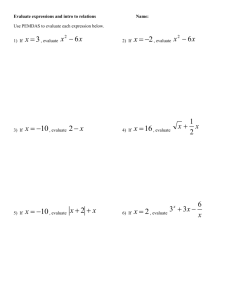Classifying Chemical Reactions Worksheet
advertisement

IIT/FIELD MUSEUM – High School Transformation Project Classifying Chemical Reactions Glencoe Chemistry: Matter and Change Unit 3 Bonding and Reactions Chapter 10 Chemical Reactions Section 10.2 Classifying Chemical Reactions Context of the Lesson After students learn what chemical reactions are and how to express them in chemical equations, they need to learn to identify the five (5) fundamental types of chemical equations: synthesis, decomposition, single-replacement, double-replacement and combustion. This lesson provides students with a guided-inquiry experience that allows them to understand the types of chemical reactions by leading them through several exercises using analogies. After this lesson, students will be ready to learn aqueous chemical reactions. Main Goals & Objectives Students will sort mathematical equations into categories then repeat the exercise with chemical equations. Afterwards, students will listen to a story with analogies to chemical changes and then create their own analogy stories. From performing these activities, students will be able to: Classify chemical reactions as synthesis, decomposition, single-replacement, doublereplacement or combustion reactions. Identify the characteristics of the fundamental types of chemical reactions. Develop models for chemical reactions and identify the limitations of the models. Nature of Science: Integrated Theme Explain that scientists’ creativity influences their doing inquiry so they may have different observations and interpretations of the same phenomena. Explain that scientists’ background knowledge influences their doing inquiry so they may have different observations and interpretations of the same phenomena. Scientific Inquiry: Integrated Theme Explain that scientific investigations all begin with a question, but do not necessarily test a hypothesis. Explain that there is no single scientific method and provide at least two different methods. Explain that inquiry procedures are guided by the question asked. Explain that all scientists performing the same procedures may not get the same results. Explain that inquiry procedures can influence the results of an investigation. Explain that research conclusions must be consistent with the data collected. General Alignment to Standards STATE GOAL 12: Understand the fundamental concepts, principles and interconnections of the life, physical and earth/space sciences C. Know and apply concepts that describe properties of matter and energy and the interactions between them 12.C.5a Analyze reactions (e.g., nuclear reactions, burning of fuel, decomposition of waste) in natural and man-made energy systems. Materials Per pair of students: “Mathematical Equations Challenge” sheet* “Chemical Equations Challenge” sheet* Scissors *Teacher note: The two equation sheets should be copied on differently colored paper because although, they will be cut up into pieces, they still need to be distinguishable. Per student: Notebook Reaction Types Worksheet Safety Remind students that lab materials are to be used ONLY as directed Review safety issues regarding scissors and other sharp objects The Lesson Day One Bell Ringer: On the board or overhead, write: “We humans classify many things—living organisms, food groups, types of cars, just to name a few. What is the purpose of all this classifying? Write three reasons that classification is useful.” Have students think, pair, share their ideas. Activity Assign students to teams of two and distribute the “Mathematical Equations Challenge” worksheet (one per team). Instruct students to cut the sheet into strips, with one equation on each strip. Then sort the equations into groups based on their observations. Circulate among the students and observe their equation groupings. If students seem really confused, suggest that they look for five possible groups or not at the actual numbers, but at the way the equations look overall. When a team has successfully grouped their equations into five groups (which will look remarkably like synthesis, decomposition, etc.), have them explain their groupings. Then give them the “Chemical Equations Challenge” worksheet and instruct them to repeat the procedure done with the math equations. Indicate that they use their groups of mathematical equations to help them sort the chemical equations. Almost all students will classify the equations into the five (5) fundamental reaction types. Once a team has successfully grouped the chemical equations, direct them to write the chemical equations in their notebooks in the same groupings as the strips of paper. They should then prepare to explain to the class how they developed their classification schemes. As a class discussion, ask teams to explain the development of their classification schemes. Encourage all students to share their thought processes and point out that their different methods were influenced by each student’s creativity and interpretation. Indicate that this is a normal aspect of the nature of science. Also, point out that the students combined their previous classifying skills with new knowledge about chemical reactions to develop their classification schemes. Indicate that this is a common aspect of the scientific inquiry. Homework Read Section 10.2 (pages 284-291) and match the five classes of reactions with the five groups you made in class. On the “Reaction Types Worksheet,” identify each reaction by its class. Day Two Bell Ringer: On the board or overhead, write: “Last night you read about five types of chemical reactions. How would you describe each one in a sentence? Write a description of what happens in each type of reaction: synthesis, decomposition, single replacement, double replacement and combustion.” Review Check in the worksheet students completed for homework, then go over the correct answers as students check their work. An answer presented via the overhead projector can speed up this process. Discuss any items that the students found difficult and provide clues to identifying the five reaction types as necessary. Example clues: one product—synthesis one reactant—decomposition one lonely element and one compound on each side—single replacement two compounds on each side—double replacement oxygen as reactant, oxides as products—combustion Tell students that you are going to read them a story. Ask them to put down their pens and relax. (You may wish to dim the lights and light a candle to “set the mood” for the romantic story. You may also enhance your “street creed” by acknowledging that the story is very hokey, but that it does the job!) The Story One day, as you are walking out of school after your last class, you see the most attractive person you’ve seen in a long while. You ask your friends if they know who this person is. One of your friends knows the person and tells you the attractive person’s name. In fact, your friend is good friends with this person’s friend and offers to find out what lunch period this new apple of your eye is in. The next day at lunch, your friend takes you over to the table where the person is sitting and introduces you. You and your friend sit down with the attractive person and friend, and the four of you talk and enjoy lunch together. You find that not only is this person cute, but funny and enjoyable to be around. The person seems to enjoy your company, too, and you exchange e-mail addresses and phone numbers. That night, you get a phone call from the person and talk for a while. You enjoy it and learn that the two of you have a lot in common. Before long, the two of you are a couple, and you decide to go to the next school dance together. You’re so excited—this person is so much fun to be around that you’re sure it’s going to be a great night. The day of the dance comes and everything is perfect—your clothes, your date’s clothes, the decorations, the music—it’s all great and you’re having a terrific time. The DJ puts on a slow song and you find that you really like dancing in your date’s arms. The next song is a fast one and you and your date join a group of friends dancing nearby. For part of the song, you find yourself dancing across from your best friend’s date, while your friend is dancing beside your date. It’s fun to be part of such a casual group, but it’s also nice to go back to your own date for the next slow song. Later in the evening, you are warm and thirsty, so you head out into the hallway, where there is punch to drink. Your date stays behind, fast-dancing as part of a large group. When you return a few minutes later, the music has changed, and you can’t find your date anywhere. Then you spot your date, across the dance floor, slow-dancing with someone else! You feel confused, angry and foolish standing alone on the dance floor, so you hurry back out into the hall. When your date comes looking for you a few songs later, you tell your date what you saw and how you felt. The conversation turns into an argument, with heated emotions and raised voices. You and your date decide that perhaps you had different ideas about the evening and you break up. A few weeks later, you’re walking out of school, when you see ahead of you the most attractive person you’ve spotted in a long while… Ask the students what the story had to do with chemistry. The students should be able to identify the parts of the story that represent the five classes of chemical reactions. Synthesis: two students joining as a couple Double replacement: dancing with the other couple Single replacement: your date dancing with someone else Combustion: the argument Decomposition: breaking up Discuss with students the ways in which the analogy works and the ways it doesn’t. (Example of a flaw—combustion releases energy, just as the argument was heated, but the argument doesn’t have an outside reactant to represent oxygen.) Direct the students to individually write a story that contains analogies for all five types of fundamental chemical reactions. The story can be about whatever you want—a few ideas are football games, telephone calls and cooking—except that you cannot copy any analogy you heard in the class story. The story doesn’t need to be long—just long enough to include comparisons to all five reaction types. Homework Complete your story, then highlight and identify the five (5) analogies for the five (5) fundamental chemical reactions. Then write a sentence or two explaining the flaws/weaknesses in your analogy—how is this story not a perfect comparison to real chemical reactions? Modifications Advanced classes/students may complete the equation-sorting exercise quickly and have time to begin working on the first day’s homework, the “Reaction Types Worksheet,” in class. Students who need assistance organizing their thoughts while reading may benefit from a worksheet with a blank table to organize their notes. A sample is provided at the end of this lesson. Limited English Language students may need copies of the story to help improve comprehension. Assessment Students’ understandings of the five types of reactions are assessed through: Observation and discussion of their classification schemes Correctly completed classifications of mathematical and chemical equations Solutions to “Reaction Types Worksheet” Analogy stories Rubric for analogy story: Reaction Type Analogy Story _____ / 5 all reactions accurately are represented _____ / 1 complete sentences _____ / 2 grammar and spelling _____ / 2 creativity and story flow _____ / 10 total points Classifying Chemical Reactions Worksheet Name: ____________________________________ Period: _____ Classify each reaction as synthesis, decomposition, single replacement, double replacement or combustion. The equations are not balanced. 1. PbCl2 + AgNO3 Pb(NO3)2 + AgCl 2. NH3 + HCl 3. AlCl3 + Na2SO4 4. Zn + S 5. Al2(SO4)3 + BaCl2 6. Al2S3 7. H2SO4 + Fe NH4Cl ___________________________ ___________________________ Al2(SO4)3 + NaCl ZnS ___________________________ ___________________________ BaSO4 + AlCl3 Al + S ___________________________ ___________________________ H2 + FeSO4 ___________________________ 8. C12H22O11 + O2 CO2 + H2O ___________________________ 9. Mg(OH)2 + H2SO4 MgSO4 + H2O ___________________________ 10. NaOH + Na2SO4 + Cu(OH)2 ___________________________ 11. C4H12 + O2 12. Fe + O2 13. Mg3(PO4)2 + H2 14. NH4NO3 15. Cl2 + KBr CuSO4 H2O + CO2 Fe2O3 ___________________________ ___________________________ Mg + H3PO4 ___________________________ N2O + H2O ___________________________ KCl + Br2 ___________________________ Classifying Chemical Reactions Reading Notes—Section 10.2 (pages 284-291) Reaction Type Synthesis Decomposition Single Replacement Double Replacement Combustion Sample Equation Clues for Recognizing Reaction Type Teacher Notes: Synthesis—two reactants join to form one product **clue: one product A + B AB 2 Na + Cl2 2 NaCl Decomposition—one reactant breaks into several products **clue: one reactant CD C + D 2 H2O 2 H2 + O2 Single Replacement—an element in a compound switches places with a lone element **clue: lonely element on each side A + BC AC + B 2 KBr + Cl2 2 KCl + Br2 Double Replacement—two compounds swap their positive and negative parts **clue: two compounds on each side AB + CD AD + CB AgNO3 + NaCl AgCl + NaNO3 Combustion—an energy-releasing reaction with oxygen to produce one or more oxides **Clue: O2 is a reactant, products are compounds with oxygen in them AB + O2 AO + BO C + O2 CO2 Mathematical Equations Challenge Names of Group Members: __________________________ ________________________ Challenge Question: The mathematics professors have generally agreed that there are five (5) different possible types of “addition” expressions. Using your observations skills, I am sure that you will recognize similarities and subtle differences in each of the 21 mathematical equations. Can you classify each of these mathematical equations into the correct five different categories? Hints: Use scissors to cut out each equation from the accompanying sheet. No mathematics textbooks or calculators can be used at any time. Report: In the space below, your group’s reaction classification scheme should contain: a general description of each category two sample equations for each category Classification Scheme: Mathematical Equations Challenge 1. 28 + 54 = 82 2. 22 + 55 = 25 + 52 3. 40 + 22 = 44 + 18 4. 75 + 12 = 15 + 72 5. 131 = 44 + 18 + 69 6. 14 + 6 = 16 + 4 7. 45 + 2 = 42 + 5 8. 79 = 52 + 27 9. 20 + 58 + 16 = 94 10. 17 + 42 = 12 + 47 11. 33 + 29 = 44 + 18 12. 71 + 3 = 73 + 1 13. 62 + 49 = 111 14. 46 = 18 + 15 + 13 15. 12 + 50 = 18 + 44 16. 35 + 20 + 52 = 107 17. 59 + 48 = 49 + 58 18. 38 + 24 = 18 + 44 19. 67 + 1 = 61 + 7 20. 36 + 13 = 33 + 16 21. 48 + 14 = 44 + 18 Chemical Equations Challenge Names of Group Members: _________________________ ________________________ Challenge Question: Can you classify each of these chemical reactions into five different categories? Hints: Use scissors to cut out each reaction from the accompanying sheet. Do NOT balance the equations as this may confuse you. No textbooks can be used at any time. Report: In the space below, your group’s reaction classification scheme should contain: a general description of each category two sample equations for each category Classification Scheme: Chemical Equations Challenge 1. SiO2 + Mg Si 2. SO2 + O2 SO3 3. CaCl2 + K3PO4 4. CO O2 5. H2CO3 6. CaCO3 + HCl H2CO3 + CaCl2 7. C4H10 + O2 H20 + CO2 8. P4O10 + H2O H3PO4 9. Cu 10. NCl3 11. Ag2SO4 12. NaHCO3 13. Li 14. H2SO4 15. BaO 16. C10H22 17. Al2(SO4)3 + 18. CuBr + Br2 19. CH4 + O2 + N2 + Cu(NO3)2 (NH4)2SO4 + H20 + + + Hg Cl2 NH4I Na2CO3 LiOH Ca + KCl H2O + CO2 + + CO2 HgNO3 + MgO Ca3(PO4)2 CO2 + + HNO3 O2 + + H2O + H2 CaSO4 + Ba(NO3)2 CO2 Ca(OH)2 + AgI H2 + H2O Al(OH)3 CuBr2 CO2 + H2O H2O + CaSO4 Instructor's Notes Chemical Equations Challenge Frustration with the Traditional Lecture Approach During my first year of teaching I taught this material in a traditional lecture approach, and I was very disappointed at the number of students who were not able to classify these chemical reactions despite having been exposed to my "brilliant" lecture. I felt that my organized, carefully scripted lesson should -have been an automatic success. How could they not see things exactly as they had been told, exactly as their own notes told them? I decided to take a chance during my second year of teaching. Would they be able to see the patterns and trends in these chemical equations before I assigned the formal terms synthesis, decomposition, single replacement, double replacement and combustion? I was nervous at first but was absolutely stunned at how easily my students were able to do this. I have experienced success with this in every class (regular or honors), and I am now a firm believer in the constructivist belief of "experience it first, name it afterwards." I am extremely proud of creating this activity because I enjoy teaching the types of reactions after this activity. To see them nodding along with me as the formal names are introduced to them, and hearing comments like "We saw that one!" is much more pleasant than hearing the usual "How did you know it was a single replacement reaction again?" Groups and Incentives to Finish Early This cooperative activity takes about 30-50 minutes to make sure that everyone gets finished. I score this as 10 points, and award the extra credit to the first three groups to finish. Three extra points for being first, two points for the second group, one extra for being third. This keeps the groups focused and minimizes the chances of groups merging. Teacher's Role in the Classroom Once the groups have been assigned and students understand the task, the teacher becomes the facilitator. The teacher can walk around the classroom to monitor the students' progress. Teachers should remain silent because students will often seek assistance through a teacher's facial cues, comments, etc. To keep the groups on track, a teacher cannot simply let the students go without some timely encouragement. I often make the following comments to maintain a productive classroom atmosphere: "We are looking for patterns and trends.' "What do you start with? What do you end with?' "Think about it... with only five different groups, they can't be too element specific." "Is this right?" "Well, explain to me why you put these together." Student justifies the categories... "Ok, I like this one and that one, but I don't like this group here." THEN WALK AWAY. "There seems to be one equation that just doesn't fit in this group." THEN WALK AWAY. Two extensions for groups who finish early? If a group finishes earlier than the rest, ask them to look closely at the double replacement category. If they truly see the patterns, they should be able to predict the products of the reactions. I will then give them both starting materials of a double replacement reaction. "Give me the products and then I will believe you really see what I see." Another extension is for students to write the formal names of the types of chemical equations on the chalkboard. Ask the students to try to match the formal name with their five different categories, "Don't be surprised if your students get all five correct in their first try!!! Shocking, but true!!! Why aren't the equations balanced? The equations provided are not balanced and students are specifically told not to balance them. This helps to mini-mire the potential for distraction since the coefficients do not influence the classification of the chemical equations. Teachers who feel obligated to have the equations balanced can ask students to do this after completing the activity. Recommended Classification Scheme: There are many different ways to categorize chemical reactions, but the equations were specifically selected with the classification scheme described in a popular chemistry textbook.1 Synthesis: A chemical reaction with only 1 product. SO2 + O2 SO3 Decomposition: A chemical reaction with only 1 reactant. H2CO3 CO2 + H2O Single Replacement: An element and compound produce a new element and new compound. H2SO4 + Ca CaSO4 + H2 Double Replacement: Two compounds react by exchanging ions producing two new compounds. NaCl + AgNO 3 AgCl + NaNO 3 Combustion: A hydrocarbon and oxygen react to produce carbon dioxide and water. CH4 + O2 CO2 + H2O R. C.; Smith, R G.; Price, J. Merrill Chemistry, 1995, Glencoe/McGraw-Hill, p. 228-230. Flinn Scientific Foundation Chemical Equations Simulation for Teachers Synthesis (Only one number on the right side) 35 + 20 + 52 = 28 + 54 = 82 62 + 49 = 111 107 Decomposition (Start with one number and end with many) 46 = 18 + 15 79 = 52 + 27 131 = 44 + 18 + 13 + 69 Double Replacement (Switch ones' positions to form two new numbers) 17 + 42 = 12 + 47 22 + 55 = 25 + 52 59 + 48 = 49 + 58 Single Replacement (Single digit replaces one's position) 45 + 2 = 42 + 5 67 + 1 = 61 + 7 14 + 6 = 16 + 4 right side Combustion (Numbers on the 40 + 22 = 44 + 18 12 + 50 = 18 + 44 33 + 29 = 44 + 18 Flinn Scientific Foundation are always 18 and 44)