 staining (Milligan, et al 1991). The analysis was performed by an automated procedure using Image J software (National Institutes of Health). Immunohistochemical images were processed by color deconvolution plug-in using H-DAB vector (http://rsbweb.nih.gov/ij/plugins) (Ruifrok and Johnston, 2001) to separate microglia staining from hematoxylin counter-staining. The images of microglia were then subjected to the same threshold to eliminate background and converted into black and white scale. The resulting images were analyzed automatically by particle analysis function and data are expressed as a proportion of Ox-42 positive area to the total area of the PVN. The increase in the proportion of the area occupied by microglia was used as a measure of activation as reported previously (Popovich, et al 1997). 14 Statistical analysis All results are expressed as means ± SEM for groups of 5-10 animals measured individually. Results from cell culture experiments are expressed as means ± SEM of three different cell preparations assayed individually. . To address the differences among all individual groups, we analyzed the data by one-way ANOVA followed by the post hoc Newman-Keuls’ test for all determinations with significant omnibus ANOVA. P<0.05 was considered as statistically significant. All statistics were performed with the use of GraphPad Prism 5.02 software (GraphPad Software Inc., San Diego, CA). 15