Economic and Environmental Geology Notes (Geos 340)

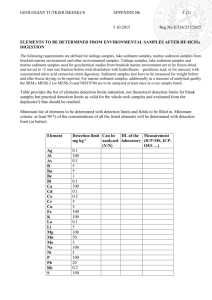
Economic and Environmental Geology Notes (Geos 340)
Introduction, syllabus. Field trip planning
Processes.
Conceptual
Chemical separation (oxidation/reduction, dissolution/precipitation, examples Fe oxidation, write reaction, multiple examples dissolution/pption)
Physical separation (gravity-density settling, magnetic, sorting by stream/wind)
Biological activity (changes in solubility/precipitation either direct or indirect, production of fossil fuels, examples) notes: properties important both for ore/resource production and for environment. Also define pollutant distribution and migration.
Earth Processes (radioactive, internal heat driven largely)
magmatic processes (energy sources, phase equilibria, partitioning, fractional crystallization, settling, Cr layered intrusions combination of fractional crystallization and settling-magma injection, magma mixing, double diffusive mixing, crystal fractionation, convection, density currents, nucleation densities, crystallization in thermal gradients, Ni compatible, found in mafic-ultramafic - F, Be, Li, Ree incompatible found in pegmatites - explain relation to degree of fractionation from mantle composition) note on double diffusive convection: The best known double-diffusive instabilities are ``salt-fingers'' (Stern, 1960).
These arise when hot salty water lies over cold fresh water of a higher density and consist of long fingers of rising and sinking water. A blob of hot salty water which finds itself surrounded by cold fresh water rapidly loses its heat while retaining its salt due to the very different rates of diffusion of heat and salt. The blob becomes cold and salty and hence denser than the surrounding fluid. This tends to make the blob sink further, drawing down more hot salty water from above giving rise to sinking fingers of fluid. There are many other different forms of double-diffusive convection. notes for Layered basic intrusions and ophiolites
Layered basic intrusions occur throughout the geologic record on the continents. They are generally large, funnelshaped bodies of crytalline igneous rock that as an overall composition close to that of a mid-ocean ridge basalt
(though this was not known at the time these rocks were first studied). More importantly, most of them apparently crystallized in situ from a single batch of very primitive magma. During the 1950's, this allowed the evolving field of experimental petrology the opportunity to test the phase relationships they were determining for basaltic systems.
Another and very important aspect of layered basic intrusions is their sulfide mineralization, which accounts for some of the richest Cu and PGE ore deposits in the world. The layering in an LBI can take many forms, the most important are:
Mineral or modal layering
Grain size layering
Cryptic layering
The earliest theories for the origin of layering in igneous rocks envisioned the gravitational settling of crystals to the floor of a magma chamber, as pictured at the right. However, there are a variety of problems with this model. The most obvious is that the density relationships often do not allow it -- plagioclase, which is lighter than the magma is often found in alternating bands with olivine, which is denser than the magma.
1
Modern theories for the origin of layering take other processes into account such as varying nucleation rates for the different mineral phases, crystallizazion along thermal gradients and double diffusive convection.
Despite the fact that actual crystal settling probably has little to do with the formation of igneous layered rocks, these rocks are generally still refered to as cumulates .
Metamorphic process ( contact , oxide and sulfide ores, Au, Ag, hydrothermal from intrusion, more water rich sialic more mineralization often, and limestone more reactive than shale or ss, emeralds, ruby, sapphire in these environments, very rare, exogenetic region vs endogenetic, zonation [with distinct mineral assemblages and associated ore types]. regional , dewatering=hydrothermal, remobilization, effect of metamorphism on physical properties
(building stone) as well as chemical, garnets refractories like Al-rich minerals, and abrasives like corundum, metamorphic grade) zonation due to rock type, tectonic setting. from Kula notes.
Hydrothermal process (driven by magmatic or metamorphic, generally T dependent, ppt/solution depends on T, pH, Eh, P, Cl, S, Na, K, CO2; discuss solubility, effect of H+, effect of Cl making soluble species in water, leChatlier principle. Get gradients and zones due to systematic variations in T, etc. Epithermal([Au, Ag, Sb], mesothermal[Cu, Pb, Zn], hypothermal[Sn, W], note commonly observed associations. Also wall rock alteration zones, metasomatism: potassic zone (K-spar), phyllic zone (micas), argillic zone (clay minerals, no Ca-rich minerals, and no
Kspar), propyllitic zone (least distinction from unaltered, has carbonates, no biotite)
Notes on reactions (concepts also true for water pollutants and sediment)
PbCl2 = Pb+ + 2Cl- or PbCl2 = PbCl- + Cl-
S precipitates, acidity affects, warmer = more soluble. transport often as Cl, pption as S, hydrothermal zoning often is controlled by Cl, not S, solubilities.
Black smokers, dissolved metals and sulfur ppt where encounters sea water, changing T, pH, etc.
Some ore deposits presently being mined on continents thought to have originated in such an environment. Talk more about when talk about Cu ores.
Diagenesis: low-T modification, recrystallization, cementation. sometimes mineral changes such as dolomite.
Cellulose (plant) = loss of S, P, H, concentration of C, = ranks of coal (lignite, bituminous, anthracite)
low cellulose marine plankton (high in lipids, some proteins and carbohydrates)= kerogen = petroleum, as slowly heated and compressed.
2
methane: both biogenic (CH4 produced as bacteria break down organics, swamp gas, flying saucers), and thermogenic (CH4 produced as more complex hydrocarbons break down with heating)
Earth Processes (solar, external heat driven largely)
weathering: climate, pH, Eh, T (effect of H+ on feldspar stability)
2KAlSi3O8 + 2H+ + 9H2O = H4Al2Si2O9 (kaolinite) + 4H4SiO4 (silicic acid) + 2K+
(direction of reaction when decrease pH, increase H+)
Leaching, chelating agents, 2ndary concentration processes (redox, Cu, U), Clays common product, also bauxite from Easterbrook. Note that this is also important for pollutants, solubility determines mobility.
Comment on bauxite (Al rich) versus laterite (typical red tropical soil, Fe rich) sedimentation: sorting, river, wind, energy/density, sometimes Eh! (U in pC), placer deposits.
Crystallization from surface water, usually evaporation, can also be mixing zone where activities of components change. NaCl(mined for salt), CaSO4(H2O), KCl (mined for fertilizer).
Flux of sea water to produce concentrations (such as onto shallow continental shelf or restricted basin), phosphorous from teeth bones on continental shelfs, Often pption is biologically enhanced (e.g. manganese nodules probably related to microbe activity).
Complex reactions where polluted stream encounters other water.
3
Plate Tectonics: environments: subduction, (andesitic, Cu); divergence (basaltic, alkali basaltic, REE, Ni, Cr),
Continental (Granitic, Au, REE, Be). also affects landscape, erosion, swamps, rivers etc. from Shackleton, 1986 also include diamonds, kimberlites, at continental divergent boundaries, and layered intrusives at oceanic and continental divergent boundaries, as well as granites and REE, other incompatibles in continent interior.
Petroleum and related products:
Language and economics oil not randomly distributed is in sedimentary rocks largest reservoirs are in younger rocks (less likely as rock gets older) usually less than 10000 feet about 60% of oil is in 2% of fields pools, fields, provinces. lead time: time between when you decide to do something and when it is in operation (about 5-7 years for natural gas) (note: conservation effectively produces oil without lead time) sandstone is 30% porosity, but if grains touching the permeability is too low to extract
Proven reserves: how much can get out at present day costs and technology
Ultimate reserves: how much is there and ultimately recoverable
Probably reserves: How much can get out by special extraction methods
Possible reserves: How much could get out by some imagined but possible incremental jump in science and technology
Cost of energy (e.g. per kWhour) has been decreasing since 1920’s. Now much cheaper than
1920’s or 1980’s for example. wasting assets: non-renewable resources which, once they are gone, they’re gone. As amount decreases, cost will theoretically increase to prevent complete consumption (draw cost versus demand diagram, at some point, oil will no longer be economically viable as the world primary energy source, also used for chemicals, plastics, herbicides, pesticides, lubricants)
4
Prediction of discovery rates: in general, the rate of discovery declines as the number of feet drilled increases (can’t assume discovery rates will remain the same as today). Extraction efficiencies are also difficult to predict, depending on technological advance.
Fossil fuels presently about 80.3% of US energy use. advantages: low cost, ease of transport (for oil in particular), existing infrastructure,
Disadvantages, political, wasting asset, sulfur and other pollutants
There is no energy source that is free of environmental cost (e.g. manufacture of solar cells often has more environmental cost than use of oil).
5
Carbon Dioxide is introduced into the atmosphere by many means, including burning of fossil fuels. This introduction of excess carbon dioxide into our atmosphere has the potential of effecting our climate by "greenhouse" warming. However, there are also many processes that remove carbon dioxide from the atmosphere. Like other problems in chemical differentiation (discussed in a previous section of this course), the element carbon is not created or destroyed in these chemical processes, but rather it is partitioned into different phases. The chart below illustrates in a schematic way some of the processes involved, and the reservoirs that carbon is stored in on Earth. The percentages of carbon in each reservoir is shown (upper number in parentheses), as well as the percentage of human-released carbon that is thought to be in each reservoir (lower number in brackets).
1: burning of carbon or carbon-containing molecules: C + O2 = CO2.
2: respiration, the oxidation of carbon by living things to generate energy
3: weathering of rocks that contain carbon to release CO2
4: heating of limestone to produce Calcium for use in cement. This releases CO2.
A: burial of living things in sediment and conversion of living carbon to coal or oil.
B: Photosynthesis takes carbon from the atmosphere and fixes it in sugars, cellulose, etc
C: Animals eat plants
D: CO2 partitions between water and air.
E: CO2 in water mixes between the upper and lower parts of the ocean.
F: living things and inorganic processes extract CO2 from water as sediment.
G: sediments are lithified, becoming rock.
H: CO2 partitions between the air and ice (as snow falls, for example)
I: Ice containing CO2 melts
J: rivers flow into oceans. footnote: (a), the carbon in terrestrial plants includes carbon present in animals.
6
Oil chemistry: natural gas (CH4) , wet and dry (wet contains other gases such as ethane (C2H6), propane
(Ch3H8), butane (C4H10), (draw pictures of C and H chains), CO, CO2, H2S, He, N2, H2) petroleum, hundreds of hydrocarbon compounds. three major types of petroleum:
Paraffin rich (light crude oils): C n
H
2n+2
, straight or branched chains fom methane to paraffin wax: products include natural gas, LPG, gasoline, kerosene
Naptha rich (are rare crudes): C n
H
2n
, including carbon ring: products include heavy fuel oils and asphalt residue
Aromatic hydrocarbons (heavy crude oils): C n
H
2n-6
, at least one benzene ring: products include benzene and toluene. from Shackleton
Sweet (low S) vs Sour (higher S): Talk about environmental impact, S+H = H
2
S, acid rain.
Temp effects: biogenic (methane) and thermogenic (cracking, organic to kerogen and petroleum and then conversion of kerogen to thermogenic wet gas), diagenesis (<50C, a few hundred meters depth), catagenesis (50-150C, up to about 3500-5000m, to 1.5kb), metagenesis ( kerogen=insoluble organic matter
Sequence: lipids, proteins, carbohydrates[which contain C,H, P, S, N, O compounds] to CH4 and residue, then to kerogin and petroleum, then to petroleum and wet gas, then to dry gas. Product is also affected by the type of kerogen (depends on type of organic source, plant or animal, terrestrial or marine). cracking=decreasing number of C in chains.
7
Degradation also plays a role in petroleum production: from craig et al 2001 from Shackleton oil accumulation source rock (vitrinite reflectivity indicator of maturity of organic sediment, comparable to coal rank, it is the reflectance of vitrinite particles in source rock.) reservoir rock (porous and permeable) generally >30% porosity to get adequate permeability
(more than close-pack sandstone). More permeable = sandstone, conglomerate, limestone.
Petroleum resides in pore space. traps: Structural (show anticlinal, fault, salt dome-explaining how upturn caused by plastic diapir), stratigraphic (show lens, transg-regression sanstone ‘V’ tilted, impermeable rocks above an angular unconformity). Cementation trap (explain differential cementation), hydrologic trap can’t have too much fracturing of the impermeable rock.
8
from Tarbuck and Lutgens.
Bore Hole Logging:
More clays, more gamma, lower porosity and higher density, higher velocity
From Shackleton.
9
Coal accumulation:
Organic production vs decay rate (T, H2O, pH, affect both, bacterial activity affect decay, consider balance of production vs decay) consider subsidence, transgression rates to make room for deposition cyclothems, alternating near sea level changes most coal in carboniferous, or late Cretaceous to early Cenozoic (hell creek and fort union groups) cellulose in Devonian, thus no coal before that
Coal rank: increasing heating, pressure, loss of O, P, N, S, H, increasing C craig et al.
10
Shackelton craig et al
Coal Types: humic: from organic debris that produces peat (trees), begins as peat, most abundant, may be autochthonous sapropelic: fine grained from algal accumulations in O2-poor pools (similar to kerogen), may be allochthonous, examples: boghead and cannel coals.
Coal particles, Macerals (like coal minerals) vitrinite: jellified plant residue, translucent to golden, some retaining some cell structure
Exinite: smaller, resistant plant debris such as spore cases, algal material,
Fusinite: charcoal like opaque carbonized cell structure micrinite: opaque residue sclerotinite: fungal residue.
11
or
Anthraxylon: translucent material from woody parts of trees
Attritus: opaque material and translucent material that is not anthraxylon in situ (autochthonous): site evidence pg 21 accumulated in place of growth
evidence:
plant debris not sorted
underlying rock has roots penetrating
little to no clastic material
existing modern thick peat accumulations in place
stratigraphy suggests accumulation at or near sea level (cyclothems)
Drift (allochthonous): site evidence pg 21. especially ash connection with clastics material transported to location
evidence
underlying sediment doesn’t fit model of where plant debris might accumulate
sedimentary structures:humus and plant material rafts, sedimentary structures, cross bedding, soft sediment deformation etc
fine grain size (e.g. cannel coals), high ash content (silicate material)
not all coals are associated with cyclothems
Heavy Oils and Tar Sands: bitumen, so viscous it won’t flow. very dark (includes asphalt) volatile loss of shorter hydrocarbon chains, oxidation of shorter chains, also organics of terrestrial origin typically have longer chains, bacteria may degrade shorter chains, or light fraction driven off or gasified by heating.
Oil Shale: kerogen (waxy, long hydrocarbon chains), liquefies when heated above 500C. mostly lipid-rich e.g. blue-green algae, but O2-poor fresh and saltwater with high organics. Generally differs from bitumen in that it has not been heated sufficiently to cause organics to break down in process of forming petroleum....bitumen on the other hand has been so heated, but through near surface fractionation has lost its lighter materials.
12
Economic and Environmental Geology: Lab 1
I recommend doing the labs in groups of 2 or 3, so you can discuss and debate. However, each person needs to spend 20-30 minutes looking at the thin sections.
Do the introductory physical geology oil trap exercise (handed out previously)
Examine the 9 samples of coal (labeled): Describe each, looking for significant features, indications of sulfur or ash content, macerals, rank, whatever. List them in order of rank in your report.
Examine the 3 samples of petroleum. Note color, viscosity, odor (smear a bit on glass to get in idea of color and viscosity).
Examine the 4 thin sections under microscope (use both petrographic and dissecting-type scopes). Look for features of the organic material, maceral types, sedimentary structures and features, fabric, anything you can see). Samples are from an organic-rich shale in the lower-most
Hell Creek formation (upper Cretaceous). These are pretty interesting thin sections, so don’t damage or lose them! P709-SB-8, 712-BB2-4. CHS-712-BB2-5 (2 slides) NEEDLESS TO
SAY: NEITHER THIN SECTIONS NOR SCOPES ARE TO LEAVE THE ROOM!!!
Note: 3 of the 4 new thin sections are a bit too thick, so quartz shows higher interference colors in many cases. some things to see in P709-SB-8, from unit 5b at Concordia Hadrosaur Site : interlaminated organic and silty layers, convolute laminae, siltier lenses, blobs of variable matrix-quartz indicative of soft sediment deformation, carbonized and hematized organics
(fusinite, micrinite), small pieces of ‘amber’ (vitrinite), Qz, muscovite, (NOTE: I haven't spotted any fusinite yet in the new thin section, but if you find some, point it out to me!) some things to see in 712-BB2-4, and 712-BB2-5 from bone conglomerate unit 6 at BB2 CHS: angular Qz, 0.2mm, authogenic growth around Qz crystals, resembles matrix, mud clasts
(intraclasts-0.5-2mm, larger in BB2-5, also can see laminations in some muclasts), bone fragments (in at least 1 on the sections), vitrinite, translucent organic fragments with growth rings (anthraxylon), some carbonized micrinite, fusinite--(haven't spotted any fusinite yet in these new sample), woody fragments, muscovite, phlogopite, mica, feldspar (Carlsbad twinning, also albite twinninsg). Highly birefringent clear mineral-calcite? calcite cement, where present.
13
Economic and Environmental Geology : review of mapping and cross-sections, and application to oil traps. (these are taken from introductory physical geology classes.)
The following questions are modified from an introductory geology test by Andre Lehre, 2000.
What are the oldest and youngest rock units in the map area?
Are there any unconformities shown on the map area? Explain.
What type of structure is the main geological structure?
On a separate sheet of paper, sketch a cross-sectional view from the upper left to the lower right of the map area.
Some speculators are forming the Silver River Oil Exploration Company. Their land is shown on your geologic map. You wonder if you should buy in.
Discuss the property in terms of the following:
Are there potential source rocks (explain)?
Are there potential reservoir rocks (explain)?
Is there a trap (explain)?
Is there any other potential trap outside the Silver River Property
(explain)?
On a separate sheet of paper, sketch a cross-sectional view from the lower left to the upper right of the map area, showing locations of any potential traps.
14
15
Fission and Fusion unstable = higher energy than stable forms (see Fig). When go from high energy to low energy, energy is released. In general Fe is most stable nucleus. By combining smaller nuclei, or splitting larger nuclei, can often get an energy return. Some unstable nuclei will spontaneously decay at a characteristic rate (radioactivity). Gamma decay, Beta decay, Alpha decay.
Fission: Some unstable nuclei can be split into two smaller nuclei, such as by bombarding with neutrons.
235
92
U + n =
141
56
Ba +
92
36
K + 3n + energy : neutrons + protons number is conserved. chain reactions and the purpose of the control rods (mousetrap and ping-pong ball demo?) need a means to control chain reaction = control rods (boron-steel).
Also, need to slow down neutrons so they interact more (collision-capture) with nuclei. Do this with a moderator (C, deuterium, plain water). separating 235 U from 238 U. What is difference? mass. separate by gaseous diffusion. Mention mass spectrometer. neutron bombardments and the production of unstable nuclei (chart of the nuclides?) (sometimes these unstable nuclei are themselves fissionable and can be used as fuel, such as the production of
233
U from
232
Th exposed to neutron bombardment) or
239
94
Pu. Breeder reactor, fast neutrons
(no moderator),
238
92
U +n =
239
92
U - Beta =
239
93
Np;
239
93
Np - Beta =
239
94
Pu which is fissionable
(also example of Beta decay).
Strong political opposition in the late 1970’s and early 1980’s effectively sidelined fission power in the US, confirmed by accident in US resulting in a minor leak of radioactive material (three mile island) and major accident in USSR. Legal challenges and increasingly high legal hurdles made it cost ineffective. US discontinued development of nuclear power by the mid 1980’s. Countries that lack oil and coal have developed it more extensively (e.g. France and
Japan).
Talk more about geological concentration of Uranium ore when talk about ore-forming processes. But key is valence state of U: U
4+
insoluble, U
6+
soluble. Will concentrations occur in oxidizing or reducing conditions? (Talk about it, try to figure out)
Fusion: runs the sun
2
1
H +
2
1
H =
3
2
He + n + energy very high energy reaction, occurs at extremely high temperatures and pressures (like in the sun) difficult to contain, matter exists as plasma at those temperatures (what destroyed shuttle last winter).
2 containment approaches: toroidal magnetic field, or inertial approach in which bombard target and shock waves, heat waves contain material. parallel reaction: 2
1
H + 2
1
H = 3
1
H + n and 2
1
H + 3
1
H = 4
2
He + n.
Different potential fusion reaction, using lunar
3
He:
16
2
1
H (deuterium) +
3
2
He =
4
2
He + p + energy (proton easier to contain than n, less radiation problem, less radioactive waste).
Other energy sources:
All energy sources are nuclear: sun is nuclear (fusion), geothermal is nuclear (radioactive decay), most others are ultimately from the sun.
Solar (direct, electricity): environmental cost of manufacturing cells, not easily transported
Weather-related: Hydroelectric, wind, wave (hard to transport, not where want it often) remnant kinetic energy: tides geothermal: corrosion, disruption of natural areas
Biomass (still get CO2, competes directly with food production and timber, etc)
All have potential to affect environment in unexpected ways. Reroutes energy otherwise used as part of overall weather-climate system.
Class discussion on energy sources.
LAB: Activity for Yucca Mt site (Xerox copies): talk about differences among low level waste, high level waste, and transuranic waste, and possible disposal sites. Also talk about difference between storage and disposal.
17
Protons on Y axis, neutrons on X axis
Beta decay- up one and to the left one alpha decay - down two and to the left two blue: half life greater than 5x108 green: half life between 30 dyas and 5x108 red: half life between 10 min and 30 days yellow: half life less than 10 min square open box: alpha decay fillled box beta decay, or electron?
18
ORE FORMING PROCESSES
Formation of mineral deposits (think of processes that could concentrate ores.... come up with 2)
2 key things: transport (need sufficient concentration and transport volume), and change of conditions to cause dissolution/precipitation reactions.
Fluids are almost always involved (facilitate transport). Could have vapor transport. Solid transport is relatively slow, thus less significant.
A. mechanical accumulation of materials from a fluid medium....e.g. placer deposits
B. chemical precipitation from a medium
1. crystallization from a magma, layered intrusives (Cr), partitioning, concentration by fractional crystallization, liquid-liquid immiscibility (show immiscibility curve on a phase diagram between phase A and B, talk about sulfide immiscibility, a function of S concentration.)
2. precipitation from an aqueous fluid, e.g. vein deposits, evaporates, supergene enrichment, etc., remind of LeChatelier principle. This is most important process on Earth
3. Precipitation from a vapor phase, coal burning, C, Cl, vapors on the Moon?
4. Solid-solid reactions, metamorphism, replacement (usually metasomatic though), unmixing (introduce to immiscibility in solids? - example of perthite, exsolution of Na and
K feldspars, show phase diagram)
Most ore-forming processes on earth are related to water (no water on the Moon, other processes?)
Fluids (usually hydrothermal if produce ores):
Ground water (generally acidic and oxidizing above water table, alkaline and reducing below)
(meteoric water
connate water)
volcanic exhalations, Juvenile water (magmatic water) (usually mixed with meteoric water, usually high S, not so much metals, usually low Ph, acidic, and low Eh, reducing. gases contain Fe, Mn, Cu, Pb, Zn, S. Also Cr, Ni, Cu, Mn, Pb, Fe in East Pacific Rise.
metamorphic water (Au deposits in pC terranes may be from metamorphic water, not a lot of stuff in metamorphic water usually, but metamorphic water can MOVE elements whose source is other rock etc.)
sea water (maintain rough concentration balance through time, get evaporates, some Fesulfides in Black Sea, Manganese nodules (may have volcanic association).
geothermal brines (>300C) (water heated by deep circulation, geothermal gradient high or magma reservoirs). Higher Temperature raises solubility, larger mass transport and if T decreases it will precipitate. Can get ores of Cu, Pb, Zn, Ag.
General characteristics of ancient ore fluids composition: N-Cl-Ca brines, salinity <2% to 60%, temperature: 50-500C, usually 200-500C.
Hg = <200C
19
epithermal Au, Ag = 130-330C base metals = 75-500C porphyry Cu-Mo = >300C pressure: usually <20km (5-6kbars) pH: buffering by ions keeps from being extreme, probably 5.5-8 mostly driving energy: magmatic heat, metamorphic heat, gravity sulfide content: usually low in S ions compared to metals.
Concentrating Processes leaching weathering and sedimentation supergene enrichment (water table effect, due to descending fluids) hypogene enrichment (due to ascending fluids) metamorphism (usually disperses) granitization vein migration and changes in T, P, fO2, pH etc. gravity settling filter pressing
20
Fe Ores:
Magmatic: layered intrusives have magnetite that can accumulate much like Chromite. (e.g.
Bushveld complex in South Africa). Not major source of Fe presently, but get V from there.
Consider V partitioning: can exist in 3 valence states V3+, V4+, V5+. As V3+ it can substitute for Fe3+. The other valences don’t fit as easily into the magnetite structure (need a chargebalancing vacancy, such as 3 V5+ substitutes for 5 Fe3+, leaving 2 holes, or a V5+ substitutes for a Fe2+ and a Fe3+ leaving one hole. (Q: will partitioning increase or decrease with more
O2)?
Consider the following experimental results (from Dave Lindstrom PhD. thesis)
Will V accumulate in magnetite most readily where fO2 is low or high?
Metamorphic (interaction of igneous fluids with wall rock): limestone especially easily replaced by magnetite or hematite. Usually small.
Hydrothermal (sea floor volcanism): Black smokers, cooling of hydrothermal water where encounters sea water, oxidation, pH changes.
Weathering: remind of the solubility graph from before. Can leave Fe behind, leach other elements. laterite soils. In tropical soils (acidic, warm, high rainfall) feldspars and quartz dissolve leaving iron oxides and hydroxides (when exposed to drying conditions such as clearcutting, Fe(OH3) can dehydrate to form Goethite, FeO-OH which is hard and unworkable)
Sedimentary Processes:
Bog iron deposits: organics and organic acids provide low Eh and solubility of Fe, oxidize when encounters fresh water.
21
Craig et al.
Ironstones: (Phanerozoic, may depend on organic matter) Near shore, usually marine but sometimes fresh water, explain oolite formation.
Fe
2
O
3
+ 2CO
2
= 2FeCO
3
+ 1/2O
2
LeChatelier Principle, what conditions need to change to get siderite? What if increased CO2? Decreased O2?
Banded Iron Formation (the Biggie): (Precambrian, in range 2.6-1.8bya, associated with greater solubility of Fe in ancient oceans compared to today; note stromatolites in rocks ~3.5by). features: age 2.6-1.8bya banding (alternating layers of iron oxides (hematite and magnetite), and silicates (jasper) and carbonates (siderites). facies relationships (indication carbonates may be deeper water, oxides shallower water) occur in belts.narrow, long regions complex with appearance of multiple environments, processes, Fe sources absence of detrital sedimentary materials (are almost exclusively chemical sedimentary deposits) banding association: chert (jasper) also chemical sedimentary, age range indicates significance of atmospheric fO2 (also more acidic, due to CO2). Sources of Fe probably varied, submarine volcanoes, weathering,
MN banded Fe formations: oolitic hematite in MN iron ranges indicates shallow shoals (need water movement) overlain by graded bedding, poorly sorted, turbidity current mudstones (deeper in basin), suggesting Fe restricted to more O2 rich shoreward environments?
22
From a web site-“outline of Minnesota Geology, Winona State University http://www.winona.msus.edu/geology/MRW/text/text34-45.html
, 2004
23
Geos 340, Econ and Env Geol
Lab Activity: Concept Mapping of Ore Formation, Part I: Banded Fe Formation
Fe
3+
: insoluble
Fe
2+
: soluble
2. What is the source of
Fe on land?
1. What is the source of
Fe in the ocean?
10. Why and how do we get FeCO
3
in deeper water?
3. What condition of O
2
(Eh) is necessary for significant Fe
Transport throughout the oceans?
4. When was this condition met? When was it not met?
5. What condition is necessary for significant Fe Transport?
When was this condition met?
6. Explain the condition of O
2
(Eh) necessary for Fe
2
O
3
precipitation in this region.
7. What might cause this condition to occur?
8. Why might it be restricted to nearshore environments?
9. When was this condition met? When was it not?
11. Explain in words why banded Fe formations occur in the time 2.6bya to about 1.8bya, but not before that nor after that.
24
Geos 340, Econ and Env Geol
Lab Activity: Concept Mapping of Ore Formation, Part II: Supergene enrichment of Cu Ore
Fe 3+ : insoluble
Fe
2+
: Soluble
Cu
2+
: Soluble
Cu + : insoluble
1. What is happening to the chemical state of water as it falls through the air?
2. Is the Meteoric ground water high or low in O
2
(Eh)?
3. What is the valence of Fe?
Of Cu?
4. Chemically, what are Fe and Cu doing in this region?
5. What is happening here?
What do the arrows represent? water table
6. Does this region have high or low
O
2
(Eh)?
7. What is the valence of Fe? of Cu?
8. Chemically, what do Fe and Cu do in this region?
9. Put a red circle around the region where Fe will be concentrated.
10. Put a blue circle around the region where Cu will be concentrated.
11. Put a blue rectangle around the area where Cu will be depleted (that is, the region from which Cu will be extracted for later concentration in a much smaller area (volume).
12. Explain in words the process that concentrates Cu.
25
Cu Ores
magmatic: sulfide immiscibility in mafic and ultramafic magmas. hydrothermal: vein deposits examples of changes that can lead to precipitation: changes in T, changes in pH (such as due to encountering limestone), changes in Eh (such as from encountering of organic deposits). porphyry copper
26
Skarn or contact metamorphism : Granite (acidic) intrudes limestone or marble
(CaCO3), pH change results in pption. volcanogenic massive sulfide
Like black smokers. Hydrothermal waters, driven by heat from mid-ocean ridge magmatism, encounters cold, lower pH water, resulting in pption of massive deposits of sulfides not bounded by strata.
Stratiform deposits form is controlled by sediment layers, not patterns of fracturing, or massive areas related to hydrothermal circulation. reduced, organic rich (fO2) and carbonate rich (pH) sediment (e.g. shale) overlying oxidized clastics beds (e.g. red bed). Hydrothermal fluids circulate, preferentially concentrating Cu in the beds with lower fO2, higher pH.
Secondary enrichment process: Supergene enrichment:
see thought map puzzle:
Go through thought map puzzles. where oxides and carbonates of Cu (tenorite CuO, cuprite
Cu2O, azurite, malachite Cu(CO3)2(OH)2, and CU2(CO3(OH)2), and chrysocolla (silicate)? where sulfides (reduced, chalcocite, bornite, native Cu)? talk about Gossan, the iron hat. also Cu oxide-carbonate minerals as prospecting tool. talk about electrowinning, throwing old metal in for autoelectrowinning.
27
Geos 340 Economic and Environmental Geology
Ore Mineral Lab
Don’t just copy information. Rather, copy information AND test each mineral. I will expect you to be able to identify the minerals on the next test.
Minerals to do (place in this order)
Sulfides: Sphalerite (2 specimens), pyrite, chalcocite, chalcopyrite, arsenopyrite, galena, pyrrhotite, molybdenite, bornite, stibnite
Sulfates: gypsum (2 varieties, alabaster and selenite)
Oxides: magnetite, hematite (2 varieties, specular and oolitic), ilmenite
Carbonates:
Chlorides: halite
Silicates:
28
Identity:
Chemical composition
Color
Streak
Hardness diagnostic features
Association (other minerals it commonly occurs with)
Occurrence (Type of ore deposited often associated with)
Identity:
Chemical composition
Color
Streak
Hardness diagnostic features
Association (other minerals it commonly occurs with)
Occurrence (Type of ore deposited often associated with)
29
chalcocite: Cu2S, 2.5-3, orth, hex, shiny lead grey to black tarnish, may be sooty, sectile (can be cut into thin shavings with a knife), an important ore mineral, bornite, chalcopyrite, pyrite, supergene enriched zones of porphyry copper.
Pyrrhotite: Fe1-xS, 4, mon, hex, magnetic, brownish bronze, black streak, ommision solid solution, more
Fe, less magnetic, basic igneous rocks, norites, also in contact metamorphic, vein, pegmatites. pyrite: FeS2, 6-6.5, iso, brass-yellow, greenish black streak, harder than chalcopyrite and paler color, differs from marcasite by deeper color and crystal form. most widespread sulfide, both high and low
T, alters to limonite, magmatic, hydrothermal, contact, sedimentary as both primary and secondary mineral, chalcopyrite, sphalerite, galena. galena: PbS, 2.5, iso, perfect cubic cleavage, lead gray, lead grey streak, high specific gravity, softness, veins associated with sphalerite, pyrite, marcasite, chalcopyrite, calcite,dolomite, quartz, barite, fluourite, maybe be associated with silver minerals when in veins. may contain silver itself. chalcopyrite: CuFeS2, 3.5-4, tet, brittle, yellow, greenish black streak, brass yellow often tarnishes bronze or iridescent, most common Cu mineral, hydrothermal vein, replacement, both low and high temperature deposits, primary ore in porphyry copper, Sudbury, Ducktown, also in Miss Valley
Type. molybdenite: MoS2, 4.6-4.7, hex, lead grey, grayish black streak, platy, denser than graphite and has a bluer tone (compared to graphites brownish) and greener streak on glazed porcelain, granite pegmatites and aplites, high-T vein with cassiterite, fluorite, scheelite, contact metamorphic with lime silicates chalcopyrite, scheelite. sphalerite: ZnS, 3.5-4, Iso, 6 directions, resinous luster, yellow or reddish brown streak, H2S odor, galena, marcasite, chalcopyrite, calcite, dolomite lead-zinc district of Mississippi Valley Type deposits, hydrothermal veins, contact metamorphic. bornite: Cu5FeS4, 3, tet, iso, brownish-bronze when fresh but purple-blue tarnish is characteristic, grayish black streak, chalcocite, chalcopyrite, covellite, pyrrhotite, pyrite. not an important ore mineral, veins, disseminated in basic rocks, metamorphic deposits, replacement deposits, pegmatites. stibnite: Sb2S3, 2, orth, perfect cleavage plane with striations, bladed habit, crystals often curved, lead grey to black color and streak, low T hydrothermal veins, galena, cinnabar, sphalerite, barite, gold. chromite: FeCr2O4, 5.5, iso, submetallic luster, ironblack to brownish black, streak dark brown, peridotites, ultrabasic rocks, magmatic. is the only ore of Cr. hematite: Fe2O3, 5.5-6.6, rho, red streak, red to black, platy metallic variety called specularite, most abundant and important ore of Fe, volcanic, metamorphic, chemical sedimentary ores. name from
Greek for blood. magnetite: Fe3O4, 6, iso, strongly magnetic, black, magmatic segregation, metamorphic, sedimentary pC banded Fe formations. an important Fe ore. ilmenite: FeTiO3, 5.5-6, rho, slightly magnetic, iron black, streak black to brownish red, solid solution with hematite above 950C. igneous, gabbros, anorthosites, magmatic segregation, or in pegmatites and veins. magnetite, rutile, zircon, monazite. gypsum: CaSO2*2H2O, 2, mon, three unequal cleavage plane, transparent to translucent, colorless, white, gray or shades of yellow, red, brown from impurities, sedimentary, halite, dolomite, calcite, pyrite quartz. halite: NaCl, 2.5, iso, cubic cleavage, salty taste, less bitter than sylvite and has yellow flame color, transparent to translucent, colorless or white to shades of yellow, red, blue, purple with impurities, sedimentary, gypsum, sylvite, anhydrite, calcite,
30
Mn Ore:
Important in alloys, scavanges S and O during smelting (FeO +Mn = MnO + Fe: which way will reaction go?), colorant (purple) or decolerant in glasses (reducing Fe).
Soluble as Mn2+, less soluble as Mn3+, fairly insoluble as Mn4+ (MnO2, pyrolusite). Mn also soluble in more acidic solutions, and ores also occur in divalent state (pyrolucite:
BaMn9O18*2H2O, and Rhodochrosite, MnCO3, or rhodonite, MnSiO3)
Ores usually chemical sedimentary in nature. May be primary or by leaching. Mn nodules possible future source, but is not shortage of Mn (but none in US presently profitable).
Al Ore: refractories, light weight metal, activated bauxite as an absorbent.
Corundum, ruby, sapphire.
Abundant in earth (after O and Si). But, binds very strongly to O, poorly soluble = difficult to process. leaching in tropical/subtropical where abundant rainfall (leaching), near neutral pH where Al least soluble. Highly soluble materials like Na, K, Ca leach first. Then Mg and others. Al converted mainly to kaolinite (often from feldspars: see phase diagrams and reactions, compare phase diagram to le Chatelier principle). Eventually, Si in kaolinite can leach out, leaving Al oxides and hydroxides (gibbsite=Al(OH)
3
, boehmite=AlO*OH). Where conditions are slightly more acidic, Fe may also leach (narrow zone), or, more likely, if rocks are initially low in Fe. This produces more pure Al ore. low relief = slow erosion compared to rate of chemical leaching common pisolitic texture, consequence of insitu process of phase transformation.
People notes note climate zones, effects of variable acidity and intensity of leaching
From Water, Land,
31
From Easterbrook, 1993
Formation of kaolinite from K-spar, also production of gibbsite (bauxite) as H2SiO4 drops as
SiO2 is leached (Kaolinite ↔ Gibbsite + Silicic Acid).
Orthoclase + carbonic acid + water ↔ kaolinite, + potassium ions + carbonate ions, + silicic acid
4KAlSi
3
O
8
+ 4H
2
CO
3
+ 18H
2
O = Al
4
Si
4
O
10
(OH)
8
+ 4K + + 4HCO
3
- + 8H
4
SiO
4
+
Electrolysis, add NaF to decrease electrical resistance and viscosity (cryolite=Na3AlF6). basic reaction:
Al
2
O
3
= 2Al + 3O cathodic reaction: 2Al
3+
+ 6e- = 2Al
0 anodic reaction : 3O
2-
= 1.5O
2
+ 6e-
32
Mg Ore: used in refractory, alloys, many varied
Compatible, mafic and ultramafic
Often related to sea water, maagnesite (Mg carbonate), or directly from sea water or brines.
Drive off CO2 by heating (also used with limestone to produce CaO for cement, 2 nd
major producer of greenhouse gases after fossil fuels).
Si Ore: glass, to scavange oxygen in Fe smelting (slag), piezoelectric properties in crystals for timing, reduced metal in silicon chips, photovoltaic cells, other wide ranging uses. quartz arenites, quartzites, quartz veins, pegmatites.
Ni Ore: highly compatible, mafic ultramafic, chalcophile, so primary ores are often magmatic: immiscible separation of Ni sulfide melt (often associated with Pt). It is also siderophile, going with reduced Fe.
Also sometimes get residual leaching deposits (laterite), where e.g. peridotite is weathered to chlorite (Fe, Mg, chlorine mica) and serpentine (hydrated silicate).
U Ores:
Formation of U “roll type” deposits in sandstone and conglomerate, including the chemistry and geological setting. (importance of organics, oxidized ‘yellow’ zone, movement of roll with time)
UO
2
2+
is soluble (6+ state). UO
2
is insoluble (4+ state)
Understand U (and Au) placer deposits (pC!): dense reduced U minerals, (gold also dense so is an association). accumulates in streams where winnowing and sorting concentrate denser minerals that are hydraulic equivalents (explain briefly). Explain why are typically pC (oxidized
U dissolves)
Magmatic (Usually in pegmatites, alkalic igneous, and carbonatites due to incompatibility of U and preference for alkali rich-Si poor melts).
Hydrothermal deposits (usually associated with granite because of U incompatibility)
Show video Isotopes, Part I, stable isotopes.
33
Economic and Environmental Geology
Geothermometry, Cation exchange, and Stable Isotope Exercises
Geothermometry:
Inferring the temperature and pressure under which a particular ore formed is of interest in understanding the process of ore formation and predicting where we might find other ores of that same type.
Because the reactions that deposit ore minerals (or form metamorphic minerals in the second puzzle below) are functions of T and P (as well as activities of all the chemical components in the system, including oxygen [Eh or fO
2
], hydrogen ions [pH], and all the elements of the periodic table), the minerals that form in a particular ore reveal the temperature, pressure, and compositional conditions of that ore.
The set of minerals that form in an ore, or the set of minerals in a metamorphic rock, is called an assemblage . This assemblage can be used to infer T and P.
Puzzle One : Consider the following ore mineral assemblages. Put them in order of increasing temperature of formation (based on information from past lectures): assemblage one: galena, sphalerite, chalcopyrite, barite, pyrite assemblage two: cinnabar, stibnite, galena, Au, quartz, marcasite, barite assemblage three: cassiterite, scheelite, wolframite. tourmaline, topaz, molybdenite note: cinnabar is a Hg-sulfide, cassiterite is a Sn sulfide, scheelite and wolframite are tungsten oxides, marcasite is a copper sulfide. lowest T = middle T = highest T =
34
Puzzle two : Consider the diagram showing the stability conditions for minerals in a quartzbearing non-calcareous rock subjected to metamorphism. This is a composite phase diagram , with each line representing a reaction studied in the laboratory to learn under what conditions of
T and P that reaction occurs. For example, reaction 1 is the following: Analcite + Quartz
Albite + H
2
O [NaAlSi
2
O
6
٠H
2
O + SiO
2
NaAlSi
3
O
8
+ H2O]. Reactions 9 involve the polymorphs of Al
2
SiO
3
, Kyanite
Andalucite, Kyanite
Silliminite, and Andalucite
Silliminite. The key reactions that you need to solve the puzzle are given. To simplify the puzzle for yourself, you might want to highlight these reactions on the diagram.
1) A metamorphic rock contains both Kaolinite and quartz. What can you say about the maximum temperature the rock was subjected to? (remember, this phase diagram is only valid for rocks that contain quartz, so quartz exists everywhere on the diagram except for reactions where it is specifically omitted)
2) Identify the conditions of T and depth (P) that formed a metamorphic rock containing the assemblage: Silliminite, muscovite, staurolite, quartz
3) Identify the conditions of T and depth (P) that formed a metamorphic rock containing the assemblage: Andalucite, Hornblende, Chlorite, Muscovite, and Quartz.
35
Diagram from Hyndman, 1972, pg 313.
Key reactions: unnumbered reaction) Actinolite
Hornblende (not balanced)
4b) Kaolinite + quartz
pyrophyllite + H2O
6) Chlorite + Muscovite + Quartz
Cordierite + Biotite
7a) Staurolite + Quartz
Almandine + Silliminite + H2O
9) either Kyanite
Andalucite, or Andalucite
Silliminite, or Kyanite
Silliminite
36
Cation Exchange, Solubility, and Precipitation :
Cation exchange refers to the process by which cations of different elements ‘switch places’ between (for example) water and sediment, or hydrothermal water and vein wall rock. The Le
Chatelier principle applies to these reactions, just as it applies to other reactions we have examined. We have talked substantially about solubility and precipitation. These reactions depend on pH, Eh, T, and P, and other compositional variables of the overall system.
Puzzle 1 : Consider the following reaction:
Ca
2+*
+ Mg
2+’
Ca
2+’
+ Mg
2+* where ‘ indicates an ion dissolved in water, and * indicates an ion adsorbed onto a sediment particle, such as a clay mineral.
Discuss what you expect to happen where fresh river water, laden with sediment, flows into the sea. Both Mg
2+
and Ca
2+
concentrations are higher in seawater than in fresh water, but the ratio
Mg 2+ /Ca 2+ is higher in seawater than in fresh water.
Puzzle 2 : Consider a river contaminated with Cd 2+ flowing into the ocean. Cadmium is a strong poison, especially when it is dissolved in water and accessible to organism in the ecosystem. In general, Cd oxide and Cd sulfide are very insoluble in water. Thus, we might expect most of the
Cd in the river to be associated with the sediment load and not dissolved in the water. However,
CdCl
2
is much more soluble in water.
Consider the reaction:
CdO
*
+ 2NaCl
’
Na
2
O
*
+ CdCl
2
’
What might happen to the Cd associated with sediment as the contaminated river flows into the sea? Explain.
Puzzle 3 : Consider two rivers. One is flowing from a mining district with abundant sulfide minerals exposed by the mining operation. The presence of sulfide, when oxidized in the presence of water, produces sulfuric acid (1/2FeS
2
+ 1.5O
2
+ H
2
O
H
2
SO
4
+ 1/2Fe). Iron,
Copper, Lead, and many other metals are more soluble under acidic conditions than under basic conditions, thus this river contains a lot of dissolved metals. Because these metals are in solution, the water is clear.
37
The second river is flowing from an area dominated by limestone. The limestone buffers the acidity of the water at a high pH (low acidity). This is the larger of the two rivers. Its water is also clear.
Downstream, the two rivers join (the point where two rivers join is called the confluence ).
The water at the confluence turns dark and murky. Sediment of various colors, including orange and green, is seen on the bottom and along the shores in the region.
Explain what has happened. (I understand that Karl has a stop that is similar to this situation on his field trip to Utah).
Puzzle 4: A natural spring brings groundwater out into a small lake. The sediment in the aquifer from which the groundwater originates contains a lot of organic detritus. Although the groundwater is considered safe to drink, and there are many wells in the area, at the spot where the spring flows into the lake there is an orangish deposit forming that looks really bad, as though the spring water were polluted. Explain what is happening.
38
Stable Isotopes : Isotopes of a given element have the same number of protons (and thus the same chemical properties) but have different numbers of neutrons (giving them different masses). For example, two isotopes of oxygen, 16 O and 18 O, both have 8 protons. However, the first has 8 neutrons and the second has 10 neutrons. The different masses cause them to behave slightly differently with changes in temperature. Also, living things can fractionate between isotopes. In general, the fractionation affect from temperature or living things is greater for lighter elements than heavy elements because the relative difference in mass is greater (Oxygen
18 differs from Oxygen 16 by 12.5%, whereas Uranium 235 differs from Uranium 238 by only
1.3%). Thus, the isotopes of lighter elements, like O and S, can tell us about temperature. We often use isotopes of heavier radioactive elements to tell us about age, but that is another story.
Puzzle 1 : Past Climatic changes on Earth are inferred in part on the basis of changes in oxygen isotopes in ocean sediments through time. The following questions lead you through a concept map of how this works.
A particular water molecule (H
2
O) can contain either 16 O or 18 O. Which do you suppose is more likely to evaporate? (this step is the key to the rest of the puzzle, make sure your reasoning is correct before proceeding further)
We can consider a partition coefficient for
16
O between vapor and liquid,
D
16
O vapor/
16
O liquid. Will this ratio be less than one, equal to one, or greater than one?
Chart the route by which water gets into glacial ice. Where did that water come from and by what route?
Will glacial ice have a higher or lower concentration of
16
O (as a fraction of total oxygen) than the oceans?
Now, consider the two situations where there is a lot of glacial ice on earth (during the ice ages) and where there is only a little glacial ice (like today). In which case will there be a higher concentration of
16
O in the oceans (as a fraction of total oxygen)?
So, will the oceans have a higher concentration of
16
O when the climate is cold or warm?
39
Oxygen in ocean water finds its way into sediment and fossils on the sea floor (for example:
CO
3
2-
+ Ca
2+
= CaCO
3
), and there is a balance of oxygen isotopes between water and carbon dioxide and between carbon dioxide and calcium carbonate (e.g. C 16
2H
2
16
O). Will the fraction of
O
2
+ 2H
2
18 O C 18 O
2
+
16
O in ocean sediment and fossils be higher or lower during periods of cold climate?
Puzzle 2 : Isotopes can also be used as a geothermometer, to infer the temperature at which (for example) ore deposits formed. Below is an example of sulfur isotope fractionation between sulfide and sulfate. Two isotopes of sulfur are
32
S and
34
S. Before using this method as a geothermometer in a real situation, you would need to also account for the effects of other variables such as pH, and to consider the fractionation involved in the precipitation of the sulfate ions as particular minerals. The problem below is simplified so that you can get to the concept without getting lost in the complexity.
H
2
34
S +
32
SO
4
2-
↔ H
2
32
S +
34
SO
4
2where H
2
S is present as a gas, and SO
4
2-
is present in aqueous solution (this reaction occurs in ore-forming situations and was important in the formation of Carlsbad Caverns).
The equilibrium constant for this reaction is given below. An equilibrium constant is a quantitative way of expressing the Le Chatelier principle.
K = ([H
2
32
S][
34
SO
4
2-
])/([ H
2
34
S][
32
SO
4
2-
]) brackets indicate concentrations.
The equilibrium constant (K) for this reaction is a measure of the degree to which the two isotopes of S are fractionated between sulfide and sulfate. For example, if K=1, then there is no fractionation. If K = 1.1, then there is about a 10% fractionation with
34
S going more into the sulfate and
32
S into the sulfide. If K = 0.9, then there is about a 10% fractionation with
34
S going more into the sulfide and 32 S into the sulfate.
The value of K is a function of temperature, as shown in the figure below (from Stanton,
1972, Ore Petrology, an undergraduate text, pg 177).
Suppose that you find from measurements in a particular situation that there was about a
2% fractionation of these sulfur isotopes between sulfate and sulfide with the
34
S preferring the sulfate.
What was the temperature at which this fractionation occurred?
40
Surface Hydrology:
Flow of water:
Laminar: water in thin sheets that flow smoothly over and past each other
Turbulent: seemingly-random chaotic flow, eddies
In general, flow becomes more turbulent as velocity and depth increase.
Also affected by density and viscosity
Reynolds number = R =
dv/
; rho = density, mu = viscosity, d = depth, v = velocity. rule of thumb: if <500 then get laminar flow, if greater than 1200 then get turbulent flow.
Go through exercises of what would case increase or decrease in turbulent flow.
More laminar causes less energy loss. More turbulence causes greater erosion. Increasing sediment load increases viscosity and dampens turbulence.
Discharge = Q = wdv ; w=width, d=depth, v=average velocity. (more generally, cross-sectional area times average velocity)
Notice that discharge is in units of volume per unit time. Draw a rectangular box showing the meaning of the equation, a volume with dimensions w, d, and distance traveled in unit time.
How do we estimate velocities of flood events, or in paleostreams that no longer carry as much water?
(how big of boulders could current move? How big was the channel formed by the former river?)
41
v= 59 d
1/2
; d=boulder diameter, v=bed velocity (which is approximately 0.833 of avg velocity)
Manning formula (an empirical expression) v=1.49/n R
2/3
S
1/2
; v=velocity in fps, n = coefficient of roughness, R=cross-sectional area of channel divided by its wetted perimeter, s=slope.
Think through how velocity will change as change shape of channel, as change slope, as change roughness. How will turbulence of channel change as increase slope? How will it change as go from a channel with shape (1x9; or 4x25) to a channel with same cross-sectional area but with shape (3x3,
or 10x10)
Drainage Basins: could further subdivide into e.g. wild rice (MN) vs Buffalo R. or wild Rice (ND) vs Sheyenne, Maple Rivers.
The amount of water carried by a river is related to the size of the basin, amount of rainfall, rate of runoff (which is a function of the condition of the soil as well as the rate of precipitation or snow melt), and amount of water stored. precipitation = runoff + interception + storage interception = evaporation, transpiration, infiltration. Infiltration is the movement of water into unsaturated soil and sediment, and this water may find its way into streams eventually, but with a lag time, or this water may be used by plants or it may evaporate.
42
storage = snowpack, groundwater, undrained ponds and lakes that supply groundwater. This water, too, may eventually get into rivers. runoff depends on slope, permeability of the soil, vegetation, degree of soil saturation, etc. recurrence interval = the average time for floods of a particular discharge size OR LARGER to occur. up to 1973 from about 1873, or 100 years. In that time there were 7 floods >= 10000cfs, recur every 14 years, which is the value shown (10
1.15
), but they weren’t evenly distributed, occurring in 1882, 1897, 1943, 1952, 1965, 1966, 1969.
43
Recurrence interval activity:
Make a recurrence interval graph for Fargo. Predict the recurrence interval for a flood at 40.7 feet crest. Predict the discharge of a 500 year flood. Data go through 2009.
Top Floods: Red River at Fargo ND
Fargo
Year
0 2009
1
2
1997
1969
3 1897
4
2001
1882
4.5 2006
5 1989
6 1978
7 1979
8 1952
9 1943
Flow*
29400
28800
25,300
25,000
20630
20,000
19800
18,900
17,500
17,300
16,300
16,000
9.5 2007
10 1975
11 1965
12 1994
13 1995
14 1966
15 1993
16 1996
16.5 2005
17 1962
18 1984
19 1947
19.5 2008
13400
13,200
11,400
11,200
11,000
10,700
10,100
9,940
9730
9,580
9,550
9,300
9190
19.75 2007
20 1986
8770
8,640
*All flows are in cubic feet per second
Additional floods between 4000 and 8640cfs (based on daily average flow, generally somewhat less than max flow:
1905 (4250), 1907 (4420), 1910 (4700), 1916 (7440), 1916 (7720), 1917 (5200), 1920 (6120), 1922 (5200), 1929 (4400), 1943 (4240), 1944
(4120), 1945 (7650), 1946 (5880), 1947 (9200), 1950 (7680), 1050 (6250), 1951 (7990), 1953 (6700), 1953 (5550), 1962 (6400), 1962
(8510), 1962 (9570), 1962 (8160), 1963 (4820), 1967 (4230), 1972 (7080), 1972 (4140), 1974 (4040), 1975 (8380), 1980 (5180), 1982
(5800), 1984 (9450), 1984 (4050), 1985 (4600), 1986 (8600), 1986 (7210), 1986 (4000), 1993 (6180), 1998 (8210), 1998 (8590), 1998
(6110), 1999 (4270), 1999 (4090), 2002 (4210), 2003 (6680), 2004 (5380).
Discharge versus gauge for Fargo (2006) (note, values do not quite correspond to actual floods gage and discharge values, possibly due to variations in flow due to temperature, ice, diking, etc?)
8000 cfs = 24.9ft
9000 cfs = 26.7ft
10000 cfs = 28.54ft
12000cfs = 30.9ft
14000cfs = 32.4ft
16000cfs = 33.64
20000cfs = 35.75
24000cfs=37.61
28000cfs=39.3ft
31600cfs=40.7ft
44
Calculations of water volume for Red River.
In March 2010, there were about 6 inches of water equivalent in the snow pack plus rain in the
Red River basin south of Fargo/Moorhead.
So P = 6.
Taking S = 3.25 calculated for basin conditions from Env. Prot. Agency Publication EPA/600/6-
85/002a.
The runoff in inches = [(P – 0.2·S)
2 /(P+0.8·S)]=_________ inches of water over the drainage basin.
Total runoff volume = [(P – 0.2·S)
2 /(P+0.8·S)]·(size of drainage basin)
Drainage basin south of Fargo is about 6800 square miles
This means that _____________ ft
3 must run through our town (you must convert basin area into square feet).
At an average of 24000cfs (2.1x10
9
ft
3
per day- 37.61 foot river stage), how many days will it take for this amount of water to go through Fargo/Moorhead?
If the water goes through in three weeks time, what will be the average cfs?
This corresponds to what stage level?
45
Water Runoff Exercise
Consider that in the area NE of Hawley, MN, 5 inches of rain falls in 24 hours, with 10 inches of snow still on the ground. The rain falls over 25 square miles in the Buffalo River Drainage basin. Assume that the land there is 50% pasture and 50% cultivated. In both cases, the land is uncontoured (cultivation does not follow contour lines) and the soil is in fair condition (it has not been unduly depleted of organic material etc by hard pasturing or lack of crop rotation). The soil is loam, with moderately low runoff potential. Hills typically have 6% slopes. Note: 1sq mile = 259 hectares. 1 hectare = 100 square meters.
1cm=0.01m.
What is the 24 hour runoff into the Buffalo River?
Assume that all this water must get under the Highway 10 bridge in Hawley in 24 hours (this assumption ignores runoff rate). This bridge is 66 feet wide and 14 feet high. It is concrete.
The friction factor (n) for concrete is 0.03. Assume that the slope of the river is about 1 foot per mile. Assuming that the river at Hawley was already running 5 feet deep, will the rainfall cause the water to run over the road? (hint: calculate the depth of water at the
Hawley bridge: 0 points for the correct answer, 50 points for the correct calculation correctly done)
46
The following procedures are taken from EPA publication EPA/600/6-85/002a, Water Quality
Assessment: A screening procedure for toxic and conventional pollutants in surface and ground water, Revised 1985. These are the procedures approved by the EPA.
47
48
49
50
Ground Water:
Remind of meteoric (rain), connate (trapped sedimentary), juvenile sources (magmatic).
Do demo of porosity, large and small marbles. Maybe also a mixture of the two.
Porosity: volume void space/total volume x 100%
3-30% in clastic, 1-30% in Limestone and dolostone, 2-5% in crystalline igneous, volcanic
(vesicular) up to 87% weathering can produce 30-60%
Primary: space between grains: not affected significantly by grain size, but grain shape affects (think of cubes vs spheres, or plate shaped clay or micas particles vs round sand), packing affects (illustrate), sorting affects (illustrate).
Secondary: fractures, solution cavities, shrinkage cracks in fine-grained sediment
Permeability: how easily liquid moves through sediment or rock
Q=PIA (Darcy’s Law)
Q=discharge
P=permeability coefficient (hydraulic conductivity)
I=hydraulic gradient
A=cross-sectional area of aquifer.
Notice similarity with stream discharge equation. What is equivalent for P and I?
P depends on grain size. Big = Better, less friction or surface tension resistance per unit volume due to lower surface area contact. e.g. clay has high porosity but low permeability.
P also depends on the standard deviation of grain size (sorting).
Vesicular lava can also have high porosity but low permeability. Secondary permeability can develop if e.g. fractures unit bubbles. Fracturing is primary source of permeability in clay sediments since the much higher permeability in fractures (even though fracture cross-sectional area may be low relative to cross-sectional area of entire sediment) carries more water than the low-permeability bulk sediment.
Water Table: boundary between saturated and unsaturated sediment. Is not always a horizontal layer because of surface tension (capillary action), and the dynamics of water being added (rate of rainfall), and how fast the water moves away from ‘high’ areas (depends of permeability).
Movement of water will be from areas of higher water level (higher potentiometric surface or
‘head’) to lower areas. Can have mounding of water under landfills if they are not sealed to rainwater, results in migration away from landfill.
51
Aquifer-aquitard-aquiclude-aquifuge. Refer to layers of rock of sediment. In order of decreasing permeability, decreasing capacity to carry water. impermeable aquifuges, etc confine an aquifer.
52
Econonomic and Environmental Geology: Permeability Lab
Porosity: measure the porosity in closest packed large glass marbles. (hint: measure how much water it takes to fill pore space: remember that porosity = volume pore space/total volume x 100%)
Measure the porosity in closest packed small glass marbles
Permeability:
Objective: Measure the permeability in large glass marbles and small glass marbles.
Considering the equipment you have available (tubes, marbles, bbs, liter jugs, clay, stop watches) measure the effects of grain size on permeability (if you have time, also examine the effect of sorting).
Discussion of experimental approach: (hint: You need to measure discharge (volume per unit time) while keeping the hydraulic gradient, cross-sectional area, and distance water travels constant: remember that permeability is determined from the relationship: discharge = PIA.
You will need to choose appropriate units)
Units for F, I, A, and Discharge:
Measurement methods and controls:
Experimental results
53
Environmental Notes, Natural Waters (notes based on Water Quality Assessment,
EPA/600/6-85/002a, Revised 1985) air
Pollutant water biota sediment
EPA establishes criteria for pollutants in water (e.g. criteria to protect aquatic life
drinking water criteria.), for 24 hour avg (
annual average or maximum spike), freshwater (
marine).
Aldrin/dieldrin0.0019
g/l
Antimony 1600
g/l
Arsenic 40
g/l
Chlordane 0.0043
g/l
DDT
Mercury
Toluene
0.00023
g/l
0.2
g/l
2300
g/l
Think about how much this is.....how many grams in a liter. How much water would 1 gallon of
DDT contaminate to above the accepted level?
Note that pesticides have the lowest levels (they were made to kill things, after all). Other types of pollutants include organic solvents (e.g. toluene) and metals (e.g. Arsenic).
There are other criteria bearing on each pollutant as well. For example, The average concentration over 4 days for Hg cannot exceed 0.012
g/l more than once every three years.
The one hour average should not exceed 2.4
g/l more than once every three years. Mercury in saline water should not exceed 0.2
g/l (24 hour avg).
For some pollutants, the toxicity depends strongly on properties of water and limits are expressed in those terms. For example, the 24 avg of Cd cannot exceed 1.05ln(water hardness) -8.53
g/l.
Where hardness is mg/l CaCO3.
Processes that Act on at Toxicant:
Loading Process = rate at which waste discharge, atmospheric deposition, land runoff introduce toxicants into natural waters
Speciation Process = state of chemical in the water (affects toxicity, volatility, etc). Ph affects how much of an acid-base is in the neutral state (for example H2CO3 = H+ + HCO3-: if water is more acidic, in which direction with reaction go?)
Transport Process =
Solubility, partitioning, solution/dissolution, adsorption of colloids
Advection = movement of water and particles in and out of aqueous habitats, movement between water and water
Volatilization = movement between water and air. (influenced by volatilization)
Sedimentation = movement of suspended sediment or movement of toxicants between sediment and water (influenced by partitioning).
54
Transformation Processes =
Biodegradation = organisms metabolize pollutants, altering their toxicity
Photolysis = light induced chemical reactions alter a pollutants toxicity
Hydrolysis = reactions with water alter a pollutants toxicity
Reduction/Oxidation = variations in valence state alter a pollutants toxicity
The first three of these apply mainly to organic toxicants (metal toxicants cannot be broken down into harmless forms). The 4rth applies to both organics (mostly caused by microbe activity, so is also bio-related), and metals (e.g. Cr is most toxic in the hexavalent state).
Bioaccumulation Processes =
Bioconcentration = passive uptake of pollutant results in concentration in organisms (e.g. fish take in pollutant through their gills)
Biomagnification = consumption of contaminated food results in increasing levels of concentration as one goes up the food chain.
Relative influence of Transport and Transformation Processes on selected pollutants: sorption
DDT X
Dieldrin X
PCB X volatilization
X
X
X biodegradation
NO
NO
X (Only tranform. process acting on this)
Tetrachloro
ethane NO X X
DDT can be adsorbed, volatilized, hydrolyzed and will bioaccumulate, but is not biodegraded or made harmless by photolysis.
Both Transport and Transformation processes can remove a pollutant from water. The RATE at which the toxicant is removed is important. Often, rates of removal are related to the concentration of the toxicant (1rst order reaction), the square of the concentration (2 nd order reaction), and so on. rate = k
1
·C
T
, C
T
= total concentration, k
1
= rate constant (dependent on T, P, pH, Eh, etc).
More than one process may act to remove a toxicant (e.g. photolysis make break down the toxicant and it also partitions into bottom sediment). In a simplified way (and not completely correct, especially if the reactions are not all first order), the cumulative rate can be predicted from an expression such as k
1
=
k for each process
Details of Transport Processes: factors affecting sorption: chemical : solubility = f(T, X) (X includes pH, Eh, salt concentrations, hardness etc)
55
partitioning = f(T, X, and amount of minerals or colloids present) environmental: amount and type of suspended sediment organic content of suspended sediment size of sediment particles (increasing surface area/mass increases interaction)
Solubility: organic pollutants are often non-polar, giving them very limited solubility
(NaCl dissolves well, toluene does not). Solubility depends on water T and X.
Toluene solubility is 507mg/l (at 25C), NaCl solubility is 360g/l; both in water.
If the amount of a toxicant exceeds the amount that can be soluble, an immiscible phase develops (for example, common for a large toluene or crude oil spill in water).
Partitioning: reflects the tendency of a species to attach to solid or become part of the solid crystal lattice. Often expressed as a ratio of concentration in solid/concentration in liquid. It depends on the type of sediment, T and X.
Volatilization: depends on the intrinsic tendency of the species to become a vapor or to partition into a vapor phase, also on T, X. the RATE at which it becomes a vapor also depends on the species, T, and X.
As the toxicant approaches its solubility limit, its chemical behavior approaches the behavior of the pure toxicant.
Details of Transformation Processes:
Biodegradation (can be used to clean up organic toxicants):
Metabolism: the pollutant serves as a food source (energy)
Cometabolism: the pollutant is transformed but the biota gain no energy from the transformation (meaning that there MUST be an alternative source of C, or food, available; often biodegradation-assisted clean-ups are implemented by providing food for the cute little bacterias)
Degradation depends on T, X, pH, Eh, nutrients, the state of the toxicant (dissolved or adsorbed, etc) and, for cometabolism, a food source. In general, higher temperatures result in faster and more complete degradation. However, enzymes that catalyze the reactions make this more complex.
In aquatic environments, other nutrients like P or N make become the limiting factor in biodegradation rates.
Only anaerobic bacteria can work under very low Eh conditions, and biodegradation rates are usually lower. Thus, aerating water or sediment can sharply increase biodegradation rates.
Photolysis:
Often reactions are complex. Involve a photon exiting the compound to a higher energy state from which it can decay in more than one way. Reactions depend on the molecular absorption of light and solar radiation (the wavelength of light available may or may not match the wavelength
56
that is absorbed by the species), and on light attenuation in water (turbidity will obviously affect this).
In fact, DDT IS changed by photolysis, to DDE, but this compound is still toxic and has an even longer half-life in the environment than does DDT.
Abiotic Hydrolysis.
Like photolysis, may or may not produce non-toxic substance. Hydrolysis reactions are often first order. Reaction rates depend on T and pH.
Often rate constants can be expressed in the form k
H
= k
N
+ k
A
[H
+
] + k
B
[OH
-
]
Metal Toxicants:
Unlike organic toxicants, these exist naturally in water and sediments. They cannot in general be degraded by biota, sunlight, or hydrolysis, although valence state may affect both their toxicity and mobility. Metals of most concern in natural waters include Arsenic, Cadmium, Cr, Cu, Pb,
Hg, Ni, Ag, and Zn.
In assessing human influence on concentrations of these metals, it is necessary to consider background concentration (the concentration that exists regionally in the water and not simply near a point source) and historical concentrations (that background concentration that existed prior to human activity).
Consider Pb: 4-day avg Pb permitted by EPA for protection of freshwater aquatic life depends on the water hardness (like Cd). for 100mg/l of CaCO2 = 3.2
g/l.
Pb present in soils in New Jersey = 96.3
g/g (for illustration, convert the water allowance to
g/g).
Average concentration of Pb in the Earth’s crust = 15 g/g.
Values measured in the Souris, Red, Rainy, drainage basins in 1975 = from below detection limits to 6
g/l.
Because of the long use of Pb as a lubricant in gasoline, Pb is still highly concentrated in soils near major streets and highways, including in many suburban yards where children play (and eat the dirt). It is still present in many houses where Pb paint was used in the past.
To meet EPA standards in water, Pb can be diluted, or adsorbed or partitioned into sediment (and the sediment can settle out of the water). In sediment, the Pb is not as biologically accessible.
57
Conceptual examples of toxicant attenuation in surface waters .
High Flow Rates in Stream, example from North Fork of Holston
River, VA, 1978. A chloralkali plant that closed in 1972.
Settling ponds are the source of ongoing Hg contamination concentration falls in areas of low flow rates where sediment settles out. Simple dilution (from tributary waters) does not account for the change in concentration. Is the partitioning coefficient for the pollutant >1, =1, or <1?
1/3 of Hg is dissolved. Lake sediments were 0.47-2.4
g/g Hg.
Low Flow Rates in Stream
What is happening to cause the progressive decrease in toxicant concentration? Why does it not match the dilution model? What is the significance of the low flow rate?
58
Consider the observed attenuation of a toxicant in the upper stream shown is as illustrated in Case 1 or Case 2.
Explain the likely cause of each variation.
Case 1: Toxicant is not significantly volatile. The toxicant partitions into sediment which settles in the lake.
Additional attenuation occurs due to dilution from the tributary stream.
Case 2: The toxicant does not partition into the sediment (D<<1).
Attenuation occurs more rapidly as water temperature increases, suggesting either volatile loss (higher
T increase volatility) or T-related degradation of the pollutant.
Additional attenuation occurs due to dilution by the tributary stream.
59
Lab Puzzle: Given the scenario shown, predict the variation in concentration for each of pollutant X and Y.
60
Thought Exercise (do in discussions groups if possible):
Consider that pollutant X is dumped in a stream at point A.
Do a series of thought puzzles related to the figure above in which you choose how the pollutant behaves and model the resultant behavior. Construct concentration versus distance graphs for each scenario. Consider the variables:
Transformation: pollutant is unaffected by transformation pollutant is affected by hydrolysis that is T-dependent
Pollutant is affected by photolysis, dependent on water depth
Transport: volatility (T dependent) partitioning dilution amount of sediment
61
Environmental Puzzle Discussion
(imagine presenting for a client)
Examine the water-table depth contour map from 1993.
Examine the graphs showing changes in VOC concentrations with time on the western boundary of the landfill (wells farther to the west and north have shown no contamination as of 2010).
Consider: use of Cells 1 and 2 was discontinued in the 1990s due to contaminants leading into groundwater from the unlined cells. They were then excavated and lined, and trees were planted along the N and W borders to enhance VOC loss through transpiration.
Consider: water upwells to the west of the landfill, sometimes artesian!
Hypothesis: There is an impermeable clay layer between two aquifers below the landfill. This layer pinches out to the west under the wetland area. The upper wells sample above this clay layer and the lower wells sample below it.
Hypothesis: VOCs from the unlined landfill did not immediately penetrate through the clay layer. Permeation of the clay layer takes time.
Explanation: Explain to a client what is going on. Be prepared for questions. Talk about direction of ground water flow, reason for wetlands and artesian springs, the reason the graphical data shows what it shows (including influence of unlined landfill, influence of excavation and relining, influence of tree plantings, influence of impermeable clay barrier, and influence of groundwater flow), and the cause of the "hill" in groundwater table in Area 1 in 1993.
62
63
All wells are on the immediate west edge of the Clay County landfill. Wells W-11 and W16 are just west of Old Area 1, W9 and W17 are west of old area 2 (both unlined landfill cells that have since been excavated and lined). W-11 and W-9 are shallower wells (above a presumed clay layers between aquifers) and W-16 and W-17 are deeper wells.
Scale goes from Jan 1991 on the left to Jan 2011 on the right.
64