IMMUNISATION OF PREGNANT WOMEN AGAINST INFLUENZA A
advertisement

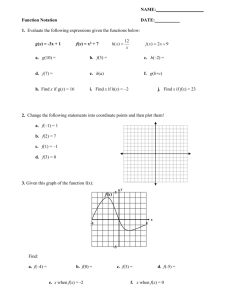
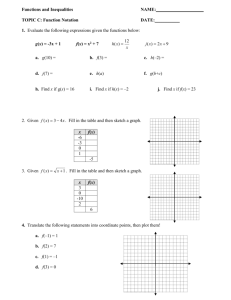
IMMUNISATION OF PREGNANT WOMEN AGAINST INFLUENZA A H1N1(v) INTRODUCTION The purpose of this paper is to brief NHS Board Operational leads on the need for further action to ensure all pregnant women have been offered the opportunity of vaccination against influenza A H1N1v before 31st December 2009. As indicated in the CMO letter SGHD/CMO/CNO (2009)15, Health Protection Scotland has been instructed by the Scottish Government to work with NHS Boards to ensure that this is achieved. RISK TO PREGNANT WOMEN FROM INFLUENZA A H1N1(v) INFECTION 1. WHO1 recently held a seminar to gather information on the experiences of 100 professionals who had managed cases of the infection. Although the overwhelming majority of those infected with the new virus have uncomplicated influenza-like illness, concern was expressed about small subsets of patients who rapidly develop very severe progressive pneumonia often associated with failure of other organs. The participants agreed that the risk of severe or fatal illness is highest in three groups: pregnant women, especially during the third trimester of pregnancy, children younger than 2 years of age, and people with chronic lung disease, including asthma. There is as yet no confirmed case of a mother infected with influenza A H1N1v passing the virus onto her unborn child. 2. Early reports from the US2 indicated that pregnant women were more than four times more likely to be admitted to hospital due to influenza A H1N1v infection than the general population. In Australia3, about 5% of hospitalised confirmed H1N1 cases have been reported as pregnant. In the UK, the ongoing review4 of hospitalised cases of laboratory confirmed pandemic influenza AH1N1v to help guide clinical management, has identified 29 (19.6%) of 148 female cases aged 16-45 years admitted to hospital who were pregnant at the time of admission. The UK Obstetric Surveillance System5 reports that based on data received up to 12 November, 18% of pregnant women admitted to hospital with influenza A H1N1v infection had had an ITU admission. 3. In Scotland, of the 1280 confirmed hospitalised cases of influenza A H1N1v infection reported to HPS by December 1st 2009, 48 (3.8%) were pregnant at the time of admission, 8 of whom had other co-morbidities recognised as risk factors for the disease. Over 50% of these pregnant women have been aged less than 25 years. Of the 48, at least 5 received intensive care. Of the 52 deaths in confirmed cases, 3 (5.8%) were pregnant at the time of admission to hospital. 4. Indicators for rates for influenza like illness in Scotland have remained relatively stable over the last few weeks. Swab-positivity rates for influenza A H1N1v infection in those presenting to GPs with symptoms have shown a similar picture. Rates of transmission of the virus may therefore be at or near to their 1 peak. However when they start falling, it is likely to take some weeks before they cease to be a significant public health problem. With the winter starting, the incidence of other respiratory pathogens (including bacterial) is likely to increase which may lead to a worsening of the clinical spectrum. 5. Because of these factors, the influenza A H1N1v virus will continue to present a risk to the health of pregnant women over the coming weeks. They remain a priority for vaccination. 6. So far many thousands of women have been vaccinated with Pandemrix in the UK. The MHRA has implemented a pharmaco-vigilance system. Up to and including Thursday 29th October 2009, MHRA6 has received a total of 50 UK reports of suspected adverse reactions to the H1N1 vaccines in pregnant women. The most commonly reported are injection site reactions and other well established adverse effects of many vaccines. The MHRA continues to conclude that the benefit – risk balance for Pandemrix remains positive. IMMUNISING PREGNANT WOMEN: CURRENT SITUATION 7. The estimated cumulative uptake rate among pregnant women as at week ending 30 November is 28.5 %. These are preliminary figures only and are derived from weekly compilation of information from 585 (57%) General Practices where H1N1 vaccination uptake data was submitted and are continuing to be scrutinised. Other information sought directly from individual practices, for example in Borders on 1 December, indicates much higher uptakes are being achieved by some practices usually over 50%. It is of note that currently the number of women coded as pregnant by the practices is about half that of the expected number of pregnant women. Steps are being taken to support and inform practices around the identification and recording of pregnancy status. 8. HPS has worked with NHS Boards to provide support and review progress in the immunisation of pregnant women. NHS Boards have shown commitment to achieving optimum uptake and have undertaken a number of important actions to ensure that pregnant women are encouraged to come forward for vaccination. Some of these actions are listed in Appendix 1 “Good Practice Identified from NHS Boards in Scotland in Maximising Uptake in Pregnant Women”. 9. There is considerable evidence of good practice in almost all Board areas, and we would advise that we seek to extend this across all so as to contribute to the maximisation of uptake in pregnant women. 10. We are aware that 7 Boards have developed a central register of pregnant women and have organised the sending of a letter to each one identified either inviting them for immunisation or informing that they will soon receive a letter from their general practitioner doing so. Others have worked with local practices to invite women. 11. Further information has emerged from Northern Ireland on practice that has contributed to increasing uptake rates. This information is contained in Appendix 2. The information for professionals in use in Northern Ireland comes with this email. 2 FURTHER ACTIONS TO ENHANCE UPTAKE 13. There is clearly scope for further action to ensure that the uptake of vaccination in pregnant women is maximised. National Actions 14. HPS is working with partner national agencies to: Revise and improve the coding of pregnant women by General Practices; Implement software to continue to extract uptake rates. 15. The Scottish Government with other UK Health Departments has taken forward a media campaign to highlight the need for, and benefits of, immunisation for At Risk groups including pregnant women. This will start today and feature over the next two weeks. The Swine Flu vaccination information leaflet for pregnant women comes with this email. A Word version of the same leaflet also comes for incorporation into any invitation letters if necessary. Further printed copies can also be ordered from the Scottish Government. Board Actions 16. Boards are asked to : a) review their activity to date, and to consider implementation of good practice where this is not already in place or completed, including close liaison with midwifery leads/obstetrics staff; b) if not already done, should ensure that all GP practices are provided with complete lists of pregnant women receiving active antenatal care. Appropriate information governance processes should be followed. Boards should also ensure that robust arrangements are in place to notify practices of newly booked pregnancies. This will allow practices to ensure that all registered pregnant women have been completely identified. When these become available, lists should be used as a final check by practices to ensure that no women have been omitted. c) by 10th December 2009, ensure that all pregnant women resident in the Board area have been invited for vaccination. If this has not already been completed in your area, you are asked to centrally organise the sending of a letter to all pregnant women reminding them of the offer of the vaccine via their registered GP. This should be done without delay. Boards which have not already done so, should also encourage practices to invite for vaccination those who have not already been vaccinated. Appendix 3 gives further details on how this might be approached by Boards, including a sample process and letter used by NHS Lanarkshire. An alternative form of words for a letter, based on that used in Northern Ireland, is provided. d) after 10th December 2009 and before the end of December 2009, liaise with General Practices to encourage any pregnant woman who has been invited and 3 has not directly refused the vaccine, but has not been vaccinated, to come forward for immunisation or be offered the vaccine opportunistically; e) liaise with midwifery leads/obstetrics staff to ensure that any pregnant woman who has not directly refused the vaccine but has not been vaccinated, is advised of the offer of vaccination. f) give a status update at the Board Operational Leads meeting on 9th December. CONCLUSION For the coming weeks influenza A H1N1 infection will continue to present a risk to pregnant women in Scotland. All pertinent national and local actions should be taken to protect them. This briefing is designed to ensure a consistent approach across the country. REFERENCES 1. World Health Organisation, “Clinical management of human infection with pandemic (H1N1) 2009: revised guidance”, WHO, November 2009 2. Jamieson et al.., “H1N1 2009 Influenza Virus Infection during Pregnancy in the USA”, Lancet 374:451-8, 2009 3. Australian Government, Department of Health and Ageing “Australian Influenza Surveillance Summary Report 2009 - Period- 5 to 11 September 2009”, Australian Government. 4. (Pandemic) Influenza Clinical Information Network - FLU-CIN, “Summary Report No 6 Date: 20 /11 /2009 (based on data available to 18/11/2009 – 600 cases)”, Presentation, November 2009 5. UK Obstetric Surveillance System, “H1N1 in Pregnancy Interim Report No.1Women admitted to hospital with laboratory confirmed H1N1v infection Date: 13/11/09 (based on data available to 12/11/09)”, Royal College of Obstetricians and Gynaecologists and National Perinatal Epidemiology Unit, Presentation November 2009. 6. Medicines and Healthcare Products Regulatory Agency “UK Suspected Adverse Reaction Analysis - Swine Flu (H1N1) Vaccines 5 November 2009” MHRA, November 2009. Martin Donaghy Medical Director Jill Carson Service Planning Manager 4 APPENDIX 1 Good Practice Identified from NHS Boards in Scotland in Maximising Uptake in Pregnant Women Good Practice…… Engaging directly with midwives and Obstetrics/Gynaecology staff. Carried out by….. Borders, Dumfries & Galloway, Western Isles. GPs phoning pregnant women directly to invite for vaccination. Some practices within Borders. Occupational Health Consultant-led clinics for pregnant health and social care workers. GGC. Support and encouragement to midwives from Obs/Gynae Consultants. Borders, GGC. Active involvement of midwifery lead in planning structures for vaccination. Various Boards including Borders, GGC, Tayside, Western Isles. Midwife-specific training sessions. Lanarkshire, Dumfries & Galloway. Letters and information sent out to all pregnant women. Various boards including Lanarkshire, Lothian. Specific arrangements for those with high-risk pregnancies. Lothian. Weekly H1N1 vaccination briefing cascaded to maternity services. Borders Jill Carson Alison Smith-Palmer HPS 1st December 2009 Version 1 5 APPENDIX 2 INFLUENZA A H1N1 IMMUNISATION IN PREGNANT WOMEN IN NORTHERN IRELAND (NI) Uptake rates in pregnant women were discussed with colleagues from the Northern Ireland Health Protection Agency. Uptake Rates So far the average uptake rate for NI is 52%. The programme is organised through the 5 Acute Trusts operating in NI and the rates range from 40 to 65%. The denominator for the uptake rate is all women booked with the Trusts for antenatal care. It therefore does not include all pregnant women. NI has a register of pregnant women (NIMAS) but this is only employed in a minority of the Trusts. It is not essential to either the call of women or the recording of immunisation status or the calculation of uptake. Indeed the Trust with the best uptake rates does not use NIMAS. The differential in uptake rates partly reflects the varying start dates of the Trust programmes. The Programme The NI Government and NI HPA decided after extensive consultation to run the immunisation of pregnant women through the Trusts rather than GPs. The decision was described as being 50:50. Pregnant women are identified either through NIMAS or the routine antenatal booking system. Special clinics in the hospitals were set up and run by midwives, many at evenings and weekends. A special invitation letter was devised which gave a number to book an appointment. A specific information sheet on immunisation and pregnancy was developed which was either sent with the letter or made available at the clinic prior to immunisation. The Trust which made the sheet available on attendance has the highest uptake rate. One Trust also sent out the GSK Patient information leaflet and this has the lowest uptake leading to a preliminary conclusion that this may have backfired. Immunisation is now being offered opportunistically at routine antenatal clinics in most Trusts. Reasons for the relatively high uptake rates Colleagues from the Northern Ireland Health Protection Agency are of the view that the main reason was the early engagement of midwives and their buy-in to the programme. The NI HPA allocated a public health Specialist Registrar and a nurse to develop this aspect of the pregnancy programme. Specific sessions to train midwives as either immunisers and/or counsellors were arranged. Key opinion formers in the profession were targeted and brought on board and served to reassure their colleagues. Obstetricians were not involved in a major way. Martin Donaghy Medical Director Health Protection Scotland November 2009 6 APPENDIX 3 EXAMPLE OF PROCESS FOR CENTRAL ORGANISATION FOR INVITING PREGNANT WOMEN Obtain lists of pregnant women from midwifery service/s. Mail aggregated lists to individual practices for reconciliation with practice lists. Mail all pregnant women directly from the Board. Ensure that there are robust arrangements in place to notify practices of newly booked pregnant women. NHS Lanarkshire Process vaccination of pregnant women began with availability of vaccine in GP and OH around end of Oct. The invite letter is the second phase. we do not have centralised electronic records for pregnant women. Midwives just had paper records but they had labels and CHI # (We considered using electronic laboratory booking bloods data, but data quality not considered good enough) each midwife collated all their pt's labels with CHI #, ~5-10% did not have CHI, in which case they hand wrote demographic details admin persons entered ~3,000+ CHI numbers on to a spreadsheet with forename (as a check) and code for their area of care (I think it took several bank aid staff over about 4 days). They looked up CHI for those with just demographics. the spreadsheet was merged with CHI database to add demographics electronically and checked with forename for accuracy (a small % were discarded for lack of accuracy) then the electronic CHI demographic data was used as mail merge ideally we would have wished for a double check of pregnancy status before letters issued, but risk balance favoured early distribution given the consequences of H1N1 illness in pregnancy mail merge letters auto stuffed with info leaflets in windowed envelopes with private and confidential on it (NB no labels used to omit a further potential source of error) 3,060 letters to be posted (over next few days) key times were getting all midwives to collate all data, wait for printed leaflets, then the envelope stuffer needed calibrated (manager for retinal screening off unexpectedly) and letters posted. midwives now issuing letters at booking. 7 SAMPLE NHS LANARKSHIRE TEXT FOR LETTER TO PREGNANT WOMEN Dear <first name> <surname> Invitation for H1N1/Swine flu vaccination I am writing to recommend vaccination against the H1N1/swine flu virus for all pregnant women and invite you to contact your GP surgery to make an appointment to be vaccinated. Swine flu is a respiratory disease caused by a new strain of the influenza virus. Swine flu is easily spread by coughing or sneezing and causes marked fever, tiredness, cough and sore throat. Some people are more likely than others to become very unwell with swine flu and pregnant women have been particularly severely affected. These effects include being more likely to need hospital treatment. The vaccine against the virus is available from your GP surgery and is a very good way of protecting you and your baby from the virus. Most people should only require a single dose, but depending on your personal circumstances, you may need two doses. Your GP will tell you whether you need to make a follow-up appointment. For more information about the vaccine, please see the attached leaflet or call the NHS 24 vaccination information line on 08000 282 816. If you are no longer pregnant, or have already made an appointment at the surgery, or you have received your swine flu vaccination already, please accept my sincere apologies and ignore this letter. 8 ALTERNATIVE WORDING FOR BOARD LETTER BASED ON LETTER USED IN NORTHERN IRELAND Dear SWINE FLU VACCINATION for PREGNANT WOMEN We would like to invite you to be vaccinated against swine flu, if you have not already received it. All pregnant women in Scotland are being offered a swine flu vaccination. It may be that you have been contacted before by your general practice, or have been advised by your midwife, or antenatal clinic to receive this vaccine. If you are still undecided, we would ask you now to reconsider this important invitation. Why are pregnant women being vaccinated? While swine flu is a mild illness for most people, it can be very serious. Pregnant women who catch this flu are much more likely to develop serious complications, including pneumonia and heart and lung problems, compared to women who are not pregnant. They are more likely to become so ill that they need to be admitted to hospital and they are ten times more likely to need intensive care compared to the general population. Sadly, some pregnant women have died as a result of swine flu infection. The vaccine can protect you and your unborn baby from the effects of swine flu. Is the vaccine safe for me and my baby? The swine flu vaccine is a new vaccine because swine flu is a new virus. It has undergone tests and has been certified as safe for use in pregnant women at all stages of pregnancy. The swine flu vaccine is similar to the seasonal flu vaccine which has been given safely to millions of women across the world. Studies looking at the effects of giving pregnant women the seasonal flu vaccination found that it caused no bad effects on pregnant women or their babies. The swine flu vaccine does not contain live virus, and cannot give you or your baby the flu. How do I get this vaccination? If you wish to accept the vaccine, we urge you not to delay further and to contact your general practice to receive it. Please find enclosed a patient information leaflet on the swine flu vaccination. Further information is available on the NHS 24 website: www.nhs24.com or its Flu Helpline: 08000 282 816. If you are no longer pregnant, or have already made an appointment at your general practice, or you have received your swine flu vaccination already, please accept my sincere apologies and ignore this letter. Yours sincerely Dr XXXXXX XXXXXX 3xx December 2009 9 ALTERNATIVE WORDING FOR BOARD LETTER BASED ON LETTER USED IN NORTHERN IRELAND Dear SWINE FLU VACCINATION for PREGNANT WOMEN We would like to invite you to be vaccinated against swine flu. All pregnant women in Scotland are being offered a swine flu vaccination. It may be that you have been contacted before by your general practice, or have been advised by your midwife, or antenatal clinic to receive this vaccine. If you are still undecided, we would ask you now to reconsider this important invitation. Why are pregnant women being vaccinated? While swine flu is a mild illness for most people, it can be very serious. Pregnant women who catch this flu are much more likely to develop serious complications, including pneumonia and heart and lung problems, compared to women who are not pregnant. They are more likely to become so ill that they need to be admitted to hospital and they are ten times more likely to need intensive care compared to the general population. Sadly, some pregnant women have died as a result of swine flu infection. The vaccine can protect you and your unborn baby from the effects of swine flu. Is the vaccine safe for me and my baby? The swine flu vaccine is a new vaccine because swine flu is a new virus. It has undergone tests and has been certified as safe for use in pregnant women at all stages of pregnancy. The swine flu vaccine is similar to the seasonal flu vaccine which has been given safely to millions of women across the world. Studies looking at the effects of giving pregnant women the seasonal flu vaccination found that it caused no bad effects on pregnant women or their babies. The swine flu vaccine does not contain live virus, and cannot give you or your baby the flu. How do I get this vaccination? If you wish to accept the vaccine, we urge you not to delay further and to contact your general practice to receive it. Please find enclosed a patient information leaflet on the swine flu vaccination. Further information is available on the NHS 24 website: www.nhs24.com or its Flu Helpline: 08000 282 816. Yours sincerely Dr XXXXXX XXXXXX 3rd December 2009 10