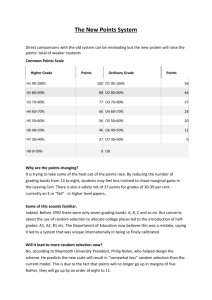
DRAFT PROPOSAL FOR AMENDMENTS
advertisement

DRAFT PROPOSAL FOR AMENDMENTS DRAFT PROPOSAL FOR AMENDMENTS UPDATED ON: 08.01.2013 The IPC has received inputs from its stakeholders on IP 2010 which require upgradation/change. The draft amendments (PROPOSED) are put on the website (www.ipc.gov.in) for comments of the users. It is expected that if no comments are received within 21 days, please note- the draft amendments will be taken for finalization. General Chapters 2.4.22.Optical Rotation and Specific Optical Rotation. Page 143 Para 5, line 5 Change from: per ml. to: per 100 ml. 2.4.26. Solubility Dapsone. Page154 Change to: Freely soluble in acetone; soluble in dilute mineral acids; sparingly soluble in ethanol (95 per cent) and very slightly soluble in water. Famotidine. Page 156 Change to: Freely soluble in dimethylformamide and in glacial acetic acid; slightly soluble in methanol; very slightly soluble in water; practically insoluble in acetone, in ethanol (95 per cent), in chloroform, in ether and in ethyl acetate. Fluocinolone Acetonide. Page 157 Change to: Soluble in acetone and in ethanol; slightly soluble in chloroform and practically insoluble in water and in light petroleum. Isosorbide Mononitrate, Diluted. Page 159 Delete the requirement. 2.4.2. Atomic Absorption Spectrometry. Page 109 Insert at the end. Calcium Solution AAS Dissolve 1.001 g of calcium carbonate, previously dried to constant weight at 105°, in 25 ml of 1M hydrochloric acid, boil to remove carbon dioxide and add sufficient water to produce 1000 ml. Calcium Solution FP contains 400 μg of Ca in 1 ml. Iron Solution AAS Dilute sufficient quantity of iron standard solution with 0.2 M nitric acid to contain 4 μg of Fe in 1 ml. Lithium Solution AAS Dissolve 1.064 g of lithium carbonate in 5 ml of hydrochloric acid and add sufficient water to produce 100 ml. Lithium Solution FP contains 2 mg of Li in 1 ml. Potassium Solution AAS. Dissolve 1.440 g of potassium chloride, previously dried to constant weight at 130°, in sufficient freshly distilled water to produce 1000 ml. Potassium Solution FP contains 600 μg of K in 1 ml. 1 DRAFT PROPOSAL FOR AMENDMENTS Silver Solution AAS Dissolve 0.790 g of silver nitrate in sufficient water to produce 1000 ml. Further dilute 1 volume of the resulting solution to 100 volumes with water. Silver solution AAS contains 5 μg of Ag in 1 ml. Sodium Solution AAS Dissolve 0.5084 g of sodium chloride, previously dried to constant weight at 130°, in sufficiently freshly distilled water to produce 1000 ml. Sodium Solution FP contains 200 μg of Na in 1 ml. Strontium Solution AAS Dissolve 1.685 g of strontium carbonate in 10 ml of a 50 per cent v/v solution of hydrochloric acid and add sufficient water to produce 1000 ml. Strontium Solution AAS contains 1 mg of Sr in 1 ml. 2.4.4. Flame Photometry. Page 110 Insert at the end. Calcium Solution FP Dissolve 1.001 g of calcium carbonate, previously dried to constant weight at 105°, in 25 ml of 1M hydrochloric acid, boil to remove carbon dioxide and add sufficient water to produce 1000 ml. Calcium Solution FP contains 400 μg of Ca in 1 ml. Lithium Solution FP Dissolve 1.064 g of lithium carbonate in 5 ml of hydrochloric acid and add sufficient water to produce 100 ml. Lithium Solution FP contains 2 mg of Li in 1 ml. Potassium Solution FP Dissolve 1.440 g of potassium chloride, previously dried to constant weight at 130°, in sufficient freshly distilled water to produce 1000 ml. Potassium Solution FP contains 600 μg of K in 1 ml. Sodium Solution FP Dissolve 0.5084 g of sodium chloride, previously dried to constant weight at 130°, in sufficiently freshly distilled water to produce 1000 ml. Sodium Solution FP contains 200 μg of Na in 1 ml. 2.5.2. Dissolution Test. Page 189 Dissolution medium. Insert at the end, For hard or soft gelatin capsules and gelatin-coated tablets that do not conform to the dissolution specification, repeat the test as follows. Where water or a medium with a pH of less than 6.8 is specified as the medium in the individual monograph, the same medium specified may be used with the addition of purified pepsin that results in an activity of 750,000 Units or less per 1000 ml. For media with a pH of 6.8 or greater, pancreatin can be added to produce not more than 1750 Units of protease activity per 1000 ml. 2 DRAFT PROPOSAL FOR AMENDMENTS 4.2. General Reagents. Page 563 Insert before Zinc, Activated Zinc Acetate, 0.25 M. Dissolve 54.9 g of zinc acetate in a mixture 600 ml of water and 150 ml of glacial acetic acid; stir to dissolve the zinc acetate. While stirring, add 150 ml of ammonium hydroxide, cool to room temperature, adjust the pH to 6.4 with ammonium hydroxide and dilute with water to produce 1000.0 ml. Page 566, Ammonia Solution, Dilute: Line 4, Change from: in a cool place. to: and at a temperature not exceeding 30°. Page 566, Ammonia Solution, Strong: Line 7, Change from: in a cool place. to: and at a temperature not exceeding 20°. Page 575, Cinnamaldehyde: Line 4, Change from: in a cool place. to: and at a temperature not exceeding 30°. Page 579, Dimethylaminobenzaldehyde Reagent: Line 6, Change from: room temperature. to: a temperature not exceeding 30°. Page 589, Iodine Bromide: Line 6, Change from: in a cool place. to: and at a temperature not exceeding 30°. Page 590 Iodine Trichloride: Line 5, Change from: in a cool place. to: and at a temperature not exceeding 30°. Page 594 Metaphosphoric- Acetic Acids Solution: Line 4, Change from: in a cool place. to: at a temperature not exceeding 30°. Page 610 Sodium Bisulphite: Line 15, Change from: in a cool place. to: and at a temperature not exceeding 30°. Page 611, Sodium Hypochlorite Solution: Line 13, Change from: in a cool place. to: and at a temperature not exceeding 30°. Page 612, Sodium Hypochlorite Solution (3 per cent Cl): Line 14, Change from: in a cool place. to: and at a temperature not exceeding 30°. 3 DRAFT PROPOSAL FOR AMENDMENTS 4.5. Volumetric Reagents and Solutions. Page 631, Dioctyl Sodium Sulphosuccinate, 0.0005M Change to: Dioctyl Sodium Sulphosuccinate, 0.01M. Dissolve 4.5 g of dioctyl sodium sulphosuccinate in warm water, cool and dilute to 1000 ml with water. Standardise the solution in the following manner. To 25 ml add 25 ml of a solution containing 20 per cent w/v of anhydrous sodium sulphate and 2 per cent w/v of sodium carbonate, 50 ml of chloroform and 1.5 ml of bromophenol blue solution and mix. Titrate with 0.01M tetrabutylammonium iodide until about 1 ml from the end point. Stopper the flask, shake vigorously for 2 minutes and continue the titration, in increments of 0.05 ml, shaking vigorously and allowing the flask to stand for about 10 seconds after each addition. Continue the titration until a blue colour just appears in the chloroform layer. 1 ml of 0.01M tetrabutylammonium iodide is equivalent to 0.004446 g of C20H37NaO7S. Page 635 Insert before Titanium Trichloride, 0.1 M, Tetrabutylammonium Iodide, 0.01M. Dissolve 4 g of tetrabutylammonium iodide in water and dilute to 1000.0 ml with water. Standardise the solution in the following manner. Pipette 25.0 ml of the solution into a flask, add 50.0 ml of 0.01 M silver nitrate solution, add 1.0 ml of 1 M nitric acid and titrate the excess of silver nitrate with 0.01 M ammonium thiocyanate using 1 ml of ferric ammonium sulphate solution as an indicator. 1 ml of 0.01 M silver nitrate is equivalent to 0.003694 g of C16H37IN. 5.2. Biological Indicators. Page 639 6.2. Identification. Line 8 Change from: 30° to 50° to: 30° to 35° 7.2. Weights and Measures: SI Units Insert at the end: Table-6 gives the conversion from part per million and Table-7 gives commonly used concentrations and their conversions. Table-6 Parts per billion (ppb) 1 10 100 1000 5000 10000 Parts per million (ppm) 0.001 0.01 0.1 1 5 10 20 µg per g Fraction Decimal Per cent w/w 0.001 0.01 0.1 1 5 10 20 1/1000000000 1/100000000 1/10000000 1/1000000 5/1000000 1/100000 2/100000 0.000000001 0.00000001 0.0000001 0.000001 0.000005 0.00001 0.00002 0.0000001 per cent 0.000001 per cent 0.00001 per cent 0.0001 per cent 0.0005 per cent 0.001 per cent 0.002 per cent 4 DRAFT PROPOSAL FOR AMENDMENTS 50 100 200 1000 5000 50 100 200 1000 5000 5/100000 1/10000 2/10000 1/1000 5/1000 0.00005 0.0001 0.0002 0.001 0.005 0.005 per cent 0.01 per cent 0.02 per cent 0.1 per cent 0.5 per cent Table-7 µg per ml mg per ml 1 10 20 50 100 200 500 1000 10000 0.001 0.01 0.02 0.05 0.1 0.2 0.5 1 10 g per ml g per 100 ml (per cent w/w) 0.000001 0.00001 0.00002 0.00005 0.0001 0.0002 0.0005 0.001 0.01 0.0001 0.001 0.002 0.005 0.01 0.02 0.05 0.1 1 Capsules. Page 721 Disintegration. Soft Capsules. Line 3 Change from: and add a disc to each tube to: The disc may be omitted if the capsules adhere to the disc or if it is likely to be attacked by the contents of capsules. Enteric Capsules. Change to: Use the apparatus described under disintegration test (2.5.1), using one capsule in each tube. Operate the apparatus for 2 hours without the discs in 0.1 M hydrochloric acid. No capsule shows sign of disintegration or of rupture permitting the escape of the contents. Replace the medium in the vessel with mixed phosphate buffer pH 6.8. When justified and authorized, a buffer solution of pH 6.8 with added pancreas powder (for example, 0.35 g of pancreas powder per 100 ml of buffer solution) may be used. Add a disc to each tube and operate the apparatus for a further 60 minutes. If the capsules fail to comply because of adherence to the discs, the results are invalid. Repeat the test on a further 6 capsules omitting the discs. Monographs Alprazolam. Page 786 Identification. B, line 1 and 3 Change from: water to: methanol Alprazolam Tablets. Page 787 Dissolution. Chromatographic system, line 2 5 DRAFT PROPOSAL FOR AMENDMENTS Change from: octadecylsilane to: octylsilane Amikacin. Page 2874 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Test solution. Dissolve 0.1 g of the substance under examination in water and dilute to 10.0 ml with water. Reference solution. A 0.01 per cent w/v solution of amikacin RS in water. Derivatise the solutions prior to analysis by transferring 0.2 ml of the solution under test to a ground-glass-stoppered vial. Add 2 ml of a 1.0 per cent w/v solution of 2,4,6-trinitrobenzenesulphonic acid. To this solution add 3 ml of pyridine and close the vial tightly. Shake vigorously for 30 seconds and heat in a water-bath at 75° for 45 minutes. Cool in cold water for 2 minutes and add 2 ml of glacial acetic acid. Shake vigorously for 30 seconds. Store the derivatised solutions at 10° prior to and during analysis. Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm) (Such as Spherisorb ODS 2), – column temperature: 30 °, – mobile phase: a mixture of 30 volumes of a 0.27 per cent w/v solution of potassium dihydrogen orthophosphate, adjusted to pH 6.5 with 2.2 per cent w/v solution of potassium hydroxide and 70 volumes of methanol, – flow rate: 1 ml per minute, – spectrophotometer set at 340 nm, – injection volume: 20 µl. The relative retention time with reference to amikacin for 4–O–(3–amino–3–deoxy–α–D–glucopyranosyl)–6–O– (6–amino–6–deoxy– α –D–glucopyranosyl)–1–N–[(2S)–4–amino–2–hydroxybutanoyl]–2–deoxy–L–streptamine RS (amikacin impurity A) is about 1.5. Inject the reference solution. The test is not valid unless the column efficiency is not less than 2000 theoretical plates and the tailing factor is not more than 1.5. Inject the reference solution and the test solution. Run the chromatogram 4 times the retention time of the principal peak. In the chromatogram obtained with the test solution, the area of any peak due to amikacin impurity A is not more than the area of the principal peak in the chromatogram obtained with the reference solution (1.0 per cent); the area of any other secondary peak is not more than 0.5 times the area of the principal peak in the chromatogram obtained with the reference solution (0.5 per cent) and the sum of areas of all the secondary peaks other than amikacin impurity A is not more than 1.5 times the area of the principal peak in the chromatogram obtained with the reference solution (1.5 per cent). Ignore any peak with an area less than 0.1 times the area of the principal peak in the chromatogram obtained with the reference solution (0.1 per cent). Amikacin Sulphate. Page 2874 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Test solution. Dissolve 0.1 g of the substance under examination in water and dilute to 10.0 m with water. Reference solution. Dissolve 7.5 mg of amikacin RS in water and dilute to 100.0 ml with water. Derivatise the solutions prior to analysis by transferring 0.2 ml of the solution under test to a ground-glass-stoppered vial. Add 2 ml of a 1.0 per cent w/v solution of 2,4,6-trinitrobenzenesulphonic acid. To this solution add 3 ml of pyridine and close the vial tightly. Shake vigorously for 30 seconds and heat in a water-bath at 75° for 2 hours. Cool in cold water for 2 minutes and add 2 ml of glacial acetic acid. Shake vigorously for 30 seconds. Store the derivatised solutions at 10° prior to and during analysis. 6 DRAFT PROPOSAL FOR AMENDMENTS Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm) (Such as Spherisorb ODS 2), – column temperature: 30 °, – mobile phase: a mixture of 30 volumes of a 0.27 per cent w/v solution of potassium dihydrogen orthophosphate, adjusted to pH 6.5 with 2.2 per cent w/v solution of potassium hydroxide and 70 volumes of methanol, – flow rate: 1 ml per minute, – spectrophotometer set at 340 nm, – injection volume: 20 µl. The relative retention time with reference to amikacin for 4–O–(3–amino–3–deoxy–α–D–glucopyranosyl)–6–O– (6–amino–6–deoxy– α –D–glucopyranosyl)–1–N–[(2S)–4–amino–2–hydroxybutanoyl]–2–deoxy–L–streptamine RS (amikacin impurity A) is about 1.5. Inject the reference solution. The test is not valid unless the column efficiency is not less than 2000 theoretical plates and the tailing factor is not more than 1.5. Inject the reference solution and the test solution. Run the chromatogram 4 times the retention time of the principal peak. In the chromatogram obtained with the test solution, the area of any peak due to amikacin impurity A is not more than the area of the principal peak in the chromatogram obtained with the reference solution (1.0 per cent); the area of any other secondary peak is not more than 0.5 times the area of the principal peak in the chromatogram obtained with the reference solution (0.5 per cent) and the sum of areas of all the secondary peaks other than amikacin impurity A is not more than 1.5 times the area of the principal peak in the chromatogram obtained with the reference solution (1.5 per cent). Ignore any peak eluting before the principal peak and any peak with an area less than 0.1 times the area of the principal peak in the chromatogram obtained with the reference solution (0.1 per cent). Amikacin Injection. Page 2875 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Test solution. Dilute a volume of the injection containing about 0.1 g of Amikacin to 10.0 ml with water. Reference solution. A 0.013 per cent w/v solution of amikacin sulphate RS in water. Derivatise the solutions prior to analysis by transferring 0.2 ml of the solution under test to a ground-glass-stoppered vial. Add 2 ml of a 1.0 per cent w/v solution of 2,4,6-trinitrobenzenesulphonic acid. To this solution add 3 ml of pyridine and close the vial tightly. Shake vigorously for 30 seconds and heat in a water-bath at 75° for 2 hours. Cool in cold water for 2 minutes and add 2 ml of glacial acetic acid. Shake vigorously for 30 seconds. Store the derivatised solutions at 10° prior to and during analysis. Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm) (Such as Spherisorb ODS 2), – mobile phase: a mixture of 30 volumes of a 0.27 per cent w/v solution of potassium dihydrogen orthophosphate, adjusted to pH 6.5 with 2.2 per cent w/v solution of potassium hydroxide and 70 volumes of methanol, – flow rate: 1 ml per minute, – spectrophotometer set at 340 nm, – injection volume: 20 µl. The relative retention time with reference to amikacin for 4–O–(3–amino–3–deoxy–α–D–glucopyranosyl)–6–O– (6–amino–6–deoxy– α –D–glucopyranosyl)–1–N–[(2S)–4–amino–2–hydroxybutanoyl]–2–deoxy–L–streptamine RS (amikacin impurity A) is about 1.5. 7 DRAFT PROPOSAL FOR AMENDMENTS Inject the reference solution. The test is not valid unless the column efficiency is not less than 2000 theoretical plates and the tailing factor is not more than 1.5. Inject the reference solution and the test solution. Run the chromatogram 4 times the retention time of the principal peak. In the chromatogram obtained with the test solution, the area of any secondary peak is not more than the 1.5 times the area of the principal peak in the chromatogram obtained with the reference solution (1.5 per cent) and the sum of areas of all the secondary peaks is not more than three times the area of the principal peak in the chromatogram obtained with the reference solution (3.0 per cent). Ignore any peak eluting before the principal peak and any peak with an area less than 0.1 times the area of the principal peak in the chromatogram obtained with the reference solution (0.1 per cent). Amiodarone Hydrochloride. Page 802 Insert before Related substances. Impurity H. Determine by thin-layer chromatography (2.4.17), coating the plate with silica gel F254. CAUTION- Prepare the solutions immediately before use and keep protected from bright light. Mobile phase. A mixture of 5 volumes of anhydrous formic acid, 10 volumes of methanol and 85 volumes of dichloromethane. Test solution. Dissolve 0.5 g of the substance under examination in 5.0 ml of the dichloromethane. Reference solution (a). Dissolve 10 mg of (2-chloroethyl)diethylamine hydrochloride (amiodarone hydrochloride impurity H) in 50.0 ml of dichloromethane. Dilute 2.0 ml of this solution to 20.0 ml with dichloromethane. Reference solution (b). Mix 2.0 ml of the test solution and 2.0 ml of reference solution (a). Apply to the plate 50 µl of test solution, reference solution (a) and 100 µl of reference solution (b). Allow the mobile phase to rise 15 cm. Dry the plate in air and spray the plate with potassium iodobismuthate solution and then with dilute hydrogen peroxide solution, examine immediately in day light. Any spot correspond to amiodarone hydrochloride impurity H in the chromatogram obtained with the test solution is not more than the principal spot in the chromatogram obtained with reference solution (a) (0.02 per cent). The test is not valid unless the chromatogram obtained with the reference solution (b) shows clearly visible spot of amiodarone impurity H. Amiodarone Tablets. Page 2877 Related substances. After chromatographic system, para 3, Change to: Inject the reference solution and the test solution. Run the chromatogram twice the retention time of the principal peak. In the chromatogram obtained with the test solution, the area of any peak corresponding to amiodarone impurity A is not more than the 2.5 times the area of the principal peak in the chromatogram obtained with the reference solution (0.5 per cent) and the area of any other secondary peak is not more than the area of the principal peak in the chromatogram obtained with the reference solution (0.2 per cent). The sum of areas of all the secondary peaks is not more than 5 times the area of the principal peak in the chromatogram obtained with the reference solution (1.0 per cent). Ignore any peak with an area less than 0.25 times the area of the principal peak in the chromatogram obtained with the reference solution (0.05 per cent). Atenolol. Page 847 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Test solution. Dissolve 100 mg of the substance under examination in the mobile phase and dilute to 100.0 ml with the mobile phase. Dilute 1.0 ml of the solution to 10.0 ml with the mobile phase. Reference solution. Dilute 1.0 ml of the test solution to 100.0 ml with the mobile phase. Chromatographic system 8 DRAFT PROPOSAL FOR AMENDMENTS – a stainless steel column 30 cm x 3.9 mm, packed with octadecylsilane bonded to porous silica (5 µm), – mobile phase: dissolve 1.1 g of sodium 1-heptanesulfonate and 0.71 g of anhydrous dibasic sodium phosphate in 700 ml of water. Add 2 ml of dibutylamine, and adjust to pH 3.0 with 0.8 M phosphoric acid, add 300 ml of methanol, – flow rate: 0.6 ml per minute, – spectrophotometer set at 226nm, – injection volume: 50 µl. Inject the reference solution. The test is not valid unless the column efficiency is not less than 5000 theoretical plates and the tailing factor is not more than 2.0. Inject the reference solution and the test solution. Run the chromatogram six times the retention time of the principal peak. In the chromatogram obtained with the test solution, the area of any secondary peak is not more than 0.25 times the area of the principal peak in the chromatogram obtained with reference solution (0.25 per cent). The sum of areas of all the secondary peaks is not more than 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (0.5 per cent). Atenolol Tablets. Page 848 Assay. Lines 5 and 6 Change from: sintered glass funnel (Porosil G3) to: fine glass micro-fibre filter paper (Whatman GF/C) Atorvastatin Tablets. Page 850 Insert before Other tests. Uniformity of Content. (For tablets containing 10 mg or less) Complies with the test stated under Tablets. Determine by liquid chromatography (2.4.14) as described in the Assay using the following test solution. Test solution. Transfer one tablet in 50.0-ml volumetric flask, add 3 ml of water and allow to disperse the tablet in water, add 20 ml of methanol and mix with the aid of ultrasound, make up to volume with methanol and filter. Dilute 10.0 ml of the filtrate to 25.0 ml with the solvent mixture. Azathioprine. Page 855 Identification Change to: Determine by infrared absorption spectrophotometry (2.4.6). Compare the spectrum with that obtained with azathioprine RS or with the reference spectrum of azathioprine. Azithromycin. Page 857 Related substances. Reference solution (a). Change to: Reference solution (a). A 0.008 per cent w/v solution of azithromycin RS in the solvent mixture. After chromatographic system, insert. Name Azithromycin impurity L 1 Azithromycin impurity M 2 Azithromycin impurity E 3 Relative retention time 0.29 0.37 0.43 Correction factor 2.3 0.6 9 DRAFT PROPOSAL FOR AMENDMENTS Azithromycin impurity F4 Azithromycin impurity D5 Azithromycin impurity J6 Azithromycin impurity I7 Azithromycin impurity C8 Azithromycin impurity N9 Azithromycin impurity H10 Azithromycin impurity A11 Azithromycin impurity P Azithromycin (Retention time: about 45-50 minutes) Azithromycin impurity O12 Azithromycin impurity G13 Azithromycin impurity B14 0.51 0.54 0.54 0.61 0.73 0.76 0.79 0.83 0.92 1.0 1.23 1.26 1.31 0.3 0.7 0.1 0.2 - 1 azithromycin 3’- N-oxide 3’-(N,N-didemethyl)-3’-N-formylazithromycin 3 aminoazithromycin 4 3’-N-demethyl-3’-N- formylazithromycin 5 14-demethyl-14- (hydroxymethyl)azithromycin 6 13-O-decladinosylazithromycin 7 3’-N-demethylazithromycin 8 3”-O-demethylazithromycin 9 3’-de(dimethylamino)-3’-oxoazithromycin 10 3’-N-[[4-(acetylamino)phenyl]sulfonyl]-3’-N-demethylazithromycin 11 6-demethylazithromycin 12 2-desethyl-2-propylazithromycin 13 3’-N-demethyl-3’-N-[(4-methylphenyl)sulfonyl]azithromycin 14 3-deoxyazithromycin 2 Last para. Insert at the end ;ignore the peaks eluting before azithromycin impurity L and after azithromycin impurity B. Azithromycin Oral Suspension. Page 860 Assay. Solvent mixture. Change from: 40 volumes of acetonitrile and 60 volumes of 3.484 per cent w/v solution of dipotassium hydrogen phosphate with the pH previously adjusted to 6.5 with orthophosphoric acid. to: 40 volumes of acetonitrile and 60 volumes of water. Azithromycin Tablets. Page 861 Related substances. Reference solution (a). Change to: Reference solution (a). A 0.008 per cent w/v solution of azithromycin RS in the solvent mixture. After chromatographic system, insert. Name Azithromycin impurity L 1 Azithromycin impurity M 2 Azithromycin impurity E 3 Relative retention time 0.29 0.37 0.43 Correction factor 2.3 0.6 10 DRAFT PROPOSAL FOR AMENDMENTS Azithromycin impurity F4 Azithromycin impurity D5 Azithromycin impurity J6 Azithromycin impurity I7 Azithromycin impurity C8 Azithromycin impurity N9 Azithromycin impurity H10 Azithromycin impurity A11 Azithromycin impurity P Azithromycin (Retention time: about 45-50 minutes) Azithromycin impurity O12 Azithromycin impurity G13 Azithromycin impurity B14 0.51 0.54 0.54 0.61 0.73 0.76 0.79 0.83 0.92 1.0 1.23 1.26 1.31 0.3 0.7 0.1 0.2 - 1 azithromycin 3’- N-oxide 3’-(N,N-didemethyl)-3’-N-formylazithromycin 3 aminoazithromycin 4 3’-N-demethyl-3’-N- formylazithromycin 5 14-demethyl-14- (hydroxymethyl)azithromycin 6 13-O-decladinosylazithromycin 7 3’-N-demethylazithromycin 8 3”-O-demethylazithromycin 9 3’-de(dimethylamino)-3’-oxoazithromycin 10 3’-N-[[4-(acetylamino)phenyl]sulfonyl]-3’-N-demethylazithromycin 11 6-demethylazithromycin 12 2-desethyl-2-propylazithromycin 13 3’-N-demethyl-3’-N-[(4-methylphenyl)sulfonyl]azithromycin 14 3-deoxyazithromycin 2 Last para. Insert at the end ;ignore the peaks eluting before azithromycin impurity L and after azithromycin impurity B. Water. Delete the requirement. Benzhexol Hydrochloride. Page 884 Related substances. Chromatographic system, mobile phase Change to: a mixture of 200 volumes of water, 0.2 volume of triethylamine, adjusted to pH 4.0 with orthophosphoric acid and 800 volumes of acetonitrile, Calcitriol Capsules. Page 961 Assay. Test solution. Change to: For capsules containing 0.25 µg or less calcitriol use the mixed contents of 10 capsules as test solution. For capsules containing more than 0.25 µg of calcitriol, dilute a quantity of mixed contents of 10 capsules with the mobile phase to obtain a concentration of 1.5 µg of calcitriol per ml. Cefadroxil Oral Suspension. Page 2891 Related substances. Reference solution (c). 11 DRAFT PROPOSAL FOR AMENDMENTS Change to: Reference solution (c). Dissolve 10 mg of 7- amino desacetoxycephalosphorinic acid RS (cefadroxil impurity B RS ) in 10.0 ml of phosphate buffer pH 7.0 and dilute to 100.0 ml with mobile phase A . Dilute 5.0 ml of this solution to 50.0 ml with mobile phase A. Cefotaxime Sodium Injection. Page 1017 Water Change to: Loss on drying (2.4.19). Not more than 3.0 per cent, determined on 1.0 g by drying in an oven at 105º. Cefpodoxime Tablets. Page 1020 Dissolution. Line 13 Change from: cefpodoxime proxetil RS in the same medium to: cefpodoxime proxetil RS prepared by dissolving in minimum quantity of methanol and diluted with the dissolution medium. Chloroquine Phosphate. Page 1061 Assay. Change to: Assay. Determine by liquid chromatography (2.4.14). Test solution. Dissolve 15 mg of the substance under examination in water and dilute to 100.0 ml with water. Reference solution (a). A 0.015 per cent w/v solution of chloroquine phosphate RS in water. Reference solution (b). A solution containing 0.015 per cent w/v of chloroquine phosphate RS and 0.0015 per cent w/v of hydroxychloroquine sulphate RS in water. Chromatographic system – a stainless steel column 10 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm), – mobile phase: a mixture of 78 volumes of buffer solution prepared by dissolving 6.8 g of monobasic potassium phosphate in 1000.0 ml of water, add 1.0 ml of perchloric acid, adjusted to pH 2.5 with orthophosphoric acid and 22 volumes of methanol, – flow rate: 1.2 ml per minute, – spectrophotometer set at 224 nm, – injection volume: 10 µl. The relative retention time with respect to chloroquine phosphate for hydroxychloroquine sulphate is about 0.8. Inject reference solution (b). The test is not valid unless the resolution between the peaks due to chloroquine phosphate and hydroxychloroquine sulphate is not less than 1.5. The column efficiency is not less than 2000 theoretical plates, tailing factor is not more than 1.5 and the relative standard deviation for replicate injections is not more than 2.0 per cent. Inject reference solution (a) and the test solution. Calculate the content of C18H26ClN3 .2H3PO4. Chloroquine Phosphate Tablets. Page 1063 Assay. Change to: Assay. Determine by liquid chromatography (2.4.14). Test solution. Weigh and powder 20 tablets. Disperse a quantity of powder containing about 15 mg of Chloroquine Phosphate in water and dilute to 100.0 ml with water, with the aid of ultrasound for 20 minutes and filter. Reference solution (a). A 0.015 per cent w/v solution of chloroquine phosphate RS in water. 12 DRAFT PROPOSAL FOR AMENDMENTS Reference solution (b). A solution containing 0.015 per cent w/v of chloroquine phosphate RS and 0.0015 per cent w/v of hydroxychloroquine sulphate RS in water. Chromatographic system – a stainless steel column 10 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm), – mobile phase: a mixture of 78 volumes of buffer solution prepared by dissolving 6.8 g of monobasic potassium phosphate in 1000.0 ml of water, add 1.0 ml of perchloric acid and adjusted to pH 2.5 with orthophosphoric acid and 22 volumes of methanol, – flow rate: 1.2 ml per minute, – spectrophotometer set at 224 nm, – injection volume: 10 µl. The relative retention time with respect to chloroquine phosphate for hydroxychloroquine sulphate is about 0.8. Inject reference solution (b). The test is not valid unless the resolution between the peaks due to chloroquine phosphate and hydroxychloroquine sulphate is not less than 1.5. The tailing factor is not more than 1.5 and the relative standard deviation for replicate injections is not more than 2.0 per cent. Inject reference solution (a) and the test solution. Calculate the content of C18H26ClN3 .2H3PO4 in the tablets. Cholecalciferol Tablets. Page 2896 Change Uniformity of content and Assay to: Uniformity of content. Complies with the test stated under Tablets. Determine by liquid chromatography (2.4.14), as described under Assay using the following test solution. Test solution. For tablets containing less than 250 µg, add 2 ml of water to one tablet in an amber-coloured flask and disperse with the aid of ultrasound. Add 3 to 6 ml (As appropriate depending upon strength) of dimethyl sulphoxide, mix, extract with hexane by shaking for 30 minutes to get final concentration same as reference solution (a), centrifuge the hexane layer and use the clear supernatant liquid. For tablets containing more than 250 µg, prepare the solution in the same manner but using 4 ml of water, 12 to 24 ml (As appropriate depending upon strength) of dimethyl sulphoxide and quantity of hexane to obtain a final concentration same as reference solution (a). Assay. Determine by liquid chromatography (2.4.14). NOTE- Carry out the following procedure as rapidly as possible in subdued light and protected from air. Test solution. Weigh and powder 20 tablets. Weigh accurately a quantity of the powder containing about 250 µg of Calciferol, disperse in 4 ml of water. Add 6 to12 ml (As appropriate depending upon strength) of dimethyl sulphoxide, mix, extract with quantity of hexane by shaking for 30 minutes, to get final concentration same as reference solution (a), centrifuge the hexane layer and use the clear supernatant liquid. Reference solution (a). A 0.001 per cent w/v solution of cholecalciferol RS or ergocalciferol RS, as appropriate. Reference solution (b). Dissolve 50 mg of cholecalciferol RS or ergocalciferol RS as appropriate in 10 ml of toluene without heating and dilute with the mobile phase to 100.0 ml. Dilute 5.0 ml of this solution to 50.0 ml with the mobile phase. Reflux 5.0 ml of this solution, under nitrogen, using a water-bath, for 60 minutes to obtain a solution of cholecalciferol, precholecalciferol and trans-cholecalciferol. Cool and dilute the refluxed solution to 50.0 ml with the mobile phase. Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with porous silica or ceramic microparticles (3 to 10 µm) (Such as Nucleosil 50-S 5 µm), – mobile phase: a mixture of 997 volumes of hexane and 3 volumes of 1-pentanol, – flow rate: 2 ml per minute, – spectrophotometer set at 254 nm, – injection volume: 10 or 20 µl. 13 DRAFT PROPOSAL FOR AMENDMENTS Inject a suitable volume of reference solution (b). Adjust the sensitivity so that the height of the peak due to cholecalciferol is more than 50 per cent of the full-scale deflection. Record the chromatograms after five more injections. The approximate relative retention times calculated with reference to cholecalciferol are 0.4 for precholecalciferol and 0.5 for trans-cholecalciferol. The resolution between precholecalciferol and transcholecalciferol should be not less than 1.0; if necessary adjust the proportions of the constituents and flow rate of the mobile phase to obtain the required resolution. Inject reference solution (a). Adjust the sensitivity so that the height of the peak due to cholecalciferol or ergocalciferol is more than 50 per cent of the full-scale deflection. Inject reference solution (a) and the test solution. Calculate the content of cholecalciferol, C27H44O, or ergocalciferol, C28H44O in the tablets. Colchicine Tablets. Page 1131 Uniformity of content. Change to: Uniformity of content. Complies with the test stated under Tablets. Determine by liquid chromatography (2.4.14), as described in the Assay using following modification. Test solution. Disperse one tablet in 40 ml of the solvent mixture, with the aid of ultrasound for 15 minutes and dilute to 50.0 ml with the solvent mixture and filter. Reference solution. Dissolve an accurately weighted quantity of colchicine RS in the solvent mixture and dilute with the solvent mixture to obtain a solution having a known concentration similar to the expected concentration of the test solution. Calculate the content of C22H25NO6 in the tablet. Assay. Change to: Assay. Determine by liquid chromatography (2.4.14). NOTE- Protect the solutions from light and prepare the solutions immediately before use. Solvent mixture. 50 volumes of methanol and 50 volumes of water. Test solution. Weigh and powder 20 tablets. Disperse a quantity of the powder containing about 0.6 mg of Colchicine in 50 ml of the solvent mixture with the aid of ultrasound for 15 minutes, rinsing down the walls of the flask at about 8 minutes and dilute to 100.0 ml with the solvent mixture and filter. Reference solution. A 0.0006 per cent w/v solution of colchicine RS in the solvent mixture. Chromatographic system - a stainless steel column 25 cm x 4.6 mm, packed with octylsilane bonded to porous silica (5 µm), - mobile phase: dilute 45 ml of 0.5 M monobasic potassium phosphate with water to 450 ml. Add about 530 ml of methanol, cool to room temperature and dilute to 1000 ml with methanol, adjusted to pH 5.5 with 0.5 M phosphoric acid, - flow rate: 1 ml per minute. - spectrophotometer set at 254 nm, - injection volume: 20 µl. The retention time for colchicine is between 5.5 and 9.5 minutes. 14 DRAFT PROPOSAL FOR AMENDMENTS Inject the reference solution. The test is not valid unless the column efficiency is not less than 4500 theoretical plates and the relative standard deviation for replicate injections is not more than 2 per cent. Inject the reference solution and the test solution. Calculate the content of C22H25NO6. Colchicine and Probenecid Tablets. Page 1132 Assay. For colchicine, last line Change from: taking 425 as the specific absorbance at 350 nm. to: taking 440 as the specific absorbance at 350 nm. Crosscarmellose Sodium. Page 1139 Identification. A. Line 1, Change from: 0.00001 per cent to: 0.0004 per cent Cyclophosphamide Injection. Page 1146 Assay. Change to: Assay. Determine by liquid chromatography (2.4.14). Test solution. Determine the weight of the content of 10 containers. Weigh accurately a suitable quantity of the mixed contents of the 10 containers containing about 50 mg of Cyclophosphamide, dissolve in water and dilute to 100.0 ml with water. Reference solution (a). A 0.05 per cent w/v solution of cyclophosphamide RS in water. Reference solution (b). Dissolve 25 mg of cyclophosphamide RS in 25 ml of water, add 5.0 ml of ethylparaben solution prepared by dissolving 185 mg of ethylparaben in 250 ml of ethanol (95 per cent)and dilute to 1000 ml with water. Chromatographic system – a stainless steel column 30 cm x 3.9 mm, packed with octadecylsilane bonded to porous silica (5 µm), – mobile phase: a mixture of 30 volumes of acetonitrile and 70 volumes of water, – flow rate: 1.5 ml per minute, – spectrophotometer set at 195 nm, – injection volume: 25 µl. The relative retention time with respect to ethylparaben for cyclophosphamide is about 0.7. Inject reference solution (b). The test is not valid unless the resolution between cyclophosphamide and ethylparaben is not less than 2.0 and the relative standard deviation of replicate injections is not more than 2.0 per cent. Inject reference solution (a) and the test solution. Calculate the content of C7H15Cl2N2O2P in the injection. Dapsone. Page 1162 Related substances. Reference solution (b). Change to: Reference solution (b). Dilute 2.0 ml of reference solution (a) to 10.0 ml with methanol. 15 DRAFT PROPOSAL FOR AMENDMENTS Last para, line 8 Change from: reference solution (a) to: reference solution (a) (1.0 per cent) Last line Change from: reference solution (b) to: reference solution (b) (0.2 per cent) Dicyclomine Tablets. Page 1206 Identification. B. Lines 4 to 6 Change from: Add a quantity of the powdered tablets containing 20 mg of Dicyclomine Hydrochloride to: Add about 20 mg of extracted powder obtained in identification test A, Activated Dimethicone. Page 1230 Assay. For polydimethylsiloxane – Lines 3 and 11 Change from: carbon tetrachloride to: toluene For silicon dioxide – Line 4 Change from: carbon tetrachloride to: toluene Line 6 Change from: n-hexane to: toluene Disodium Edetate. Page 1234, Impurity A. After chromatographic system, para 1, line 4, Change from: more to: less Docetaxel Injection. Page 2903 Related substances. Chromatographic system, line 1 Change from: 25 cm to: 5.0 cm Assay. Chromatographic system, line 1 Change from: 25 cm to: 5.0 cm Donepezil Hydrochloride. Page 1248 Sulphated ash. Change from: Sulphated ash (2.3.19). to: Sulphated ash (2.3.18). Donepezil Tablets. Page 1249 Dissolution. Change to: Dissolution (2.5.2). Apparatus No. 1, Medium. 900 ml of 0.1 M hydrochloric acid, Speed and time. 50 rpm and 45 minutes. 16 DRAFT PROPOSAL FOR AMENDMENTS Withdraw a suitable volume of medium and filter. Determine by liquid chromatography (2.4.14). Test solution. Dilute the filtrate, if necessary, with the dissolution medium. Reference solution. Dissolve an accurately weighed quantity of donepezil RS in the dissolution medium, dilute to obtain a solution having a known concentration similar to expected concentration of test solution. Chromatographic system as described under Assay using 50 µl injection volume. Inject the reference solution and the test solution. D. Not less than 75 per cent of the stated amount of C24H29NO3,HCl. Doxofylline Tablets. Page 1254 Related substances. Last para, line 3 Change from: 0.2 times to: 0.5 times Line 4 Change from: (0.2 per cent) to: (0.5 per cent) Efavirenz, Emtricitabine and Tenofovir Tablets. Page 2904 Dissolution. Last para, line 1 Change from: Not less than 80 per cent to: Not less than 75 per cent Enoxaparin Sodium. Page 1276 Identification. A. After chromatographic system, para1 Change to: Inject the reference solution. The test is not valid unless the column efficiency is not less than 10000 theoretical plates for refractive index detector. Benzyl alcohol. After chromatographic system, para 1and 2 Change to: Inject the reference solution and the test solution. In the chromatogram obtained with the reference solution, calculate the ratio (R1) of the height of the peak due to benzyl alcohol to the height of the peak due to the internal standard. In the chromatogram obtained with the test solution, calculate the ratio (R 2) of the height of the peak due to benzyl alcohol to the height of the peak due to the internal standard. Calculate the percentage content (m/m) of benzyl alcohol using the following expression: m = mass of the substance under examination, in grams. The content is not more than 0.1 per cent of benzyl alcohol Enoxaparin Injection. Page 1279 Anti-factor IIa activity. Lines 1 and 2 Change from: Not less than 20.0 and not more than 35.0 anti-factor IIa IU per mg. to: Not less than 2000 and not more than 3500 anti-factor IIa IU per ml. 17 DRAFT PROPOSAL FOR AMENDMENTS Anti-factor Xa to anti-factor IIa ratio. Line 2 and 4, Change from: IU per mg to: IU per ml Erythromycin. Page 1290 Assay. Line 4 Change from: water to: sterile phosphate buffer pH 8.0 Erythromycin Stearate Tablets. Page 1292 Disintegration. Change to: Dissolution (2.5.2). Apparatus No. 1, Medium. 900 ml of 2.72 per cent w/v solution of sodium acetate, adjusted to pH 5 with glacial acetic acid, Speed and time. 50 rpm and 45 minutes. Withdraw a suitable volume of medium and filter. To 5 ml of the filterate, add 40 ml of glacial acetic acid and 10 ml of a 0.5 per cent w/v solution of 4- dimethylaminobenzaldehyde in glacial acetic acid and dilute to 100 ml with a mixture of 35 volumes of glacial acetic acid and 70 volumes of hydrochloric acid. Allow to stand for 15 minutes and measure the absorbance of the resulting solution at the maximum at 485 nm (2.4.7). Calculate the content of C37H67NO13 in the medium from the absorbance obtained from a solution of known concentration of erythromycin stearate RS prepared in the same manner. D. Not less than 70 per cent of the stated amount of C37H67NO13. Erythromycin Tablets. Page 1291 Assay. Para 2, line 2 and 3 Change from: 0.4 g of Erythromycin and triturate with 10 ml of sterile phosphate buffer pH 8.0 to: 0.25 g of Erythromycin and triturate with 10 ml of methanol. Ethinyloestradiol Tablets. Page 1307 Assay. Delete the follwoing Take 1 ml of this solution and dilute to 10 ml with the same solvent. Etoposide. Page 1322 Related substances. Determine by liquid chromatography (2.4.14). Solvent mixture. Equal volumes of mobile phase A and mobile phase B. Test solution (a). Dissolve 40 mg of the substance under examination in the solvent mixture and dilute to 10.0 ml with the solvent mixture. Test solution (b). Dissolve 50 mg of the substance under examination in the solvent mixture and dilute to 50.0 ml with the solvent mixture. Reference solution (a). Dilute 1.0 ml of test solution (a) to 100.0 ml with the solvent mixture. Dilute 1.0 ml of this solution to 10.0 ml with the solvent mixture. Reference solution (b) To 10 ml of test solution (b), add 0.1 ml of a 4 per cent v/v solution of glacial acetic acid and 0.1 ml of phenolphthalein solution. Add 1 M sodium hydroxide until the solution becomes faintly pink (about 0.15 ml). After 15 minutes, add 0.1 ml of a 4 per cent v/v solution of glacial acetic acid. Reference solution (c) A 0.1 per cent w/v solution of etoposide RS in the solvent mixture. 18 DRAFT PROPOSAL FOR AMENDMENTS Chromatographic system – a stainless steel column 12.5 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm), – column temperature 40º, – mobile phase: A. a mixture of 1 volume of triethylamine, 1 volume of anhydrous formic acid and 998 volumes of water, B. a mixture of 1 volume of triethylamine, 1 volume of anhydrous formic acid and 998 volumes of acetonitrile, – flow rate. 1 ml per minute, – a gradient programme using the conditions given below, – spectrophotometer set at 285 nm, – injection volume. 10 µl. Time Mobile Mobile phase A phase B (min) (per cent v/v) (per cent v/v) 0-7 75 25 7 - 23 75 - 27 25 - 73 23 - 25 27 - 75 73 - 25 The relative retention time with reference to Etoposide (Retention time: about 5 minutes) for lignan P (etoposide impurity D) is about 0.4, for 4’-desmethylepipodophyllotoxin (etoposide impurity E) is about 0.8; for α-etoposide (etoposide impurity C) is about 1.1; for cis-etoposide (etoposide impurity B) is about 1.2; for (5R,5aR,8aR,9S)-9-[[4,6O-[(R)-ethylidene]-β-D-glucopyranosyl]oxy]-5-[4-[[(5R,5aR,8aR,9S)-5-(4-hydroxy-3,5-dimethoxyphenyl)-6-oxo5,5a,6,8,8a,9-hexahydroisobenzofuro[5,6-f][1,3]benzodioxol-9-yl]oxy]3,5-dimethzxoxyphenyl]-5,8,8a,9tetrahydroisobenzofuro-[5,6-f][1,3]benzodioxol-6(5aH)-one (etoposide impurity N) is about 3.1; and for (5R,5aR,8aR,9S)-9-[[2,3-bis-O-(dichloroacetyl)-4,6-O-[(S)-ethylidene]-β-L-glucopyranosyl]oxy]-5-(4-hydroxy-3,5dimethoxyphenyl)-5,8,8a,9-tetrahydroisobenzofuro[5,6-f][1,3]benzodioxol-6(5aH)-one (etoposide impurity O) is about 4.2. The correction factor for Etoposide impurity O is 1.7. Inject reference solution (b). The test is not valid unless the resolution between the peaks due to etoposide and to cisetoposide is not less than 3.0. Ignore any peak due to phenolphthalein. Inject reference solution (a) and test solution (a). The area of any peak corresponding to etoposide impurities B, C, D, E and N is not more than the twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent), the area of any peak corresponding to etoposide impurity O is not more than 1.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.15 per cent), the area of any other secondary peak is not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.1 per cent), the sum of the areas of all the secondary peaks is not more than 10 times the area of the principal peak in the chromatogram obtained with reference solution (a) (1 per cent). Ignore any peak with an area less than 0.5 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05). Fentanyl Citrate. Page 1339 Related substances. Reference solution (b). Insert at the end, To prepare degradation compound N-phenyl-1-(2-phenylethyl)piperidin-4-amine (fentanyl citrate impurity D). After chromatographic system, para 2, Change to: Inject reference solution (a) and the test solution. In the chromatogram obtained with the test solution the area of any secondary peak is not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (0.25 per cent). The sum of the areas of all the secondary peaks is not more than twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.5 per cent). Ignore any peak with an area less than 0.2 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent). 19 DRAFT PROPOSAL FOR AMENDMENTS Finasteride Tablets. Page 1351 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Solvent mixture. 50 volumes of acetonitrile and 50 volumes of water. Test solution. Dissolve a quantity of powdered tablets containing about 100 mg of Finasteride in 30 ml of the solvent mixture and dilute to 50.0 ml with the solvent mixture, centrifuge and filter the supernatant liquid. Reference solution. Dilute 1.0 ml of the test solution to 100.0 ml with the solvent mixture. Chromatographic system – a stainless steel column 25 cm x 4.0 mm, packed with octadecylsilane bonded to porous silica (5 µm) (Such as Spherisorb ODS 2), – column temperature: 60°, – mobile phase: a mixture of 10 volumes of acetonitrile, 10 volumes of tetrahydrofuran and 80 volumes of water, – flow rate: 1.5 ml per minute, – spectrophotometer set at 210 nm, – injection volume: 20 µl. -------------------------------------------------------------------------------------------------------------------------------------------------------- Name Relative retention time Finasteride impurity A1 Finasteride (Retention time: about 28 minutes) Finasteride impurity B2 Finasteride impurity C3 0.9 1.0 1.2 1.4 1 N-(1,1-dimethylethyl)-3-oxo-4-aza-5a-androstane-17b-carboxamide Methyl 3-oxo-4-aza-5a-androst-1-ene-17b-carboxylate 3 N-(1,1-dimethylethyl)-3-oxo-4-azaandrosta-1,5-diene-17b-carboxamide 2 Inject the reference solution. The test is not valid unless the column efficiency is not less than 2000 theoretical plates and the tailing factor is not more than 2.0. Inject the reference solution and the test solution. Run the chromatogram twice the retention time of the principal peak. In the chromatogram obtained with the test solution the area of any peak corresponding to finasteride impurities A, B and C, is not more than 0.3 times the area of the principal peak in the chromatogram obtained with the reference solution (0.3 per cent). The area of any other secondary peak is not more than 0.1 times the area of the principal peak in the chromatogram obtained with the reference solution (0.1 per cent) and the sum of the areas of all other secondary peaks is not more than 0.6 times the area of the principal peak in the chromatogram obtained with the reference solution (0.6 per cent). Ignore any peak with an area less than 0.05 times the area of the principal peak in the chromatogram obtained with the reference solution (0.05 per cent). Fluoxetine Hydrochloride. Page 1369 Related substances. After chromatographic system, para 2, lines 6 to 8 Change from: the peak to valley ratio for fluoxetine impurity C is not greater than 1.1. If the ratio is greater than 1.1, to: the peak to valley ratio for fluoxetine impurity C is not greater than 11. If the ratio is greater than 11, Folic Acid. Page 1384 Related substances. Reference solution (b). Line 3 Change from: with the mobile phase. to: with the same solution. 20 DRAFT PROPOSAL FOR AMENDMENTS Reference solution (e). Line 3 Change from: with the mobile phase. to: with the same solution. After chromatographic system, para 1 , insert at the end and for 2-amino-7-(chloromethyl)pteridin-4(1H)-one (folic acid impurity F) is about 2.2. Framycetin Sulphate. Page 1388 Alcohols. Delete the requirement. Gemifloxacin Tablets. Page 1409 Assay. Test solution. Change to: Weigh and powder 20 tablets. Disperse a quantity of powder containing about 100 mg of gemifloxacin, in water and dilute to 100.0 ml with water and filter. Dilute 10.0 ml of this solution to 100.0 ml with water. Gentamicin Sulphate. Page 1410 Para 2, line 2 Change from: anhydrous basis to: dried basis Water. Change to: Loss on drying. Not more than 18.0 per cent, determined on 1.0 g by drying in an oven at 110°, at a pressure not exceeding 0.7 kPa for 3 hours. Glibenclamide. Page 1414 Assay. Line 4 Change from: red colour to: pink colour Glycerin. Page 1422 Assay. Change to: Assay. Weigh accurately about 0.1 g, mix thoroughly with 45 ml of water, add 25.0 ml of a mixture of 1 volume of 0.1 M sulphuric acid and 20 volumes of 0.1 M sodium periodate. Allow to stand protected from light for 15 minutes. Add 5 ml of a 50 per cent w/v solution of ethylene glycol, allow to stand protected from light for 20 minutes and titrate with 0.1 M sodium hydroxide using 0.5 ml of phenolphthalein solution as indicator. Carry out a blank titration. 1 ml of 0.1 M sodium hydroxide is equivalent to 0.00921 g of C3H8O3. Hydrochloric Acid. Page 1450 Residue on evaporation. Insert at the end, by evaporating to dryness on a water-bath and dry at 105°. Dilute Hydrochloric Acid. Page 1450 Residue on evaporation. Insert at the end, by evaporating to dryness on a water-bath and dry at 105°. Hydroxypropylcellulose. Page 1464 Apparent viscosity. Insert at the end. For a product of low viscosity, use a quantity of the substance under examination sufficient to prepare a solution of the concentration stated on the label. 21 DRAFT PROPOSAL FOR AMENDMENTS Hyoscine Butylbromide. Page 1466 Identification. Change to: Identification. A. Determine by infrared absorption spectrophotometry (2.4.6). Compare the spectrum with that obtained with hyoscine butylbromide RS or with the reference spectrum of hyoscine butylbromide. B. Gives the reactions of bromides (2.3.1). Hyoscine Butylbromide Injection. Page 1467 Identification. B. Change to: When examined in the range 230 nm to 360 nm (2.4.7), a 0.15 per cent w/v solution in 0.01 M hydrochloric acid shows absorption maxima at about 252 nm, 257 nm and 264 nm and a less well-defined maximum at about 247 nm. Hyoscine Butylbromide Tablets. Page 1468 Identification. B. Change to: When examined in the range 230 nm to 360 nm (2.4.7), a 0.15 per cent w/v solution in 0.01 M hydrochloric acid shows absorption maxima at about 252 nm, 257 nm and 264 nm and a less well-defined maximum at about 247 nm. Lamivudine and Zidovudine Tablets. Page 1561 Dissolution. Line 3 Change from: 50 rpm to: 75 rpm Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Test solution. Disperse a quantity of the powdered tablets containing about 150 mg of Lamivudine in methanol and dilute to 100.0 ml with methanol, filter. Reference solution (a). A 0.01 per cent w/v solution of thymine in methanol. Reference solution (b). A solution containing 0.3 per cent w/v of zidovudine RS and 0.15 per cent w/v of lamivudine RS in methanol. Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with octadecylsilane chemically bonded to porous silica (5 μm), – column temperature: 35°, – mobile phase: A. a buffer solution prepared by dissolving 1.945 g of ammonium acetate in 900 ml of water, adjusted to pH 3.8 with glacial acetic acid and diluting to 1000.0 ml with water, B. methanol, – flow rate; 1 ml per minute, – a gradient programme using the conditions given below, – spectrophotometer set at 270 nm, – injection volume: 10 μl. Time (in min.) 0 Mobile phase A (per cent v/v) 90 Mobile phase B (per cent v/v) 10 22 DRAFT PROPOSAL FOR AMENDMENTS 10 25 40 45 90 80 90 90 10 20 10 10 Inject reference solution (b). The test is not valid unless the tailing factor of the lamivudine and zidovudine peaks is not more than 2.0. Inject reference solution (a) and the test solution. In the test solution, the content of thymine is not more than 2.0 per cent and of any unknown impurity not more than 0.5 per cent. The sum of all the impurities is not more than 3.0 per cent, calculated by area normalization. Lamivudine, Nevirapine and Stavudine Dispersible Tablets. Page 1563 Usual strengths. Change to: Usual strengths. Lamivudine 30 mg, Nevirapine 50 mg and Stavudine 6 mg; Lamivudine 60 mg, Nevirapine 100 mg and Stavudine 12 mg. Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). NOTE — Prepare the solutions immediately before use. Solvent mixture. 70 volumes of a solution containing 0.2 per cent v/v of ortho-phosphoric acid and 30 volumes of methanol. Test solution. Weigh and powder 20 tablets. Weigh accurately a quantity of the powder containing 75 mg of Lamivudine, dissolve in the solvent mixture and dilute to 100.0 ml with the solvent mixture, filter. Reference solution. Weigh accurately about 75 mg of lamivudine RS, 130 mg of nevirapine RS and 20 mg of stavudine RS, dissolve in 20 ml of methanol and dilute to 100.0 ml with the solvent mixture. Dilute 1.0 ml of the solution to 100.0 ml with the solvent mixture. Chromatographic system – a stainless steel column 25 cm 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm), – mobile phase: A. methanol, B. a buffer solution prepared by dissolving 1.925 g of ammonium acetate in 1000 ml of water and adjusted to pH 3.0 with trifluoroacetic acid, – flow rate: 1.2 ml per minute, – a gradient programme using the conditions given below, – spectrophotometer set at 266 nm, – injection volume: 10 µl. Time Mobile phase A Mobile phase B (min.) ( per cent v/v) ( per cent v/v) 0 5 95 15 15 85 30 45 55 40 60 40 48 60 40 51 5 95 60 5 95 The relative retention time with respect to stavudine, for carboxylic acid is 0.36, for thymine is 0.42, for lamivudine is 0.74 and for nevirapine is 2.19. 23 DRAFT PROPOSAL FOR AMENDMENTS Inject the reference solution. The test is not valid unless the column efficiency is not less than 2000 theoretical plates and tailing factor is not more than 1.5 for each component. Inject the reference solution and the test solution. In the chromatogram obtained with the test solution, the area of any secondary peak is not more than 3 times the area of principal peak due to lamivudine in the chromatogram obtained with the reference solution (3.0 per cent) and the sum of areas of all the secondary peaks is not more than 5 times the area of principal peak due to lamivudine in the chromatogram obtained with the reference solution (5.0 per cent). Insert before Other tests. Uniformity of content. (For tablets containing 10 mg or less). Comply with the test stated under Tablets. Determine by liquid chromatography (2.4.14) as described in the Assay using the following solution as the test solution. Test solution. Disperse 1 powdered tablet in the solvent mixture and dilute to 25.0 ml with the solvent mixture. Lansoprazole. Page 1568 Related substances. Reference solution (b) Change to: Reference solution (b). A 0.0025 per cent w/v solution of lansoprazole RS in methanol. Dilute 1.0 ml of this solution to 10.0 ml with the solvent mixture. Lansoprazole Sustained-release Capsules. Page 1570 Dissolution. Lines 13 and 14. Change from: D. Not more than 10 per cent of the stated amount of C16H14F3N3O2S. to: Complies with the acceptance criteria given under acid stage. Levocetrizine Hydrochloride. Page 1573 Insert before structure Levocetrizine Dihydrochloride Levonorgestrel Tablets. Page 2925 Identification. Insert before A. Test B may be omitted if tests A and C are carried out. Test A may be omitted if tests B and C are carried out. Insert before Tests. C. To a quantity of the powdered tablets containing 37.5 mg of levonorgestrel, extract with five 40 ml quantities of dichloromethane and filter. Combine the filtrates, evaporate to dryness on a water-bath. Dissolve the residue in 5 ml of dichloromethane. The optical rotation (2.4.22) of resulting solution is not less than -0.18º. Optical rotation. Delete the requirement. Insert before Related substances. Dextronorgestrel. Determine by liquid chromatography (2.4.14). Solvent mixture. A mixture of 80 volumes of methanol and 20 volumes of water. Test solution. Weigh accurately a quantity of powdered tablets containing 3 mg of the Levonorgestrel in 25.0-ml volumetric flask. Add about 15 ml of the solvent mixture and heat in a water bath at 60º for 10 minutes, shaking occasionally. Dilute to volume with the solvent mixture. 24 DRAFT PROPOSAL FOR AMENDMENTS Reference solution (a). A 0.0006 per cent w/v solution of levonorgestrel RS in the solvent mixture. Reference solution (b). A 0.0012 per cent w/v solution of norgestrel RS in the solvent mixture. Reference solution (c). A 0.000012 per cent w/v solution of levonorgestrel RS in the solvent mixture. Chromatographic system – a stainless steel column 15 cm 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm), – mobile phase: dissolve 5 g of gamma-cyclodextrin in 500 ml of water and dilute to 1000 ml with methanol, – flow rate: 1.5 ml per minute, – spectrophotometer set at 242 nm, – injection volume: 20 µl. Inject reference solutions (b). The test is not valid unless the resolution between the peaks corresponding to levonorgestrel and dextronorgestrel is not less than 1.5. Inject reference solutions (a), (b), (c), and the test solution. In the chromatogram obtained with the test solution the area of peak corresponding to dextronorgestrel is not more than the area of the principal peak in the chromatogram obtained with reference solution (c) (0.1 per cent). Lignocaine and Adrenaline Injection. Page 1585 Assay. For adrenaline - Change to: Determine by liquid chromatography (2.4.14). Solvent mixture. Dissolve 8.0 g of tetramethylammonium hydrogen sulphate, 2.2 g of sodium heptanesulphonate, add 2 ml of 0.1M disodium edetate to a mixture of 900 ml of water and 100 ml of methanol , adjusted to pH 3.5 using 1M sodium hydroxide. Test solution. Dilute a volume of injection to obtain a solution containing 0.0005 per cent w/v of adrenaline. Dilute 5.0 ml of the resulting solution to 10 ml with the solvent mixture. Reference solution (a). Dilute 5.0 ml of a 0.01 per cent w/v solution of adrenaline acid tartrate RS to 100.0 ml with the solvent mixture. Reference solution (b). A mixture of 5 ml of reference solution (a) with 5 ml of a 0.001 per cent w/v solution of noradrenaline acid tartrate in the mobile phase. Chromatographic system – a stainless steel column 10 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm) (Such as Nucleosil C18), – mobile phase: a solution prepared by dissolving 4.0 g of tetramethylammonium hydrogen sulphate, 1.1 g of sodium heptanesulphonate, add 2 ml of 0.1M disodium edetate to a mixture of 950 ml of water and 50 ml of methanol and adjusted to pH 3.5 with 1M sodium hydroxide, – flow rate: 2 ml per minute, – spectrophotometer set at 205 nm, – injection volume: 20 µl. Inject reference solution (b). The test is not valid unless the resolution between the two principal peaks is not less than 2.0. Inject reference solution (a) and the test solution. Calculate the content of C9H13NO3. Lithium Carbonate Tablets. Page 1596 Dissolution. Line 5, 25 DRAFT PROPOSAL FOR AMENDMENTS Change from: dilute to 100.0 ml with water. to: dilute to 100.0 ml with water. Filter and dilute with water if necessary. Loperamide Tablets. Page 1601 Dissolution. Line 4 and 5 Change from: Withdraw 10 ml of the medium from each vessel, mix and filter. Test solution. The mixed filtrate from the dissolution medium. to: Withdraw a suitable volume of the medium and filter. Test Solution. Use the filtrate. Medroxyprogesterone Acetate. Page 1640 Related substances. Last para, Change to: Inject test solutions (a) and (b). Record the chromatograms for 1.5 times the retention time of the principal peak. In the chromatogram obtained with test solution (b) the area of any secondary peak is not more than 0.5 times the area of the principal peak in the chromatogram obtained with test solution (a) (1.0 per cent) and the sum of the areas of all the secondary peaks is not more than 0.75 times the area of the principal peak in the chromatogram obtained with test solution (a) (1.5 per cent). Ignore any peak with an area less than 0.025 times the area of the principal peak in the chromatogram obtained with test solution (a) (0.05 per cent). Menthol. Page 1649 Related substances. Chromatographic system, line 6 Change from: column 80° to: column 80°, after 2 minutes, increase the temperature of the column to 240° at a rate of 8° per minute and maintain at this temperature for 15 minutes. After chromatographic system, para 1 and 2 Change to: Inject 1 µl of each solution. Run the chromatogram obtained with the test solution for 3 times the retention time of the principal peak. In the chromatogram obtained with the test solution the sum of the areas of all the secondary peaks is not more than the area of the principal peak in the chromatogram obtained with reference solution (a). Ignore any peak with an area less than the principal peak in the chromatogram obtained with reference solution (b). Meropenem Injection. Page 1656 Amendment list 4 Sodium carbonate. Insert Sodium carbonate. 80 per cent to 120 per cent of the labeled amount of sodium. Minoxidil. Page 1697 Heavy metals. Change to: Dissolve 1.0 g in 25 ml of methanol, the solution complies with the limit test for heavy metals, Method C (20 ppm). Montelukast Sodium. Page 1704 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). NOTE—Carry out the test protected from light. 26 DRAFT PROPOSAL FOR AMENDMENTS Solvent mixture. 10 volumes of water and 90 volumes of methanol. Test solution (a). Dissolve 50 mg of the substance under examination in the solvent mixture and dilute to 50.0 ml with the solvent mixture. Test solution (b). Dilute 1.0 ml of test solution (a) to 10.0 ml with the solvent mixture. Reference solution (a). Dilute 1.0 ml of test solution (a) to 100.0 ml with the solvent mixture. Dilute 1.0 ml of this solution to 10.0 ml with the solvent mixture Reference solution (b). Dissolve 10 mg of montelukast for peak identification RS (containing montelukast impurities B,C, D, E and F) in the solvent mixture and dilute to 10.0 ml with the solvent mixture. Reference solution (c). In order to prepare montelukast impurity G in-situ, transfer 1 ml of reference solution (b) in a colourless glass vial and expose to ambient light for about 20 minutes. Reference solution (d). Dissolve 65 mg of montelukast dicyclohexylamine RS in the solvent mixture and dilute to 50.0 ml with the solvent mixture. Dilute 10.0 ml of the solution to 100.0 ml with the solvent mixture. Chromatographic system – a stainless steel column 5 cm x 4.6 mm, packed with phenylsilyl bonded to porous silica (1.8 µm), – column temperature: 30º, – mobile phase: A. mix 1.5 ml of trifluoroacetic acid with 1000 ml of water, B. mix 1.5 ml of trifluoroacetic acid with 1000 ml of acetonitrile, – a linear gradient programme using the conditions given below, – flow rate: 1.2 ml per minute, – spectrophotometer set at 238 nm, – injection volume: 10 µl. Time Mobile phase A Mobile phase B (in min.) (per cent v/v) (per cent v/v) 03 60 40 316 6049 4051 Inject reference solution (c). The test is not valid unless the resolution between montelukast and montelukast impurity G is not less than 2.5 and montelukast impurity F is not less than 1.5. The relative retention time with reference to montelukast for [1-[[[1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-hydroxy-1methylethyl)phenyl]propyl]sulfinyl]methyl]cyclopropyl]acetic acid (montelukast impurity C) is about 0.4; for [1[[[(1R)-1-[3-[(Z)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1-hydroxy-1methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid (montelukast impurity G) is about 0.8; for 1[[[(1R)-1-[3-[(1R)-1-[[[1-(carboxymethyl)cyclopropyl]methyl]sulfanyl]-2-(7-chloroquinolin-2-yl)ethyl]phenyl]-3[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid (montelukast impurity D) and 1-[[[(1R)-1-[3-[(1S)-1-[[[1-(carboxymethyl)cyclopropyl]methyl]sulfanyl]-2-(7-chloroquinolin-2-yl)ethyl]phenyl]-3[2-(1-hydroxy-1-methylethyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid (montelukast impurity E) is about 0.9; for [1-[[[(1R)-3-(2-acetylphenyl)-1-[3-[(E)-2-(7-chloroquinolin-2yl)ethenyl]phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid (montelukast impurity F) is about 1.2 and for [1[[[(1R)-1-[3-[(E)-2-(7-chloroquinolin-2-yl)ethenyl]phenyl]-3-[2-(1methylethenyl)phenyl]propyl]sulfanyl]methyl]cyclopropyl]acetic acid (montelukast impurity B) is about 1.9. Inject reference solution (a) and test solution (a). In the chromatogram obtained with test solution (a) the area of any peak corresponding to montelukast impurity B is not more than three times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.3 per cent), the area of any peak corresponding to montelukast impurity C is not more than twice the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent). The sum of the areas of all the secondary peaks is not more than six times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.6 per cent). Ignore any peak with an area less than 0.5 times of the area of the principal peak in the chromatogram obtained with reference solution (a) (0.05 per cent). 27 DRAFT PROPOSAL FOR AMENDMENTS Multiple Electrolyte and Dextrose Injection Type V. Page 1715 Assay. For magnesium. Para 1, Insert at the end, Carry out a blank titration using 17.5 mg of CaCl2.2H2O dissolved in 50 ml of distilled water, add 5.0 ml of ammonia-ammonium chloride solution and dilute to 250 ml with water. Titrate this solution with 0.005 M disodium edetate using 50 mg of eriochrome black T mixture as indicator. Calculate the content of magnesium with the volume obtained by subtracting the volume of EDTA required for calcium from the consumption of 0.005 M EDTA in the titration of magnesium. Neotame. Page 1769 Para 2, line 2 Change from: dried to: anhydrous Nifedipine Sustained-release Tablets. Page 1781 Dissolution. Last para, Change to: D. Not less than 60 per cent cumulative of both the medium of the stated amount of C17H18N2O6. Olanzapine Tablets. Page 1812 Assay. Chromatographic system, line 10 Change from: 220 nm to: 260 nm Omeprazole Capsules. Page 1814 Dissolution. B. Line 6 to 8 Change from: Using the filtered medium as the test solution, carry out the determination as described in the Assay. to: Immediately transfer 5.0 ml of the solution to a test tube containing 1.0 ml of 0.1 M sodium hydroxide. Prepare the standard solution having a known concentration similar to the expected concentration and in similar manner to the test solution. Note:- A query was received from Dr. Hitesh Jogia of Dr. Reddy’s Labs of dated 26-09-12, corrected typoerror. This monograph is not available in BP and USP. Ondansetron Orally Disintegrating Tablets. Page 2938 Disintegration. Change to: Disintegration (2.5.1). Not more than 30 seconds. Ondansetron Tablets. Page 1819 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Solvent mixture. Equal volumes of acetonitrile and buffer solution prepared by dissolving 2.7 g of monobasic potassium phosphate in 1000 ml of water adjusted to pH 5.4 with 1M sodium hydroxide. Test solution. Weigh and powder 20 tablets. Weigh accurately a quantity of the powder containing about 50 mg of Ondansetron, disperse in 70 ml of the mobile phase, with the aid of ultrasound for 20 minutes, dilute to 100.0 ml with the mobile phase, centrifuge. Reference solution (a). A 0.0005 per cent w/v solution of ondansetron hydrochloride RS in the solvent mixture. 28 DRAFT PROPOSAL FOR AMENDMENTS Reference solution (b). A solution containing 0.005 per cent w/v solution of ondansetron impurity A RS and 0.01 per cent w/v of ondansetron hydrochloride RS in the mobile phase. Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with nitrile group bonded to porous silica (5 µm), – mobile phase: a mixture of 20 volumes of acetonitrile and 80 volumes of buffer solution prepared by dissolving 2.7 g of monobasic potassium phosphate in 1000 ml of water, adjusted to pH 5.4 with 1M sodium hydroxide, – flow rate: 1.5 ml per minute, – spectrophotometer set at 216nm, – injection volume: 10 µl. Name Relative retention time 2-methyl-imidazole Ondansetron impurity C1 Ondansetron impurity D2 Ondansetron impurity A3 Desmethylondansetron4 Ondansetron 0.22 0.40 0.47 0.87 0.90 1.0 Correction factor 1.89 0.83 0.77 1.11 1.1 - 1 1,2,3,9-tetrahydro-9-methyl-4H-carbazol-4-one. 1,2,3,9-tetrahydro-9-methyl-3-methylene-4H-carbazol-4-one. 3 3[(dimethylamino)methyl]- 1,2,3,9-tetrahydro-9-methyl-4H-carbazol-4-one. 4 1,2,3,9-tetrahydro-9-methyl-3-[(1H-imidazol-yl-)methyl]-4H-carbazol-4-one. 2 Inject reference solution (b). The test is not valid unless the resolution between the ondansetron impurity A and ondansetron is not less than 2.0. Inject reference solution (a) and the test solution. In the chromatogram obtained with the test solution the area of any secondary peak due to 2-methyl imidazole , desmethylondansetron, ondansetron impurity A and ondansetron impurity C is not more than 0.2 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent). The area of any secondary peak due to ondansetron impurity D is not more than 0.1 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.1 per cent). The area of any other secondary peak is not more than 0.2 times the area of the principal peak in the chromatogram obtained with reference solution (a) (0.2 per cent). The sum of areas of all the secondary peaks is not more than the area of the principal peak in the chromatogram obtained with reference solution (a) (1.0 per cent). Oseltamivir Phosphate. Page 1827 Loss on drying. Change to: Water (2.3.43). Not more than 0.5 per cent, determined on 0.5 g. Oseltamivir Capsules. Page 1828 Water. Delete the requirement. D‐Panthenol. Page 1856 Structure change to: 29 DRAFT PROPOSAL FOR AMENDMENTS HO H OH H N OH Me Me O Paracetamol Oral Suspension. Page 2940 Related substances. After chromatographic system, para 2, lines 5 and 6 Change from: not more than the area of the corresponding peak in reference solution (b) (0.1 per cent) to: not more than five times the area of the corresponding peak in reference solution (b) (0.5 per cent). Paracetamol Syrup. Page 1860 Assay. Test solution. Change to: Test solution. Mix an accurately weighed quantity of the preparation under examination containing 50 mg of Paracetamol in 70 ml of the mobile phase, dilute to 100.0 ml with the mobile phase. Further dilute 5.0 ml of the solution to 20.0 ml with the mobile phase. Pentazocine Hydrochloride. Page 1873 Assay. Line 2, Change from: anhydrous glacial acetic acid to: anhydrous glacial acetic acid, add 10.0 ml of mercuric acetate solution. Pilocarpine Eye Drops. Page 2943 pH. Change from: 2.5 to 4.2 to: 3.0 to 5.5 Polyoxyl 35 Castor Oil. Page 1931 Heavy metals. Last line Change from: lead standard solution (1 ppm) Change to: lead standard solution (10 ppm) Polyoxyl 40 Hydrogenated Castor Oil. Page 1931 Heavy metals. Last line Change from: lead standard solution (1 ppm) Change to: lead standard solution (10 ppm) Pregabalin. Page 1960 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Solvent mixture. Dissolve 1.2 g of monobasic potassium phosphate in 1000 ml of water, adjust the pH to 6.9 with 5 M potassium hydroxide. Test solution. Dissolve 125 mg of the substance under examination in the solvent mixture and dilute to 10.0 ml with the solvent mixture. 30 DRAFT PROPOSAL FOR AMENDMENTS Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with octylsilane bonded to porous silica (5 µm) (Such as Kromasil 100), – mobile phase: A. dissolve 1.2 g of monobasic potassium phosphate in 940 ml of water, adjusted to pH 6.9 with 5 M potassium hydroxide and 60 ml of acetonitrile, B. dissolve 1.2 g of monobasic potassium phosphate in 700 ml of water, adjusted to pH 6.7 with 5 M potassium hydroxide and 300 ml of acetonitrile, – a gradient programme using the conditions given below, – flow rate: 1.5 ml per minute, – spectrophotometer set at 210 nm, – injection volume: 40 µl. Time (in min) 0-4 4-45 45-46 46-60 Mobile phase A (per cent v/v) 100 100 0 100 Mobile phase B (per cent v/v) 0 0 100 1000 0 The relative retention time with reference to pregabalin for lactum impurity is 6.11. Inject the test solution. The area of any peak corresponding to lactum impurity is not more than 0.5 per cent. The area of any other secondary peak is not more than 0.5 per cent and the sum of areas of all the secondary peaks is not more than 1.0 per cent, calculated by area normalization. Primaquine Tablets. Page 1964 Change the monograph to: Primaquine Tablets Primaquine Phosphate Tablets Primaquine Tablets contain not less than 90.0 per cent and not more than 110.0 per cent of the stated amount of primaquine, C15H21N3O. Usual strength. The equivalent of 2.5 mg of primaquine (13 mg of Primaquine Phosphate is approximately equivalent to 7.5 mg of primaquine). Identification A. Dissolve a quantity of the powdered tablets containing 60 mg of primaquine in a mixture of 10 ml of water and 2 ml of 2 M sodium hydroxide and extract with two quantities, each of 20 ml, of chloroform. Wash the chloroform extracts with water, dry over anhydrous sodium sulphate, evaporate to dryness and dissolve the residue in 2 ml of chloroform. The residue complies with the following test. Determine by infrared absorption spectrophotometry (2.4.6). Compare the spectrum with that obtained with primaquine phosphate RS or with the reference spectrum of primaquine. B. Extract a quantity of the powdered tablets containing 25 mg of primaquine with 10 ml of water and filter. To 2 ml of the filtrate, add 3 ml of water and 1 ml of a 5 per cent w/v solution of ceric ammonium sulphate in 2 M nitric acid; a deep violet colour is produced immediately. Tests Dissolution (2.5.2). Apparatus No. 1, Medium. 900 ml of 0.01 M hydrochloric acid, Speed and time. 50 rpm and 60 minutes. 31 DRAFT PROPOSAL FOR AMENDMENTS Withdraw a suitable volume of the medium and filter. Determine by liquid chromatography (2.4.14). Test solution. Dilute the filtrate, if necessary, with the dissolution medium. Reference solution. Dissolve an accurately weighed quantity of primaquine phosphate RS in the dissolution medium and dilute with dissolution medium to obtain a solution having a known concentration similar to the expected concentration of the test solution. Chromatographic system as described in the Assay Inject the reference solution. The test is not valid unless the relative standard deviation for replicate injections is not more than 3.0 per cent. Inject the reference solution and the test solution. D. Not less than 75 per cent of the stated amount of C15H21N3O. Uniformity of content. (For tablets containing 10 mg or less) Comply with the test stated under tablets. Determine by liquid chromatography (2.4.14), as described in the Assay, using the following solutions. Test solution. Powder one tablet, dissolve in 20 ml of the mobile phase with the aid of ultrasound for 3 minutes and dilute to 50.0 ml with the mobile phase and filter. Reference solution. Dissolve an accurately weighed quantity of primaquine phosphate RS in the mobile phase and dilute with the mobile phase to obtain a solution having a known concentration similar to the expected concentration of the test solution. Calculate the content of C15H21N3O in the tablet. Other tests. Comply with the tests stated under Tablets. Assay. Determine by liquid chromatography (2.4.14). Test solution. Weigh and powder 20 tablets. Weigh accurately a quantity of the powder containing 10 mg of primaquine, dissolve in 30 ml of the mobile phase and sonicate for 20 minutes. Make up the volume to 50.0 ml with mobile phase. Reference solution. A 0.02 per cent w/v solution of primaquine phosphate RS in the mobile phase. Chromatographic system – a stainless steel column 15 cm x 3.0 mm, packed with octadecylsilane bonded to porous silica (3 µm), – column temperature. 30º, – mobile phase: a mixture of 40 volumes of 1- pentane sulphonate sodium solution prepared by dissolving 960 mg of sodium-1- pentane sulphonate and 1 ml glacial acetic acid to 400 ml of water and 60 volumes of methanol, – flow rate. 0.6 ml per minute, – spectrophotometer set at 264 nm, – injection volume. 5 µl. Inject the reference solution. The test is not valid unless the column efficiency in not less than 2000 theoretical plates, the tailing factor is not more than 2.0 and the relative standard deviation for replicate injections is not more than 2.0 per cent. Inject the reference solution and the test solution. Calculate the content of C15H21N3O. Storage. Store protected from moisture. Labelling. The label states the strength in terms of the equivalent amount of primaquine. Prochlorperazine Maleate. Page 1972 Identification. A. Line 8 Change from: prochlorperazine maleate RS to: prochlorperazine maleate RS treated in the same manner Prochlorperazine Tablets. Page 1972 Identification. A. Line 9 32 DRAFT PROPOSAL FOR AMENDMENTS Change from: prochlorperazine maleate RS to: prochlorperazine maleate RS treated in the same manner Psoralen. Page 2001 Assay. Change to: Weigh accurately about 0.1 g and dissolve in sufficient methanol to produce 100.0 ml. Dilute 2.0 ml of this solution to 100.0 ml with methanol. Further dilute 1.0 ml of this solution to 10.0 ml with methanol and measure the absorbance of the resulting solution at the maximum at about 247 nm (2.4.7). Calculate the content of C11H6O3 from the absorbance obtained by repeating the operation using a final solution of 2 µg per ml of psoralen RS in methanol in place of the substance under examination. Quinine Dihydrochloride Injection. Page 2023 Identification A. Test solution. Line 2 Change from: Quinine Dihydrochloride Bisulphate to: Quinine Dihydrochloride Rabeprazole Sodium. Page 2037 Storage. Change to: Storage. Store protected from light and moisture, at a temperature between 2º to 8 º. Do not freeze. Ramipril and Hydrochlorothiazide Tablets. Page 2041 Related substances. Last para, last line Insert at the end Ignore the peak due to hydrochlorthiazide. Uniformity of content. Change to: Uniformity of content. Comply with the test stated under Tablets. Determine by liquid chromatography (2.4.14), as described under Assay. Test solution. Disperse 1 intact tablet in water and dilute to 25.0 ml with the mobile phase. Dilute 3.0 ml of this solution to 25.0 ml with the mobile phase and mix. Assay. Change to: Assay. Determine by liquid chromatography (2.4.14). Test solution. Weigh and powder 20 tablets. Weigh accurately a quantity of powder containing about 25 mg of Ramipril, disperse in water and dilute to 250.0 ml with the mobile phase, filter. Dilute 3.0 ml of this solution to 25.0 ml with the mobile phase and mix. Reference solution (a). A 0.024 per cent w/v solution of ramipril RS in the mobile phase. Reference solution (b). A 0.06 per cent w/v solution of hydrochlorothiazide RS in the mobile phase. Reference solution (c). Dilute reference solution (a) and (b) with the mobile phase to obtain a solution having a known concentration similar to the test solution. Chromatographic system – a stainless steel column 25 cm x 4.6 mm, packed with octadecylsilane bonded to porous silica (5 µm) (Such as Thermo quest Hypersil), – mobile phase: a mixture of 55 volumes of water, 45 volumes of acetonitrile and 0.1 volume of triethylamine, adjusted to pH 3.0 with orthophosphoric acid, 33 DRAFT PROPOSAL FOR AMENDMENTS – flow rate: 1 ml per minute, – spectrophotometer set at 210 nm, – injection volume: 20 µl. Inject reference solution (c). The test is not valid unless the relative standard deviation for replicate injections for each peak is not more than 2.0 per cent. Inject reference solution (c) and the test solution. Calculate the content of C23H32N2O5 and C7H8ClN3O4S2 in the tablet. Salbutamol Sulphate. Page 2085 Related substances. Last para, line 9 Change from: 0.05 per cent to: 0.17 times Last line Change from: reference solution. to: reference solution (0.05 per cent). Sildenafil Citrate. Page 2100 Related substances. After chromatographic system, para 1, Change to: The relative retention time with reference to sildenafil for citric acid is about 0.11, for N-oxide is about 0.29 and for chlorosulphonyl is about 1.6. Inject reference solution (b). The test is not valid unless the relative standard deviation for replicate injections is not more than 5.0 per cent. Loss on drying. Change to: Water (2.3.43). Not more than 2.5 per cent, determined on 0.2 g. Sodium Bicarbonate Injection. Page 2111 Bacterial endotoxins. Change to: Bacterial endotoxins (2.2.3). Not more than 1.7 Endotoxin Units per ml. Sodium Methylparaben. Page 2132 Para 2, line 1, Change from: 99.0 per cent to: 95.0 per cent Chlorides. Line 1 Change from: 0.2 ml to: 0.4 ml Sodium Propylparaben. Page 2134 Para 2, line 1, Change from: 99.0 per cent to: 94.0 per cent Chlorides. Line 1 Change from: 0.2 ml to: 0.4 ml 34 DRAFT PROPOSAL FOR AMENDMENTS Sorbic Acid.Page 2955 Heavy metals. Change to: Heavy metals. 12 ml of 5 per cent w/v solution in ethanol (95 per cent) complies with the test for heavy metals, Method D (10 ppm), using 5 ml of lead standard solution (1ppm Pb) and 5 ml of ethanol (95 per cent). Thiopentone Sodium. Page 2216 Insert before Storage Thiopentone sodium intended for use in the manufacture of parenteral preparations without a further appropriate sterilization procedure complies with the following additional requirement. Sterility (2.2.11). Complies with the test for sterility. Storage. Insert at the end Labelling. The label states whether or not the material is intended for use in the manufacture of parenteral preparations. Tiotropium Bromide Powder for Inhalation. Page 2228 Para 1, lines 1 and 2 Change from: Tiotropium Bromide to: Tiotropium Bromide Monohydrate Labelling. Line 1 Change from: active ingredient to: tiotropium Vinorelbine Injection. Page 2304 Related substances. Change to: Related substances. Determine by liquid chromatography (2.4.14). Test solution. Dilute a volume of injection containing 10 mg of Vinorelbine to 10 ml with the mobile phase. Reference solution (a). A 0.14 per cent w/v solution of vinorelbine tartrate RS in the mobile phase. Reference solution (b). Dilute 1.0 ml of reference solution (a) to 100.0 ml with the mobile phase. Reference solution (c). A solution containing 0.14 per cent w/v of vinorelbine tartrate RS and 0.001 per cent w/v of vinorelbine impurity A RS in water. Expose a portion of this solution in a suitable xenon lamp for about 1 hour, in order to generate an additional degradation product. Chromatographic system as described in the Assay. The relative retention time with respect to vinorelbine for 3’,4’,7,8-tetradehydro-3,4’-dideoxy-3,6-epoxy-6,7dihydro-C’-norvincaleukoblastine (photodegradation product) is about 0.8, for 4-O-deacetylvinorelbine (vinorelbine impurity A) is about 1.2. Inject reference solutions (a) and (c). The test is not valid unless the relative retention between vinorelbine tartrate and vinorelbine impurity A is not less than 1.1 in the chromatogram obtained with reference solution (c) and relative standard deviation for replicate injection is not more than 2.0 in the chromatogram obtained with reference solution (a). Inject reference solution (b) and the test solution. Run the chromatograms three times the retention time of the principle peak. In the chromatogram obtained with the test solution, the area of any peak corresponding to vinorelbine impurity A is not more than 0.3 times the area of the principle peak in the chromatogram obtained with 35 DRAFT PROPOSAL FOR AMENDMENTS reference solution (b) (0.3 per cent); the area of any peak corresponding to photodegradation product is not more than the area of the principle peak in the chromatogram obtained with reference solution (b) (1.0 per cent); the area of any other secondary peak is not more than 0.2 times the area of the principle peak in the chromatogram obtained with reference solution (b) (0.2 per cent) and the sum of areas of all the secondary peaks is not more than twice the area of the principle peak in the chromatogram obtained with reference solution (b) (2.0 per cent). Zidovudine, Lamivudine and Nevirapine Tablets. Page 2334 Dissolution. Line 3 Change from: 50 rpm to: 75 rpm Zinc Sulphate Dispersible Tablets. Page 2970 Identification. B, line 1, Change from 0.2 ml to: 0.1 ml Assay. Line 4, Change from: xylenol orange to: xylenol orange triturate Line 5, Change from: methenamine to: hexamine Herbs and Herbal Products Haridra Dry Extract. Page 2508 Assay. Reference solution. Change from: Reference solution. A solution containing 0.01 per cent w/v each of bisdemethoxycurcumin RS, demethoxycurcumin RS, curcumin RS in tetrahydrofuran. Dilute 5 ml of this solution to 50 ml with the mobile phase. to: Reference solution. A solution containing 0.04 per cent w/v of curcumin RS, 0.01 per cent w/v of demethoxycurcumin RS and 0.002 per cent w/v of bisdemethoxycurcumin RS in tetrahydrofuran. Dilute 5 ml of this solution to 50 ml with the mobile phase. Chromatographic system, line 1. Change from: 0.46 mm to: 4.6 mm Line 3. Change from: 40 volumes to: 56 volumes Line 4. Change from: 60 volumes to: 44 volumes After Chromatographic system, para 1, line 4. Change from: 1.3 to: 1.24. Line 5. Change from: 1.14 to: 1.12. 3.3 Liquid/HPTLC/Gas Chromatograms of Herbs Haridra Dry Extract. Page 522 Change Chromatograms to: 36 DRAFT PROPOSAL FOR AMENDMENTS HPLC Chromatogram of curcumin RS, demethoxycurcumin RS and bisdemethoxycurcumin RS HPLC Chromatogram of Haridra Dry Extract 37 DRAFT PROPOSAL FOR AMENDMENTS Blood and Blood-related Products Human Albumin. Page 2568 Abnormal toxicity. Line 2 Change from: using Method B to: for antisera and vaccines. Vaccines and Immunosera for Human Use Pneumococcal Polysaccharide Vaccine. Page 2424 Abnormal toxicity (2.2.1): Delete the requirement Veterinary Preparations. Page 2621 General Requirements Insert after Para 1 The active pharmaceutical ingredients (drug substances), excipients (pharmaceutical aids), pharmaceutical preparations (dosage forms) and other articles described in the monographs are intended for human and veterinary use (unless explicitly restricted to one of these users). 38