Concentration and Dilutions - drbperiodcsem2
advertisement

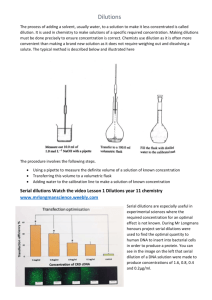
Jackielyn Jabson Friday, March 16, 2014 Solutions Unit Dilution Lesson Date: Friday, May 16, 2014 IN CLASS PowerPoint on Dilution Sheet Name: Concentration & Dilutions Sheet Number: H3 HOMEWORK ↘ Complete “More Dilution Practice” on Concentration & Dilutions sheet (H3) ↘ Textbook questions on p. 405 #2 – 7 (Dilutions Review) and p. 411 #1-10 (more Concentration of Solutions Review) Answers to More Dilution Practice Questions: http://sch3uking.wikispaces.com/file/view/Dilution%20problemsANswers%20fuill.pdf/389045782/Dilution%20 problemsANswers%20fuill.pdf Concentration and Dilutions Lesson Summary: What is Dilution? Dilution is the process of reducing the concentration of a solution by adding more solvent. The number of moles of solute remains the same because solute is neither being taken out or being added. More solvent (water – a universal solvent) is only being added to the solution. The result of the dilution would be that there is less solute (in moles) per volume of the solution. More moles of solute per volume Less moles of solute per volume = = Concentrated solution Diluted solution How to Prepare a Dilution? Dilutions involve transferring accurate volumes of a concentrated solution into another container. In order to prepare dilutions, a volumetric pipette and volumetric flask are used because there is less surface area for air due to their small diameter and thus will be more accurate; compared to a graduated cylinder which has a larger diameter. 1 Jackielyn Jabson Friday, March 16, 2014 Dilution Calculations: During dilution, the amount of solute (in moles) does not change. The concentration changes because the volume of the solution increases. Remains constant (amount of solute) Decreases (because volume Increases (because water is added) increases) 𝒄 = 𝒏 𝒗 The final concentration of the dilute solution can be determined by using a mathematical equation: Dilution Equation Concentrated Solution Diluted Solution C1V1 = C2V2 Initial Concentration NOTE: Final Initial Concentration Volume Final Volume ↘ Since moles remain constant, when you have two solutions, a concentrated solution and diluted solution, the two equations above can be made equivalent to one another ↘ Concentration and volume have an inverse relationship (as one increases, the other decreases) ↘ Initial & Final Volumes and Concentrations MUST all have the same units EXAMPLE A stock solution of 1.00M of NaCl is available. How many millilitres are needed to make a 100.0mL of 0.750M? Step 1: Determine the information given Given C1 = 1.00M V1 =? C2 = 0.750M V2 = 100mL Step 2: Write down the equation and plug in the information given C1V1 = C2V2 1.00M x V1 = 0.750 x 100mL Step 3: Rearrange equation to solve for the unknown (in this case the unknown is the initial volume) 𝐕𝟏 = 0.750𝑀 𝑥 100𝑚𝐿 1.00𝑀 Step 4: Solve equation 𝐕𝟏 = 75𝑚𝐿 ∴ 75mL are needed. NOTE: ↘ The given volume was NOT needed to be converted from millilitres into litres because the mol/litre (M) is canceled out. Be aware that your answer is in millilitres. ↘ BEWARE if the question asked for the answer to be litres, the 75mL would have to be converted to litres (by dividing 75mL by 1000L) 2 Jackielyn Jabson Friday, March 16, 2014 Resources on Dilution Textbook Chapter: 8.7 Preparing Dilutions p. 403 – p. 405 Further explanation regarding dilutions and more practice questions with answers provided: http://dl.clackamas.edu/ch105-04/dilution.htm More Practice Questions with Answers: http://mrdeakin.pbworks.com/f/Dilutions+Practice+Problems+%26+Answers.pdf Easy to Understand YouTube Tutorial on Dilution: https://www.youtube.com/watch?v=v6dnEp58mVk 3