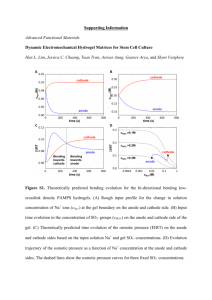
View/Open - Lirias

Culture of Cell-Encapsulating PEG Hydrogels under Mineralizing Conditions
Ç. Demirkaya
1
, Y.C. Chai
2,3
, A.K. Kudva
3,4
, J. Patterson
3,4
1
Department of Bioengineering, Ege University, Turkey; and
2
Skeletal Biology and Engineering Research
Center,
3
Research Division Prometheus, and
4
Department of Metallurgy and Materials Engineering, KU
Leuven, Belgium jennifer.patterson@mtm.kuleuven.be
Introduction: Researchers are investigating methods through which biomaterials can be better integrated with natural bone as well as help to promote bone regeneration. These approaches include the creation of functional polymer scaffolds that mimic the bone extracellular matrix (ECM); direct biomineralization; and stimulate cell adhesion, proliferation, migration, and differentiation. Hydrogels are polymer-based materials with a high water content and thus suitable for use as scaffolds for tissue regeneration. They are capable of encapsulating cells in 3D and supporting nutrient-waste exchange. Hydrogels made from poly(ethylene glycol) (PEG) are one of the most studied hydrogel systems because they offer good biocompatibility, non-immunogenicity, and resistance to protein adsorption. End-functionalized multi-arm PEG precursors can be crosslinked into an enzymatically degradable hydrogel network using di-thiol containing protease substrates in a Michael-type addition reaction 1 . Using the same chemistry, various bioactive peptides, such as Arg-Gly-Asp (RGD), can be incorporated into the PEG hydrogels to enhance the adhesion of cells and promote a biomimetic environment for the encapsulated cells. In this study, we aimed to determine the mineralization potential of cell-encapsulating 3D PEG scaffolds under different culture conditions as well as to examine the effect of the treatment on cell behavior. This work builds upon observations that human periosteum-derived cells (hPDCs), a type of skeletal precursor cell, can respond in vitro to supplementation with calcium (Ca 2+ ) and phosphate (P i
) in the growth medium, leading to increased cell proliferation, upregulation of osteogenic gene expression, and mineralization. This effect has been shown in 2D culture 2 as well as in 3D culture on solid (non-degradable) scaffolds 3 . Here, we focus on degradable scaffolds.
Materials and Methods: hPDCs were encapsulated in degradable PEG hydrogels modified with an RGDcontaining peptide. Scaffolds (with and without cells) were incubated in three different culture conditions.
Scaffolds were incubated either in growth medium or in growth medium containing Ca 2+ and P i
for 21 days. A third set of PEG scaffolds was incubated in medium containing Ca 2+ and dexamethasone for 21 days before being switched to medium containing Ca 2+ and P i
for an additional 21 days. Constructs were analyzed weekly to measure cell proliferation (Presto Blue metabolic activity assay), cell attachment and morphology (staining with phalloidin), cell viability (Live/Dead assay), and cell differentiation (alkaline phosphatase activity). Staining of cryosections with H&E, picrosirius red, Von Kossa, and alizarin red was also performed. NanoCT was used to characterize the distribution and volume of mineral deposited with the scaffold.
Results, Discussion, and Conclusion: Through 42 days, hPDCs encapsulated in the PEG hydrogels and cultured in the two phase procedure (medium with dexamethasone followed by medium with Ca 2+ and P i
) continued to proliferate. Metabolic activity of encapsulated cells cultured in growth medium as well as in growth medium with
Ca 2+ and P i
peaked at 14 days and then decreased. From images of phalloidin-stained constructs, hPDCs in PEG hydrogels cultured in medium containing Ca 2+ and P i
exhibited a more spread morphology than the other conditions. Interestingly, nanoCT analysis showed that incubation of cell-free hydrogels in medium containing Ca 2+ and P i
led to mineral deposition primarily on the surface of the hydrogels whereas cell encapsulation in the hydrogels led to mineral being deposited also inside the scaffolds.
In conclusion, we have identified in vitro culture conditions that support the growth of hPDCs in a 3D PEG hydrogel scaffold and promote construct mineralization.
References: 1. Patterson J, Hubbell JA. Biomaterials . 2010; 31(30):7836-7845.
2. Chai YC et al . Tissue
Engineering: Part A . 2011; 17(7-8):1083-1097.
3. Chai YC et al . Tissue Engineering: Part C . 2012; 18(2):90-103.