Design of PCR primers
advertisement

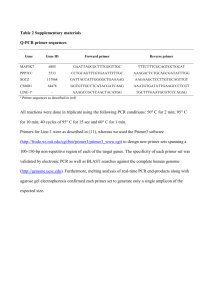
Designing primers for PCR reactions Upstream primer (sense) Always write sequence from 5’ to 3’ Start with a few bases (2-3) of random sequence (e.g. TAT)- This allows spacing before the restriction site and hence better cleavage. Incorporate your restriction site (e.g. BamHI “GGATCC”), then a “Linker” sequence before the start ATG, e.g. “GCC”, which allows more efficient translation according to Kozak’s rule. Finally, you write the sense sequence starting from the ATG and followed by the subsequent sequence. How many bases do you need? It depends, e.g. on the length of the PCR product and the primer’s GC content. For the LT cDNA (2.1 kb) I used 21bp of complementary sequence. In the case of LT, a suitable upstream primer is: --BamHI-- Kozak-start 5’ TAT GGA TCC GCC ATG GAT AAA GTT TTA AAC AGA Downstream primer (anti-sense) This is essentially the same as above but with a twist. Write the sequence out for the sense strand first. It should be complementary with the end of your sequence immediately before the stop. At the end you either put a stop codon (if you want that to be the end) or you make sure that it’s in frame with the other sequence that you want to fuse it with (e.g. a tag, ER etc.). After that follows the restriction site and random sequence for improved cutting efficiency. Finally, you convert to the anti-sense strand (anti-parallel on DNA Strider) and write it out 5’ to 3’. E.g. if the sense sequence is 5’ GATTCATAGGACTA 3’, the anti-sense strand is 5’ TAGTCCTATGAATC 3’. Finally, don’t forget to check the primer on IDTDNA’s website! (Oligo analyzer) http://www.idtdna.com/analyzer/Applications/OligoAnalyzer/ Make sure there’s not a great potential for hairpins or primer self-dimers.