Defining the Destination - Learning and Technology
advertisement

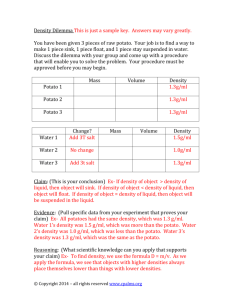
How Much Energy is in Food? How does the type and amount of food I eat affect the energy I get? Chemical reactions result in energy changes. Ryan L. Hackl Spiritwood High School Foods include chemical bonds. The foods I eat will affect the energy I need/use. 1. Can we calculate how much energy we eat in our foods? 2. How do food companies determine the amount of energy in our foods? Technology and Learning Living Sky School Division Fall 2010 Chemistry 30 (Energy Changes) DEFINING THE DESTINATION: UbD Stage One – What do we want them to learn? Outcomes: 1. Students will associate calorimeter work with energy content in foods. 2. Students will compare/contrast energy contents of various meals. Understandings: 1. Chemical reactions result in energy changes. 2. Foods are made of chemical bonds. 3. The foods I eat will affect the energy I need/use. Essential Question: How does the type and amount of food I eat affect the energy I get? Knowledge: Preknowledge: o o o Determining enthalpy of substances and using ‘kilojoules’ to measure this (Q = mcΔT) Differences between temperature and heat Using the ‘Percent Error’ formula Do: o o Students will perform the calorimetry lab Students will create a poster to show the healthy meal for an athlete Knowledge: o o o Students will perform calculations using Q=mcΔT Students will interpret nutritional information from fast food websites Students will create a food plan for an athlete EVIDENCE OF UNDERSTANDING UbD Stage Two – How will we determine what they know? Assessment: Performance Task o Perform the calorimetry lab to determine the heat content in potato chips o Locate nutritional information from fast food websites o Compare and contrast different sandwiches/foods from fast food companies o Compare traditional meals with fast food meals Other Evidence: o Summary question for peer interaction (using Q=mcΔT) LEARNING PLAN UbD Stage Three – How will we teach? Example Formats: One Three Activate o why are marathon runners advised to eat a large plate of pasta before running a race? o why do hummingbirds eat so much sugaryfood? o how much more food energy would you get if you had a high fat vs high protein vs high carbohydrate diet? o Is ‘Subway’ food more healthy than ‘McDonalds’? Where o Science Lab: calorimetry lab o Computer Lab (or laptops): locate nutritional information and create a poster / blog Acquire o Perform the calorimetry lab to find the heat content of chips o Use ‘Percent Error’ to compare nutritional information on packaging with experimental results o Use a laptop to locate nutritional information from fast food restaurants (McDonalds, Burger King, Subway, Wendy’s, etc) Explore/Enable/Equip o Collect and interpret nutritional information o Research skills o Compare energy content levels for various foods (in meal preparation) Apply o Use Microsoft Publisher (or another desktop publishing program) to create a poster to share results o (optional): Blog your results and/or present to class Exhibit/Evaluate o Poster using Microsoft Publisher and/or blog responses and/or present to class o Plan a meal for an athlete o Compare a typical meal for an athlete with a more sedentary lifestyle Two Lesson Focus o Skill development Assessment o Compare energy content from various types of potato chips Learning Experiences o Skill development (chemical) through using science lab equipment o Technology development using temperature probe o Research skills development using technology to locate/interpret nutritional information Hook o Raise an ‘activate’ question o Watch video showing endurance runner o Show bags of potato chips (used for calorimetry experiment) Rethink/Reflect o Share results with class (or blog post) and collect responses Calorimetry Lab “The Energy in Foods” Objective: To determine the energy content in different potato chips. Introduction: Why are marathon runners often asked to eat a plate of pasta before a major ‘meet’? The answer is that pasta is a good source of energy (or fuel) for the body. All foods are made of chemical bonds, and when we eat food, our bodies break down the bonds in the food to give us energy. The more complex the food (like potato chips), the more energy there is for our body to use. But how much energy is there in a potato chip? We can answer that question by using a calorimeter similar to the following to burn potato chips. The big idea? The amount of heat we lose by burning the potato chips is the same as the amount of heat we gain (and can measure) by heating the water. In formula form, this means: Qlost by food = Qgained by water Materials: Graduated cylinder Cork with wire attached Calorimeter Lighter Water Thermometer (or temperature probe) Forceps Safety glasses Potato chips (at least 2 different brands) Digital scale Petri dish or weigh boat Procedure: 1. Choose two brands of potato chips, and hypothesize which brand has more heat content (“energy”). Record your hypothesis in the observations table. 2. Using the petri dish (or weigh boat), obtain about 10.0 grams of the potato chip, and record the data in your table. Record this as “minitial” in your observation table. 3. Using the graduated cylinder, measure out 100 mL of water, and pour it into the calorimeter. Since 1 mL = 1 g for water, record “100 g” for “mwater” in the observations table. 4. Measure the initial temperature of the water. Record this as “T1” in your observations table. 5. Place the potato chips on the cork-and-wire apparatus, so that the potato chips are very near to the bottom of the calorimeter. (Refer to the picture in the introduction section). 6. Light the potato chips, and allow the chips to completely burn. Record the final mass of the ‘chip remnants’ as “mfinal” 7. Now, stir the water in the calorimeter and measure the highest temperature reading. Record this as “T2” in your observation table. 8. Repeat steps #2-7 for your second brand of potato chip. 9. Once completed, please clean up your work station, and put the cooled chip remnants in the garbage. Observations: Hypothesis: Which brand of potato chip has more energy? Why? Table 1. Data recording sheet 1. Mass of Potato Chips at start, in grams (minitial) 2. Mass of ‘chip remnants’, in grams (mfinal) 3. Mass of chips burned, in grams mchips = mfinal - minitial 4. Mass of water used, in grams *see procedure #3 5. Initial water temperature, in °C (T1) 6. Final water temperature, in °C (T2) 7. Change in water temperature, in °C ∆T = T2 – T1 Brand #1: Brand #2: ________________ ________________ Table 2. Calculations Food #1: Food #2: ________________ ________________ 4.18 J/ gC 4.18 J/ g C 1. Total mass of water used (from Table 1, line 4) 2. Change in water temperature (from Table 1, line 7) 3. Specific heat of water 4. Energy absorbed/lost by the water (J) [use Q = mcT] 5. Energy absorbed/lost (kJ) [convert J into kJ by ÷ 1000] 6. Mass of chips used (from Table 1, line 3) 7. Calculate the kJ released per gram of food used [divide energy in kJ (part 5) by mass of chips (part 6)] 8. What the potato chip package says for this food Discussion Questions: 1. Calculate the Percent Error between your calculated energy value (Table 2, #7) and the actual value from the potato package (Table 2, #8). 2. What in the potato chips made them burn so well? 3. Backpackers in the woods sometimes take a bag of chips as a ‘fire starter’. Based on your experience with this lab, why is this a good choice? 4. In terms of energy, support/refute the following statement: “Eating too many potato chips may make you less healthy, because you are taking in too much energy.” Choose either #5 or #6 or #7 and prepare a well-researched blog post or poster to highlight your learnings: 5. Using the ‘Nutritional Information’ on the websites of various fast food restaurants, determine the total energy eaten in a typical ‘full meal’ (drink, meal, dessert) for any four restaurants and compare/contrast. 6. Contrast the energy value of a ‘traditional homecooked meal’ with that from a ‘fast-food restaurant’. Include a sample meal from each, and the long-term outcomes associated with each. 7. Compare/contrast the energy and nutritional needs of an athlete with a regular person. Account for the higher needs.