Animal Monitoring Sheet for Research
advertisement

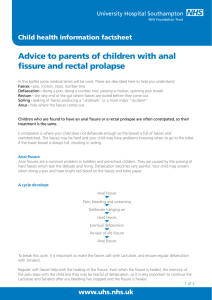
ANIMAL MONITORING SHEET PROJECT NAME:_______________________________________________________________________ Version Number: XXXXX Date: DD/MM/YYYY AEC Project No Investigator Cage/Animal ID Number Species, Strain, Sex, Age etc Amend this sheet to reflect the specific animal model in your experiment i.e. if over 15% weight loss is expected in the first 2 days of the experiment (followed by recovery), justify in your application and request approval for an increased percentage. Any additional observations tailored to the monitoring requirements for each experiment are added i.e anaemia, prolapsed anus, pregnancy, tumour size and necrosis. Remove irrelevant clinical signs, justify and inform committee under Section 3 Monitoring. Each animal must be examined and observed for abnormalities at each time point (weekly or daily, twice daily, out of hours etc as appropriate) record time intervals between same day observations. Observations must be recorded in the table (refer to scoring table) Normal clinical signs are recorded as “0” Severity of each abnormality must be scored, e.g., Breathing 3, Coat 2 , Eyes 0 Comments concerning abnormalities are recorded in the comments section of the table Surgical site appearance must be checked daily until completely healed. DATES and TIMES CLINICAL OBSERVATION (0 and above) UNDISTURBED Activity Alert/Sleeping Breathing Coat Drinking Eating Movement/gait ON HANDLING Alert Body condition Body weight (g) Percentage weight loss% Breathing Dehydration Eyes Faeces Nose Vocalisation Surgical site appearance TOTAL SCORE COMMENTS Signature of investigator/manager V2 Feb 2015 Date 1 CLINICAL SIGNS SEVERITY SCORE – Category I (1-3) SIGNS 0 1 2 isolated, abnormal posture dull or depressed coat rough 3 Activity normal huddled/inactive, isloated OR overactive little response to handling unkempt; superficial wounds, hair thinning, slight pilo-erection loose, soiled perineum Alertness/Sleeping normal Coat normal Faeces normal faeces moist Movement/ gait normal walking on tiptoe OR reluctance to move Nose normal slight OR abnormal gait wetness Vocalisation normal squeaks when palpated Surgical site appearance normal (no swelling or redness) mild redness, mild swelling of wound struggles and squeaks loudly in response to handling of a body part & signs of aggression presence of discharge and presence of moderate swelling, slight wound dehiscence. discharge moribund OR fitting unconscious bleeding or infected wounds, or severe hairloss, severe piloerection or self mutilation Presence of diarrhoea for 48 hours OR no faeces for 48 hrs OR presence of blood on faeces staggering OR limb dragging OR paralysis Obstruction of nasal passages or constant purulent discharge persistent and repetitive vocalisation without handling redness and swelling of the wound and/or separation of wound edges. *Euthanasia if resuturing and/or surgical intervention fails Other CLINICAL SIGNS SEVERITY SCORE – Category II (4-6) SIGNS 0 4 Body condition normal thin Body weight normal weight and growth rate reduced growth rate Breathing normal rapid, shallow Dehydration none Drinking normal Eating normal Eyes normal skin less elastic increased OR decreased intake over 24 hrs increased OR decreased intake over 24 hours wetness or dullness 5 6 loss of body fat, failure to grow Chronic weight loss >10%** OR failure to grow loss of muscle mass that enables palpation of the spine acute weight loss>10% * chronic weight loss 15% ** OR failure to grow for juveniles or failure to stabilise body weight laboured, irregular, or gaping mouth breaking, and/or blue skin skin tenting & eyes and abdomen sunken constantly drinking OR not drinking over 24 hours rapid, abdominal breathing skin tenting/ sunken abdomen increased OR decreased intake over 48 hours increased OR decreased intake over 48 hours obese OR not eating over 48 hours (amend for obese strains) discharge Severe eye infection with vision obstructed and signs of pain. Other *, Acute refers to the weight loss occurring within 2 days. **, Chronic refers to the weight loss occurring within 7 days; V2 Feb 2015 2 SPECIAL HUSBANDRY REQUIREMENTS Record specific diets and supplements, therapies, bedding enrichment or particular food supplements or enrichment that is not appropriate i.e sunflower seeds, paper etc EUTHANASIA/HUMANE EXPERIMENTAL ENDPOINT CRITERIA 1) Category I clinical signs: Mandatory euthanasia should be performed if an animal scored 3 for any clinical signs from column 3. Separate from the condition above, the animals will be closely monitored or euthanised based on the total scoring system as given below. Total Score 0-2 3-8 9-12 13 and above Classification/Action Good Health Health to be monitored daily Poor Health – Observe animals twice daily, if no improvement within 48 hours mandatory euthanasia. Seek further advice from the BRF Manager or AWO Mandatory euthanasia 2) Category II clinical signs: Mandatory euthanasia should be performed if an animal scored 6 for any clinical sign from column 3. This includes acute ( >10%) or chronic ( >15%) weight loss unless justified and approved by the Austin AEC. Animals with clinical signs with a single score of 4 from Category II will be closely monitored and symptoms recorded once daily. Animals with a single score of 5 require from Category II require twice daily monitoring and symptoms recorded in the morning and afternoon. Seek advice from the BRF Manager or AWO. If there no improvement twice daily monitoring must continue until animal improves/recovers (or euthanized), then once daily monitoring is required for a minimum of 5 days unless the animal deteriorates. V2 Feb 2015 Separate from the two conditions above, the animals will be closely monitored or euthanised based on the total scoring system as given above. 3