Ligation Mediated Suppresion PCR
advertisement

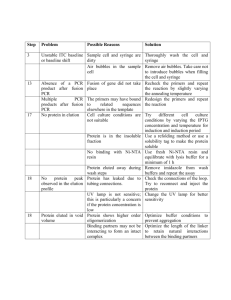
Ligation Mediated Suppression PCR (adapted from McKinney et al., 1995, Siebert, 1995, Strauss et al., 2001 and Alonso et al., 2003) PURPOSE: To analyze unknown flanking genomic sequences adjacent to a T-DNA left border 1.) Isolation of DNA -collect 2-3 young leaves in an eppendorf tube -add 100 µL extraction buffer and add proteinase K, grind tissue using a blue pestel (no large pieces of leaf should be left). -add another 100 µL of extraction buffer, vortex, and incubate in 37º for 30 min. -add 200 µL of saturated phenol and vortex. -spin at max speed in centrifuge for 2 min. -collect upper phase to new eppendorf tube. -add 200 µL of (24:1) chloroform:isoamyl alcohol, vortex, centrifuge at max speed for 2 min. -collect upper phase into a new eppendorf tube. -add 18 µL of 3M sodium acetate and add 400 µL of 100% EtOH, mix by inverting and incubate for 10 min @ 4º. -spin in centrifuge at max speed for 10 min. -pour supernatant off and wash with 500 µL of 70% EtOH -spin in centrifuge at max speed for 5 min. -pour supernatant off and wash again with 500 µL of 70% EtOH -spin in centrifuge at max speed for 5 min. -pour off supernatant -carefully pipette off excess EtOH -let pellet dry for 45 min in the hood. -resuspend DNA in 100 µL of TE -store in -20º 2.) Digestion -mix together: 50 µL gDNA (from above) 10 µL 10x Buffer 2 1 µL Hind III 39 µL dH2O TOTAL volume 100 µL -digest overnight at 37ºC 3.) Digestion Clean Up -heat inactivate at 65ºC for 20 min. -add 100 µL of chloroform -mix by inverting tubes -spin in centrifuge at max speed for 5 min. -collect upper phase into a new eppendorf tube with 200 µL of isopropanol -mix by inverting tubes -incubate at room temperature for 10 min -spin at max speed for 10 min -remove supernatant -wash with 100 µL of 70% EtOH -spin at max speed for 5 min -remove supernatant -dry in hood for 45 min. -resuspend in 20 µL of dH2O 4.) Constructing adapters for ligation *adapters for ligation to Hind III ends are made by annealing oligos ADAPS-E1(5’aattcacctgcccgg/3AmMc7/-3’) w/ a 3’ amino terminal end and ADAPL-E1(5’ctaatacgactcactatagggctcgagcggccgcccgggcaggtg-3’). Oligos may be purchased from IDT @ www.idtdna.com -dilute ADAPS and ADAPL to 100uM -combine in equal amts of ADAPS and ADAPL (i.e. add 10 µL of ADAPS add 10 µL of ADAPL) -vortex briefly -place tube in 500 mL of boiling H2O for 2 min -remove heat and let bath cool for 1 hr. (this is to ensure correct nucleotide pairing) -store adapter at -20ºC 5.) Construction of Adapter Library (ligate adapter to digestion) -mix together: 10 µL cleaned gDNA digestion 1 µL Adapter (100uM) 2 µL T4 ligase (NEB product) 2 µL 10 x T4 ligase buffer (NEB buffer) 5 µL dH2O TOTAL volume 20 µL -vortex and incubate at 16º overnight in thermocycler -heat inactivate @ 65º for 20 min -add 180 µL of TE (this is your adapter library store @ -20º) 6.) Primers for 1º and 2º PCR *Primary products are generated from amplifying primers AP1 (5’-ggatcctaatacgactcactataggc3’) and PgwLat52LB-WP1 (5’-ctatgttactagatcgaccgg-3’). *Secondary products are generated by diluting primary products by 50 fold and amplifying with primers AP2 (5’-tatagggctcgagcggccg-3’) and PgwLat52LB-WP2 (5’-caattcggcgttaattcagtac-3’). -Primers come from IDT and must be diluted to 10 µM concentration before using. 7.) Primary PCR -mix together: 0.125 µL Ex Taq (Takara) 2.5 µL 10 x Ex Taq Buffer 2.0 µL dNTP Mix 1.0 µL AP1 1.0 µL WP1 17.375 µL dH20 1.0 µL Adapter Library TOTAL volume 25 µL *Run on LMS_PCR2 (conditions recommended by Takara) 1.) incubate @ 94ºC for 2 min 2.) incubate @ 94ºC for 30 sec 3.) incubate @ 55ºC for 30 sec 4.) incubate @ 72ºC for 1 min 5.) recycle to step 2 for 29 more times 6.) incubate @ 65ºC for 10 min 7.) hold @ 4ºC forever 8.) Secondary PCR *Dilute primary PCR: -98 µL of dH20 -2 µL of primary PCR *Rxn is setup exactly like primary PCR. 9.) Run on 1% Agarose Gel 5g Agarose Low 500mL 1 x TAE 50uL EtBr *Always run a ladder to verify our bands are between 200 and 2000bp. Should only sequence the lines that give a clear band. 10.)Sequencing *Need to clean up PCR rxn using Qiagen or Eppendorf kits. Then setup the rxn to be sequenced. - mix together: 2 µL cleaned secondary PCR 1 µL secondary primer (WP2) 17 µL dH20 TOTAL volume 20 µL *Sequencing is done through UNC Lineberger Comprehensive Cancer Center (located on campus), and normal turn around time can range from 1-7days. To access sequences go to http://152.19.68.152/gafsite/Main.asp (listed as UNC-CH Genome Analysis under favorites). Run sequences through Signal website http://signal.salk.edu/cgi-bin/tdnaexpress. Unmapped lines can either be digested with a different enzyme (EcoRI),can try sequencing using PgwLat52RB primers, or try TAIL PCR. *If you use EcoRI you must use the EcoRI adapter and then proceed as normal Special Notes: After secondary (nested) PCR, only lines with clear bands are sequenced. Bands are typically between 200 and 2000bp. Lines resulting in a smear or no bands will not provide good sequence. Not all lines with distinct bands will provide good sequence either (some bands are likely artifacts). In our hands, LB mapping of EcoRI digests has up to 75% success rate. The remaining lines can be mapped by a combination of RB-mapping (with appropriate RB primers), digesting with a different enzyme (i.e. HindIII) (with appropriate Adapter modifications), or TAIL PCR. The WP1 and WP2 primers described above are specific to pBI121 and vectors with pBI121 left border regions. Similar primers can be designed to sequence from right borders. The nested primer (WP2) should be ~50 bp inside the T-DNA region to accommodate the possibility of border truncation. Multiple inserts complicate the results. It is possible that each independent insert could result in a distinct nested PCR band. Bands can be excised and sequenced. It is possible to get sequence from both bands, however, by directly sequencing the nested PCR products. Alonso JM, Stepanova AN, Leisse TJ, Kim CJ, Chen H, Shinn P, Stevenson DK, Zimmerman J, Barajas P, Cheuk R, Gadrinab C, Heller C, Jeske A, Koesema E, Meyers CC, Parker H, Prednis L, Ansari Y, Choy N, Deen H, Geralt M, Hazari N, Hom E, Karnes M, Mulholland C, Ndubaku R, Schmidt I, Guzman P, Aguilar-Henonin L, Schmid M, Weigel D, Carter DE, Marchand T, Risseeuw E, Brogden D, Zeko A, Crosby WL, Berry CC, Ecker JR (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301: 653-657 Siebert PD, Chenchik A, Kellogg DE, Lukyanov KA, Lukyanov SA (1995) An improved PCR method for walking in uncloned genomic DNA. Nucleic Acids Res 23: 1087-1088