Case Study 2, Heartworm Experimental Design
advertisement

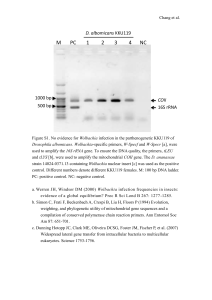
CASE STUDY: The Caruso Chronicles Part I: Caruso had not been to the veterinary clinic since Kelly and Mike found him a year ago as a puppy. The veterinarian estimated that Caruso was about two-months old. At that time he had all his shots, blood drawn and sent away to a laboratory. Caruso received a clean bill of health. “Mike, something is wrong with Caruso. Caruso’s ears perked up when he heard his name and Kelly placed her hands on each side of his face, reached her fingers behind his ears and gave him a comforting scratch behind his ears. “He’s so tired acting. You know how energetic he usually is. He just seems like he’s been getting more and more tired when he plays. Let’s take him to the clinic.” At the Clinic Dr. Veter: Hi Kelly, what’s going on with Caruso? Kelly: Caruso just doesn’t seem to be himself. For the past couple of months he’s more tired than usual” replied Kelly. Dr. Veter: Let’s draw some blood and do a couple of tests. Has Caruso been on heartworm preventative medication? Mike: No. Dr. Veter: I’d like to do a heartworm test. It won’t take very long, only 10 minutes or so. Kelly: Okay, but how does the test work? Dr. Veter: We’ll take some blood from Caruso. If Caruso is infected with the nematode that causes heartworm, a protein that circulates in the blood from the nematode will be detected and the test will turn up positive. The test is very specific and reliable. The vet took blood samples and performed a heartworm test. Dr. Veter: Good news, Caruso’s heartworm test was negative. We’ll wait to see what the blood work tests tell us. We’ll have the results in just a couple of days. We’ll give you a call. The blood samples were sent to an outside lab for analysis. A Knott test was also requested. A Knott test detects the microfilarial larval stage of D. immitis using microscopy and cell staining techniques. Dr. Veter also knew that Wolbachia, an endosymbiotic group of bacteria, were essential to the reproduction and survival of D. immitis. Wolbachia and D. immitis have a symbiotic relationship, but Wolbachia have also been implicated in severe immune responses in animals. When dead worms release Wolbachia into the blood stream the immune system goes into overdrive and has been implicated in the formation of blood clots in the lungs specifically, causing more problems, and even death in the animal. Student Questions: 1. Wolbachia are obligate endosymbionts. What does this mean? 2. What do you think would happen if an animal with a D. immitis infection was treated with an antibiotic? Part II: One week later……. Dr. Veter received Caruso’s blood work and to his surprise PCR results were also sent by the laboratory. Patient: Caruso TEST Albumin (g/L) Eosinophils Total # x 103/uL Globulin (Total Blood) Hematocrit Percent Packed Cell Volume (%PCV) Heartworm (ELISA) Knott Test Normal Range 2.7-4.4 At 2 Months 12 Months Later 3.2 2.7 0-1200 1.6-3.6 800 2.8 1300 3.8 36-60 Antigen Test for Adult Female Nematode Protein 52 Not performed 38 Negative 0 microfilaria/mL Not performed 160 microfilaria/mL Student Questions: 1. Why didn’t the veterinarian test Caruso for heartworm as a two month old puppy? 2. Hypothesize how Caruso could have a positive Knott test but have a negative heart worm test at twelve months? 3. Dr. Veter specifically asked for eosinophil and globulin levels from Caruso’s blood sample. What is the significance of this request? Optional Extension Questions: 4. The heartworm test is an application of ELISA, Enzyme-Linked Immunosorbent Assay. ELISA is a fundamental tool used in clinical immunology. How does this technique work? 5. What other diseases has ELISA been used for in the detection of infection in humans? Are there other non-disease ELISA applications? Part III: The PCR test results for Caruso were provided and analyzed by Dr. Veter. It had been a long time since he had to think about this type of test. PCR Test Results 1 2 3 4 5 6 550bp 102bp Figure 1. Detection of Wolbachia pipientis DNA by PCR Using primers against ftsZ gene fragment (550 bp) and D. immitis 18S ribosomal DNA gene fragment (102bp) Lane 1, 100 base pair ladder, Lanes 2-3 negative controls; Lane 4-5 positive control; Lane 6, Caruso’s Blood Sample. Student Questions: 1. Lane 1 contains a 100bp ladder. What purpose does the ladder serve? 2. In Lanes 2 and 3, what is the purpose of each negative control? 3. In Lane 4, two gene fragment templates were used for the positive control. One gene fragment is the 18S ribosomal DNA from D. immitis. The second is the ftsZ gene fragment from Wolbachia (WO), which plays a role in controlling WO cell cycle. a. Why was the 18S ribosomal DNA gene fragment from D. immitis used in the positive control? b. Why would a second positive control (Lane 5) be important? 4. Is Caruso, negative or positive for Wolbachia DNA? How do you know? 5. What diagnostic inference can be made about Caruso’s heartworm status? Why and is this inference supported by the blood work results? Part IV: Dr. Veter: Hi Kelly. The blood work is telling us that Caruso does have a heartworm infection. Let’s get him in here and talk about a treatment plan. Fortunately for Caruso, we’ve caught it early. In preparation for Caruso’s visit, Dr. Veter needed to decide the best course of treatment for the dog. D. immitis is not easy to treat because the nematode also harbors the endosymbiotic bacterium Wolbachia. She reviewed a recent study about heartworm treatment (Bazzocchi et al., 2008). Doxycycline (DOXY) is an antibiotic in the tetracycline family of antibiotics. Ivermectin (IVM) is an anti-parasite medication. Here is an excerpt from the article*. “A total of 20 young adult beagles were used. The animals were born and raised in a mosquito-proof environment and did not have any prior exposure to natural infection with D. immitis. Seven male and nine female adult heartworms harvested from dogs approximately 8 months post infection were introduced by intravenous transplantation (via a jugular vein) into each dog (McCall et al., 2008). Approximately 6 weeks later (Day 0), the dogs were randomly allocated to four groups of five dogs each. Beginning on Day 0, the dogs in one group were given weekly prophylactic doses of IVM (6ug/kg) orally for 34 weeks. Weekly treatment was chosen to evaluate if previously reported adulticide effect following monthly treatment could be achieved in a shorter period of time. The dogs in another group were given DOXY (10mg/kg/day) orally from Weeks 0-6, 10-12, 16-18, 22-26, and 28-34. The dogs in the third group were given a combination of IVM and DOXY at the same dosages and treatment schedules as used for the first and second groups, respectively. The remaining group served as the non-treated (or infection ) group.” Student Questions: 1. Describe the experimental design of this study. Is there anything missing or more information you need to know about? 2. Based on the above information, is this study a controlled experiment? Explain why or why not? 3. Formulate a research question based on the information. The table below shows some of the data collected from the study. 4. Why are the researchers testing the effectiveness of an antibiotic? 5. In Table 1, the researchers present range values. What is range statistically? Have the data been reported correctly? 6. What does (+) SD mean? 7. In the graph below, some of the data points have bars on them. According to the authors, what do these bars represent and what do they mean? 8. What does 95% Confidence limits represent? 9. What does percent efficacy mean? 10. Formulate a null hypothesis for this experiment. 11. What is the significance of a P value and what does P< 0.001 represent at the bottom left of the table? 12. Using information provided in the table and the graph below, are there differences between the treatment groups? Explain using the statistical information presented and percent efficacy. Is your null hypothesis supported or rejected? The graph below represents data collected over 36 weeks of treatment. Figure 1. Mean microfilarial counts (+ SDs) obtained over a 36 week period. Synthesis Question: 13. Based on the graph and Table 1, which treatment would be most effective against D. immitis? Explain. *Information presented based on the research study from Bazzocchi et al., 2008 has been modified to meet the objectives of the case study. To review the research study in full and without changes refer to literature citation section of the case study. Answer Key Part I Answers to Questions Q1: Wolbachia are obligate endosymbionts. What does this mean? Endosymbionts are organisms that live in the body or cells of another organism. An obligate endosymbiont cannot survive without the host. Wolbachia pipientis cannot survive without the host, D. immitis, in this case. Q2: D. immitis is a eukaryotic organism so antibiotics will not work on a eukaryotic organism. Wolbachia is a prokaryotic organism. Antibiotics specifically target bacterial cells. Because D. immitis has a symbiotic relationship with WO and requires WO for reproduction and survival, killing the symbiont WO would also likely impact D. immitis and interrupt its life cycle. Part II Answers to Questions Q1: Students will need to apply their understanding of the nematode life cycle and knowledge of what the heartworm test tests for (female protein antigen) to arrive at the conclusion that there would be no adult worm protein made at that young age to have an antigen reaction. Q2: Low HW burden (one compared to four females for example) and enough antigen has not accumulated. Students might also capitalized on sensitivity of the test. The level of antigen is directly related to the number of females present; the microfilaria were passed from mother to puppy transplacentally but did not have adult D. immitis at that time. Q3: Caruso had high levels of eosinophils, which are specialized white blood cells of the immune system and indicate a parasitic infection. High globulin levels indicate an immune system response to infection and production of antibodies. The host immune system will attempt to eliminate the parasite producing antibody globulins and at the same time increasing other immune system white blood cells such as eosinophils that target parasitic infectious agents. Q4: There are two forms of ELISA to detect antigens: direct and indirect ELISA assays. ELISA is used in research and applied settings. Probably some of the most familiar tests include the human HIV and hepatitis tests, but ELISA kits are used for a variety of pathogen infection testing along with autoimmune, cancer, allergy and food toxin testing. Q5: What other diseases has ELISA been used for in the detection of infection in humans? Are there other non-disease ELISA applications? See this web site for many different tests currently in use and approved by the FDA http://www.rapidtest.com/index.php?product=Parasitology-ELISA-kits&cat=17 Part III Answers to Questions Q1: Lane 1 contains a 100bp ladder. What purpose does the ladder serve? A DNA ladder is used for fragment size determination. Q2: In Lanes 2 and 3, what is the purpose of the negative control? Show a result with no WO present. The water control is essential for detecting contamination or non-specific amplification of your reaction. The second negative control, confirms a negative result in a dog not infected by D.immitis and therefore, no WO should be detected. Q3: In Lane 4, two gene fragment templates were used for the positive control. One gene fragment is the mitochondrial cytochrome oxidase 1 (CO1) gene from D. immitis. The second is the ftsZ gene fragment from Wolbachia (WO) which plays a role in regulating WO cell division. a. Why was the 18S ribosomal gene fragment from D. immitis used in the positive control? If DNA isolation is successful they will get a band for 18S ribosomal DNA even if there is no WO gene fragment. This important to know that the DNA extraction was successful and that WO is actually being amplified. Perhaps the WO ftsZ gene amplicon was not detected. This would alert the researcher that there is something wrong with the WO ftsZ gene amplification process (i.e. primer specificity) because if D. immitis is present, then WO should also be detectable. Based on the paper from Rossi et. al (2010) PCR sensitivity is extremely high in the context of WO DNA ftsZ gene detection. b. Why would a second positive control (Lane 5) be important? Shows conformation of WO DNA and therefore D. immitis infection in an infected dog. Q4: Is Caruso, negative or positive for Wolbachia DNA? How do you know? Positive. Caruso’s blood DNA sample showed a 550bp fragment identifying the WO ftsZ gene fragment. Q5: What diagnostic inference can be made about Caruso’s heartworm status from the PCR results? Why and can this inference supported by the blood work results? Caruso has heartworm because WO is symbiotic with D. immitis. The results from the blood work, the Knott’s test specifically through identification of D. immitis microfilariae, also support D. immitis infection. Part IV Answers to Questions Q1: Describe the experimental design of this study. Is there anything missing or more information you need to know about? The experimental design does not identify the gender of the animals, nor does it identify if there were differences in nematode counts before group assignment. Q2: Based on the above information, is this study a controlled experiment? Explain why or why not? Yes, this is a controlled study. The researchers included a group not receiving any treatment. The control group offers a comparison group for the researchers. Q3: Formulate a research question based on the information. This may become an important discussion point with students because the teacher can determine if students are distinguishing the difference between a question and a hypothesis. It can also be a springboard for discussion about the importance of having a question that can be tested scientifically. Q4: Why are the researchers testing the effectiveness of an antibiotic? D. immitis shares a symbiotic relationship with the bacterium Wolbachia pipientis. Killing the bacterium may have an adverse effect on D. immitis. There is a secondary implication for the health of the dog that addresses the immune response to the release of WO as noted above. Q5: In Table 1, the researchers present range values. What is range statistically? Have the data been reported correctly? Range represents how far apart the two extremes are in a data set. Range represents the difference between the minimum and maximum values and is a single number not a ‘range’. The range has been incorrectly reported. Q6: What does SD (+) mean? SD stands for standard deviation. Standard deviation is another way to represent spread of data and specifically represents how far away data values are from the mean. SD therefore represents variability. The larger the standard deviation the greater the difference from the mean, the smaller the SD the more tightly grouped the data is to the mean. Q7: Some of the data points have bars on them. What do these bars represent? In this study, as reported by the authors they represent standard deviation. SD represents how far from the mean the data point deviates. This information may be of limited use because there can be overlap of the bars and yet there may still be significant differences between the data points. SD bars wouldn’t necessarily reflect that but does show how the data varies within the group. Q8: What does 95% Confidence limits represent? This tells us how close our estimate of any sample will be to the mean. 95% of any given sample population will be covered within 2 standard deviations and would fall between the upper and lower limits identified in the paper. Q9: What does percent efficacy mean? How effective the particular treatment was at reducing adult worms. Q10: Formulate a null hypothesis for this experiment. Ho There will be no difference in reduction of adult worms between the control group and treatment type. Q11: What is the significance of a P value and what does P< 0.001 represent at the bottom left of the table? P value tells you how much statistical evidence there is to reject the notion that the differences between groups is due to chance alone. A typical level of less than 0.05 says that there is less than a 5% chance that the difference between groups is due to chance alone. Another way to look at it is that there is a 95% chance that the difference between groups is real. A P value of less than 0.001 means the statistical chance that the difference between groups is random, or due to chance, is 0.1%. Stated another way there is a 99.9% chance the difference between groups is real and not because of chance. Q12: Using information provided in the table, are there differences between the treatment groups? Explain using the statistical information presented and percent efficacy. There was not statistical difference between the IVM or DOXY treatments alone with regard to the mean number of worms recovered compared to the control group. The IVM/DOXY combination resulted in the greatest adulticidal effect at 78.26%. Based on the P value of 0.001, this is a statistically significant difference in worm reduction in the IVM/DOXY treatment group compared to the control group. Synthesis Question: Q13: Based on the Graph and Table 1, which treatment would be most effective against D. immitis? Explain. What other statistical information would you like to have to strengthen your claim particularly with respect to the graph? IVM/DOXY combination was most effective at reducing the number of adult worms and microfilaria over the 36 weeks of treatment. The control group had the highest microfilarial/mL throughout the 36 weeks compared to the IVM/DOXY and even the other two treatment groups. A P value for the data presented would help support this claim with respect to the decreases in microfilaria (Figure 1). The P value of less than 0.001 shows that there is statistical difference between the number of adult worms after the IVM/DOXY treatment compared to the control and rejects the null hypothesis. (It should be noted that in the original article the authors give a P value of less than 0.05 for control versus IVM/DOXY data presented in the graph.) Teacher Notes and Background Resources PART I: Concepts for Focus: Life cycles, prokaryotic and eukaryotic cells Student Learning Objectives: Students will apply their understanding of nematode life cycle to parasitic infections. Students will formulate hypotheses using their understanding of nematode life cycle and symbiotic relationship with Wolbachia. Background Resources Microbial Life http://serc.carleton.edu/microbelife/index.html Heartworm background http://www.metapathogen.com/heartworm/ http://www.merckvetmanual.com/mvm/index.jsp?cfile=htm/bc/11300.htm Heartworm testing info http://www.marvistavet.com/html/body_diagnosis_of_heartworm_disease.html Video of HW infection http://www.heartwormsociety.org/pet-owner-resources/canine.html Heartworm map http://www.veterinarypracticenews.com/vet-breaking-news/2011/05/13/heartworm-diagnosed-inevery-state-in-10-survey-finds.aspx Microbe wiki http://microbewiki.kenyon.edu/index.php/Dirofilaria_immitis Wolbachia Web site https://www.wolbachiawebsite.org/about.cfm Wolbachia Project http://discover.mbl.edu/index.html Werren, J. H. (1997). Biology of Wolbachia. Annual Review of Entomology, 42, 587-609. Part II: Concepts for Focus Nematode life cycle, immunology methods Student Learning Objectives Students will explain the application of clinical immunological methods to pathogen detection. Students will distinguish the difference between prokaryotic and eukaryotic cells. Background Resources Heartworm micofilaria measurement and blood testing http://www.heartwormsociety.org/veterinary-resources/canine-guidelines.html#3 http://www.drkaslow.com/html/proteins_-_albumin__globulins_.html ELISA Tutorials http://www.sumanasinc.com/webcontent/animations/content/ELISA.html http://www.biology.arizona.edu/immunology/activities/elisa/main.html Examples of other ELISA kits and uses http://www.rapidtest.com/index.php?product=Parasitology-ELISA-kits&cat=17 Bazzocchi, C., Genchi, C., Paltrinieri, S., Lecchi, C., Mortarino, M., & Bandi, C. (2003). Immunological role of the endosymbionts of Dirofilaria immitis: the Wolbachia surface protein activates canine neutrophils with production of IL-8. Veterinary Parasitology, 117(1–2), 73-83. Part III: Concepts for Focus Polymerase chain reaction, biotechnology, data interpretation Student Learning Objectives Students will explain the function of controls in a PCR test. Students will interpret and make inferences from PCR results. Background Resources PCR Virtual Lab http://learn.genetics.utah.edu/content/labs/pcr/ Biology Animation: PCR http://www.dnalc.org/resources/animations/pcr.html NCBI PCR http://www.ncbi.nlm.nih.gov/projects/genome/probe/doc/TechPCR.shtml PCR Video Lecture http://www.youtube.com/watch?v=_YgXcJ4n-kQ http://www.youtube.com/watch?v=OK7_ReXhVaQ Part IV: Concepts for Focus Data interpretation, hypothesis formation, descriptive statistics Student Learning Objectives Student will apply their understanding of experimental design. Students will use their understanding of experimental design to explain a controlled study. Students will use statistics to analyze data to arrive at conclusions. Students will formulate hypotheses and use statistics to reject or accept a hypothesis. Background Resources Basics of Standard Deviation http://www.mathsisfun.com/data/standard-deviation.html Mean, Median, and Mode Review http://www.mathsisfun.com/mean.html Scientific Process http://undsci.berkeley.edu/index.php Research Methods Knowledge Base http://www.socialresearchmethods.net/kb/contents.php Literature Citations Bazzocchi, C., Mortarino, M., Grandi, G., Kramer, L. H., Genchi, C., Bandi, C., …McCall, J. W. (2008). Combined ivermectin and doxycycline treatment has microfilaricidal and adulticidal activity against Dirofilaria immitis in experimentally infected dogs. International Journal for Parasitology, 38(12), 14011410. McCall, J. W., Genchi, C., Kramer, L., Guerrero, J., Dzimianski, M. T., Supakorndej, P., . . . Carson, B. (2008). Heartworm and Wolbachia: therapeutic implications. Veterinary Parasitology, 158, 204-214. Rossi, M. I. D., Aguiar-Alves, F., Santos, S., Paiva, J., Bendas, A., Fernandes, O., & Labarthe, N. (2010). Detection of Wolbachia DNA in blood from dogs infected with Dirofilaria immitis. Experimental Parasitology, 126(2), 270-272.