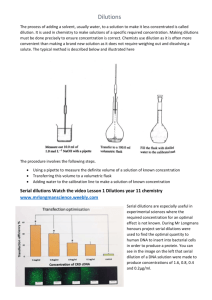
Two-fold serial dilutions
advertisement

Practical immunology 3rd class / Dept.Microbiol./Vet.Med.Coll.2015-2016. Assist.Prof.Dr.Ikram Abbas Aboud Antibodies titration , serum and antigens preservation:Titration:- The measurement of the level of specific antibodies in serum by testing increasing dilutions of the serum for antibody activity. Titer:- The reciprocal of the highest dilution of a serum that gives a reaction in an immunological test. Dilutions: Introduction: Many of the laboratory procedures involve the use of dilutions. It is important to understand the concept of dilutions, since they are a handy tool used throughout all areas of the clinical laboratory. These dilutions have to be considered as they make a quantitative difference in what is going on. Serial Dilutions: A serial dilution is any dilution where the concentration decreases by the same quantity in each successive step. Serial dilutions are multiplicative. What Does This Mean? If a solution has a 1/10 dilution the number represents 1 part of the patient sample added to 9 parts of diluents. So the volumes used would be 10-1= 9. This represents 1 part patient sample added to 9 parts of diluents. Dilutions: If a 1/8 dilution of the stock solution is made followed by a 1/6 dilution what is the final dilution. The final dilution is: 1/8 x 1/6 = 1/48 1 These type of dilutions are trickier and not used very frequently in the clinical lab. Doubling Dilutions: Doubling dilutions” are very popular. This is a series of ½ dilutions. Each successive tube wills ½ the amount of the original concentrated solution. If this is done 6 times this is what you would end up with: Doubling Dilution 6 Times: 1st dilution = 1 /2 2nd dilution = 1 /2 x 1 /2 = 1/4 3rd dilution = 1/4 x 1 /2 = 1/8 4th dilution = 1/8 x 1 /2 = 1/16 5th dilution = 1/16 x 1 /2 - 1/32 6th dilution = 1/32 x 1 /2 = 1/64 This results in a series of dilutions, each a doubling dilution of the previous one Dilution Factor: The dilution factor is the final uses the formula volume/aliquot volume. Example: What is the dilution factor if you add 0.1 mL aliquot of a specimen to 9.9 mL of diluents? -The final volume is equal to the aliquot volume PLUS the diluent volume: 0.1 mL + 9.9 mL = 10 mL -The dilution factor is equal to the final volume divided by the aliquot volume: 10 mL/0.1 mL = 1:100 dilution Practice: Problem: What is the dilution factor when 0.2 mL is added to 3.8 mL diluent? Set Up: dilution factor = final volume/aliquot volume 0.2 +3.8 = 4.0 total volume 4.0/0.2 = 1:20 dilution 2 Notes : Remember that serial dilutions are always made by taking a set quantity of the initial dilution and adding it successively to tubes with the same volume. So each successive dilution would be multiplied by the dilution factor. So in the above problem all successive tubes would have 3.8 mLs of diluents. You would then transfer 0.2 of the initial diluted sample into the next tube, mix transfer 0.2, mix and so on. If you had 4 tubes what would be the final dilution of tube 4? Calculate DF: Or if you simply wanted to know the dilution of the final tube you could just multiply them together: 1/20 x 1/20 x 1/20 x 1/20 = 1:160,000 What if we added the indicator system to the test system? You must create additional rows, if you are showing your work, to take into consideration the dilution effect that will have on the dilution. When an indicator is added it is CRITICAL to remove an aliquot from the last tube. Two-fold serial dilutions:A two-fold dilution reduces the concentration of a solution by a factor of two that is reduces the original concentration by one half. A series of two-fold dilutions is described as two-fold serial dilutions. In this manual, two-fold serial dilutions are carried out in small volumes in microwell plates. They are used in both the haemagglutination and haemagglutination inhibition tests to establish titers of the test samples. A two-fold dilution:Step 1. Use the micropipette to dispense 25 mL of PBS diluent to the first well. Step 2. Use the micropipette to transfer 25 mL of the test solution to the first well. Step 3. Use the micropipette to mix by drawing up the liquid and expelling it again. Carry out this action twice. Step 4. The well now contains 25 mL of the original test solution diluted by one half in a total volume of 50 mL. 3 4 Ten-fold serial dilutions:A ten-fold dilution reduces the concentration of a solution or a suspension of virus by a factor of ten that is to one-tenth the original concentration. A series of ten-fold dilutions is described as tenfold serial dilutions. In this manual, ten-fold serial dilutions are used in titrations of a suspension of Newcastle disease virus to establish the infectivity titer. They are carried out in small sterile test tubes. These tubes are usually made of glass and it is preferable if they have fitted lids to minimize the risk of contamination during the dilution. A ten-fold dilution:Step 1. Step 2. Step 3. Step 4. Use a micropipette to dispense 900 mL of the diluent to glass tube. Use a micropipette to transfer 100 mL of the test solution to the first well. Discard the tip. Mix by shaking by hand or using a vortex mixer. The well now contains 100 mL of the original test solution diluted by one tenth in a total volume of 1000 mL. The technique used to make a single dilution is repeated sequentially using more and more dilute solutions as the "stock" solution. At each step, 1ml of the previous dilution is added to 9ml of distilled water. Each step results in a further 10-fold change in the concentration from the previous concentration. The values shown in the tubes are the amount (in ml) of the stock solution present in each ml of the dilute solution.The dilution of the original stock solution is shown below the tubes. 5 6 ========================================================== Tube 1 2 3 4 Aliquot 0.2 0.2 0.2 0.2 Diluents 3.8 3.8 3.8 3.8 Math. 0.2/4 1/20x1/20 1/400x1/20 1/8000x1/20 Dilutions 1:20 1:400 1:8000 1:160,000 ===== ======= ======= ========== ========== 7 Tube 1 2 3 4 Dilutions 1:20 1:400 1:8000 1:160,000 RBCs 0.2 0.2 0.2 0.2 Volume 3.8 3.8 3.8 3.8 DF 1/4 -------- ------ ------- Math. 4x20 4x400 4x8000 4x160,000 Dilutions 1:80 1:800 1:32,000 1:640,000 8 There are two methods for preservation of serum and antigen:A-Physical methods:1- Short period at 4 ˚C (not exceed than 48 hours). 2- Moderate period at - 40 to - 70˚C for 1 month to 1 year. 3- Long period by lophylization. B- Chemical methods:1-Merthiolate in concentration 1/10000. 2-Phenol in concentration 0.5%. 4- Sodium azid (1% of 10% stock -- -- 1/10000). 9 10 11