Cattle Physical Examination Laboratory Manual
advertisement

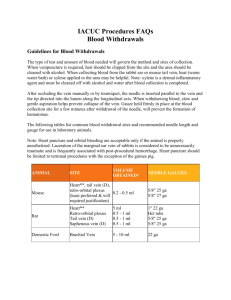
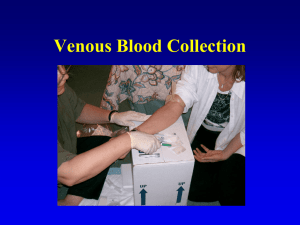
VMED 511, 512, 513, 514 Foundations in Veterinary Medicine Laboratory Manual Cattle Laboratory: Physical Examination, Diagnostic Sampling, and Treatment Techniques Station 1: Physical Examination of Cattle Systematic/Anatomic Physical Examination Physical examination in cattle should be performed when an individual animal requires treatment or when examination of a representative sample of a herd is indicated in the evaluation of a herd problem. A personal system of performing a complete physical examination should be developed to insure that important information is not omitted. The following description of a physical examination is the system utilized by Dr. Hostetler. Other clinicians may have a different system or approach to the physical examination and should be considered when you develop your systematic physical exam. By performing a systematic examination recall of normal and abnormal findings also becomes easier. Initial Observation Initially, the animal in question is observed from a distance to assess demeanor, level of alertness, and responsiveness. Additional information regarding proprioception, strength, and the presence of lameness is also gained by observation from a distance. Head and neck position in cases of suspected airway disease is best observed outside of a restraint chute. Animals in respiratory distress may collapse with further restraint. Frequently animals that are depressed will only show characteristic head and ear position when not restrained. The udder is observed to assess symmetry between fore and rear quarters and left and right quarters. Physical Examination Following restraint in a chute or on a halter, the physical examination can proceed. Initially, data points that are most affected by changes in sympathetic tone should be evaluated to avoid erroneous values. These points include collection of urine, rumination rate, heart rate and respiratory rate, respectively. Urine is collected by gently rubbing the escutcheon, the area from the base of the udder to the ventral commissure of the vulva, in an upward motion. No other part of the cow should be touched while trying to collect urine as she may resist the urge to urinate. A mid-stream sample should be collected in a clean container to allow specific gravity analysis at a later time. A laboratory multistick can be used to evaluate the pH, ketone, and protein level among other components in the urine immediately. Following urine collection and evaluation, a thermometer can be inserted in the rectum while simultaneously ausculting the left paralumbar fossa to evaluate rumen motility, the rumen should be ausculted for two minutes and the number of primary and secondary contractions recorded. The temperature can now be read and recorded. Following the auscultation of the rumen, the direct heart rate and rhythm are taken over the left cranio-ventral thorax behind the left elbow (intercostal spaces 3-5) and heart sounds are evaluated for the presence of murmurs. The respiratory system is next evaluated on the left side of the thorax. Record the respiratory rate by counting chest excursions. Auscult the lung fields to determine if abnormal sounds, crackles and wheezes, are present indicating possible lung pathology. The dorsum is palpated to determine if subcutaneous emphysema is present over the thorax indicating possible ruptured bullae secondary to severe acute pneumonia. Acoustic percussion is next performed over the left lung fields to isolate the cardiac silhouette. Determine if the fluid density line in the thorax is elevated indicating possible intrathoracic effusion or cardiomegaly. The prescapular, prefemoral and supramammary lymph nodes are next palpated to evaluate size and consistency. The jugular vein is observed and palpated to determine if jugular distension or pulses are present. The milk vein (caudal superficial epigastric vein) is palpated to qualitatively assess systemic venous hypertension and possible hematomas, seromas, or abscesses. The entire left side of the abdomen is simultaneously auscultated and percussed to determine if any “pings” consistent with gas in an intra-abdominal organ is present. Pings in the left abdomen can be indicative of a left displaced abomasum, a rumen gas cap, pneumoperitoneum, or physometra depending on location, size, and pitch. The limbs are observed for any abnormal position or swellings. The left front and rear quarters of the udder are palpated for swelling, heat or cold, and edema. The examiner then walks behind the cow to the right side to continue the examination. The right atrio-ventricular valve is evaluated behind the right elbow (intercostal spaces 35) for abnormal heart sounds and the presence of murmurs. The respiratory system is next evaluated on the right side of the thorax. Auscult the lung fields to determine if abnormal sounds, crackles and wheezes, are present indicating possible lung pathology. The dorsum is palpated to determine if subcutaneous emphysema is present over the thorax indicating possible ruptured bullae secondary to severe acute pneumonia. Acoustic percussion is next performed over the right lung fields to isolate the cardiac silhouette. Determine if the fluid density line in the thorax is elevated indicating possible intrathoracic effusion or cardiomegaly. The right prescapular and prefemoral lymph nodes are next palpated to evaluate size and consistency. The jugular vein is observed and palpated to determine if jugular distension or pulses are present. The right milk vein (caudal superficial epigastric vein) is palpated to qualitatively assess systemic venous hypertension and possible hematomas, seromas, or abscesses. The entire right side of the abdomen is simultaneously auscultated and percussed to determine if any “pings” consistent with gas in an intra-abdominal organ is present. Pings in the right abdomen can be indicative of intestinal gas secondary to ileus, right displaced abomasum, abomasal torsion or volvulus, cecal torsion, intestinal obstruction, pneumoperitoneum, or physometra depending on location, size, and pitch of the “ping”. The limbs are observed for any abnormal position or swellings. The right front and rear quarters of the udder are palpated for swelling, heat or cold, and edema. On lactating cows the mammary secretion is next evaluated. Milk is collected from each quarter in a California Mastitis Test (CMT) paddle to evaluate the milk for the presence of clinical mastitis or an increase in the relative concentration of somatic cells. The milk should be uniform white in color between the quarters and not have flakes, or clots. The milk should not be serous or bloody in appearance. The paddle is slowly tilted to about 45 degrees off the horizontal to leave only about two milliliters of milk from each quarter in the corresponding quadrant of the paddle. An equal amount of CMT reagent is added to the milk and the mixture is swirled in the paddle. The CMT reagent will lyse the cytoplasmic and nuclear membranes of the somatic cells and coagulate the nuclear DNA. The paddle is then rocked back and forth and swirled to assess the presence of coagulation of the nuclear material in the mixture. A qualitative scale ranging from negative to trace, 1, 2, or 3 is then assigned to the mixture for each quarter and recorded. As the relative concentration of somatic cells increases, the concentration of nuclear material present in the mixture increases resulting in increased coagulation of the mixture. If a positive CMT reaction occurs, composite of quarter milk cultures may be obtained for microbiologic evaluation. Following evaluation of the mammary gland a rectal examination is performed. The rectal examination should only be performed with shortly trimmed fingernails. Additionally, all rings and watches should be removed to prevent rectal lacerations and irritation. An obstetric sleeve is used with sufficient lubrication to prevent mucosal irritation. In most situations individual sleeves are used for each cow to prevent disease transmission between cows. A routine rectal examination should include the reproductive tract, the intra-abdominal urinary tract, and the digestive system. The manure should be evaluated for consistency, volume, odor, and appearance. If the rectal examination identifies a reproductive problem in a female, a vaginal examination is indicated next. The tail is tied to the animal’s neck or opposite forelimb to hold it away from the vulva. A vaginal prep is performed with an appropriate cleansing of disinfecting solution to prevent contamination of the reproductive tract during the vaginal examination. A new sterile sleeve is worn with appropriate sterile lubricant to perform the vaginal examination. The entire vaginal vault is examined for lacerations or draining tracts associated with a dystocia. The cervix is examined to evaluate the diameter of the external cervical os and whether the cervical rings are open, closed, or intact. The cervix is also examined to determine if fetal membranes are still present in a post-partum cow or if a cervical mucus plug remains in a pre-partum cow. With the conclusion of the rectal or vaginal examination the head of the animal is examined. The examiner proceeds quietly to the front of the chute, headgate, or haltered animal and begins to evaluate the cranial region. Initially, the eyes and nares are evaluated for the presence and character of discharge or redness indicating a potential viral or bacterial infection. The position of the globe in the orbit is recorded to assess enophthalmus or exophthalmus. Enophthalmus can result from rapid fluid or body fat loss, while exophthalmus usually indicates the presence of a retrobulbar mass. The corneas are examined with a penlight to evaluate the direct and indirect pupillary light responses and the presence of corneal, lenticular, or anterior chamber opacities. The upper lid is retracted to evaluate the presence of injection of scleral blood vessels resulting from potential systemic inflammation. The upper and lower lids are pinched to assess skin turgidity indicating the degree of dehydration present. If the presenting complaint includes the inability to prehend or swallow feed or water, care should be exercised while performing an examination of the head and neck to prevent exposure to a potentially rabid animal. The oral cavity is examined next to evaluate the dentition, the presence of any vesicles, erosions or ulcers, or blunted oral papillae. The age of the animal based on dentition should be double checked against the records for completeness. The presence of malodorous breath or the odors of acetone or ammonia should be recorded. A cursory neurological examination of the cranial nerves should be carried out to assess appropriate responses to stimuli. Any ear droop, facial asymmetry, or drooling should be noted in the record. Aging Cattle by Dentition: Cattle are aged by dentition to estimate the age for regulatory purposes. Correct estimation of aging requires practice. Procedure: The animal is restrained in a headgate and the head is manually restrained. The mouth is opened to expose the incisors. A general rule of thumb in the estimation of age is that the permanent incisor pairs erupt at 1 ½, 2 ½, 3 ½, and 4 years of age. The permanent incisor pairs are in wear at 5, 6, 7, and 8 years of age. Assignment: Estimate and record the age of five cows. Aging Charts from USDA Guide for Accredited Veterinarians: Station 2: Treatment Techniques Balling Gun: The balling gun is used to administer oral boluses. The cow is restrained in a headgate. A bolus is placed in the balling gun. The balling gun must be held with the bolus up to prevent dropping the bolus prior to administration. The veterinarian approaches the cow from the right side with your back to the headgate. The left leg is extended in front of the headgate opening below the cow’s head. This prevents the cow from lowering her head. With the left arm, reach over the front of the cow’s face and grasp the maxilla with the fingers inserted into the interdental space. The head is pulled toward the veterinarian to contain movement. Gentle pressure is applied to the palate which causes the mouth to open. With the right hand, the balling gun is inserted into the mouth and directed along the midline with firm gentle pressure. Care should be exercised to maintain the grasp on the balling gun to prevent accidental swallowing. The balling gun is advanced until the “finger rings” are at the commissure of the mouth. The plunger is gently depressed to eject the bolus from the gun. Excessive force should be avoided to prevent retropharyngeal placement of the bolus resulting in dysphagia and abscessation. Oro-ruminal Intubation: Oro-ruminal intubation is used to relieve bloat (ruminal tympany) and to administer liquid directly into the rumen. The cow is restrained in a headgate. A Frick speculum is used to allow passage of the tube passed the dental arcades without damaging the tube. The veterinarian approaches the cow from the right side with your back to the headgate. The left leg is extended in front of the headgate opening below the cow’s head. This prevents the cow from lowering her head. With the left arm, reach over the front of the cow’s face and grasp the maxilla with the fingers inserted into the interdental space. The head is pulled toward the veterinarian to contain movement. Gentle pressure is applied to the palate which causes the mouth to open. With the right hand, the Frick tube is inserted into the mouth and directed along the midline with firm gentle pressure. Care should be exercised to maintain the grasp on the Frick tube to prevent accidental swallowing. The Frick tube is advanced until the distal end has passed the level of the dorsal bulb of the tongue. A stomach tube is lubricated and passed through the Frick tube with firm gentle pressure. Observation of the cervical region may reveal passage of the tube indicating the tube has entered the esophagus. If the cow coughs during passage of the stomach tube, the tube should be withdrawn and re-inserted, once the tube is passed to the level of the rumen air is forced into the tube while an assistant is ausculting the left paralumbar fossa for the presence of bubbles of air in the rumen. Fluids may then be administered with a stomach pump. Upon completion of the fluid administration, the tube is once again cleared of liquid by blowing on the tube, the tube is kinked to prevent siphoning of fluid back into the tube, the tube is withdrawn prior to removal of the Frick speculum. The cow can then be released. Venipuncture (Median Sacral Vein): Tail Bleeding in cattle is performed as part of the annual herd test for Brucellosis certification, diagnostic serology for numerous bovine diseases (BVD, BLV, Neosporosis, Leptospirosis), and diagnostic sampling for clinical pathology on individual animals. Injection Techniques (following Dairy Quality Assurance and Beef Quality Assurance Guidelines) Subcutaneous injection: Subcutaneous injections are used in the administration of medications that are labeled for subcutaneous use. The injection is administered under the skin where the skin can be easily elevated from the underlying tissues. The usual site for subcutaneous injection is the lateral aspect of the neck in the cervical region. The only other approved locations for subcutaneous injections are the lateral tailhead region near the ischiorectal fossa in the administration of recombinant bovine somatotropin and ceftiofur crystalline free acid in dairy cattle. The base of the ear may also be used for the administration of ceftiofur crystalline free acid in beef cattle. The needle is inserted through the skin at an acute angle and the medication is injected into the potential space between the skin and the fascia covering the underlying tissue. Following insertion of the needle, negative pressure should be applied to the syringe, aspiration, to insure the tip of the needle is not located in a blood vessel. The needle may also be moved under the skin, in a radial fashion, to insure the needle has not penetrated the underlying fascia. Irritating or hypertonic solutions should not be administered with this technique. Typically, when medications are labeled for intramuscular or subcutaneous route, the subcutaneous route is chosen to decrease the irritation to the muscle. Many clinicians state that if a medication can be used subcutaneously then it should be used subcutaneously. Intramuscular injection: Intramuscular injections are utilized in the administration of medications labeled for intramuscular use. The medications are usually more irritating than medications labeled for subcutaneous use. In some cases the absorption of the medication from the intramuscular route is sufficiently slower than by the subcutaneous or intravenous route to be beneficial in the treatment of the condition of interest. When administering medications via the intramuscular route the medication should injected into a large muscle belly. Volume of medication administered at each injection site should be limited to 5 to 10 cc. The approved site of intramuscular injection in cattle is the lateral aspect of the cervical region in the epaxial musculature. Care must be exercised to avoid injection into the nuchal ligament which would severely decrease the rate of absorption. Following insertion of the needle, negative pressure should be applied to the syringe, aspiration, to insure the tip of the needle is not located in a blood vessel. Intravenous injection/solution administration: The intravenous route of administration is used when isotonic fluids are administered to treat or prevent dehydration and when medications labeled for intravenous use are administered. While large volumes of intravenous fluids can, in an emergency, be administered through a needle inserted into a vein, usually, a catheter is employed to maintain venous access and decrease the possibility of administering the fluids in the perivascular tissues. Catheter placement will be covered in VMED 589, Small Animal Surgery Fundamentals. The technique for administering intravenous injections is similar to the technique for venipuncture. The preferred site for intravenous injection is the jugular vein. Jugular Intravenous Administration: The cow is restrained in a headgate or held by an assistant. The cow is haltered following the guidelines presented in the restraint of cattle lab manual. The handler should turn the head tightly to the side to expose the jugular furrow. For a right handed phlebotomist: Place the left hand with fingers extended in the jugular furrow near the thoracic inlet. Apply pressure to occlude the jugular vein. Watch for the jugular vein to distend. Insert the needle directed ventrally, bevel up, through the skin into the vein. Advance the needle within the vein until the needle is completely threaded within the vein. Attach a syringe or bottle of medication and simplex onto the threaded needle. Aspirate the syringe to determine if the end of the needle is in the vein. If blood does not flow into the syringe, withdraw or advance the needle slowly, making sure to not to remove the needle from the skin. When a good flow of blood flows into the syringe during aspiration, release the occlusive pressure of the left hand and slowly inject the medication, or allow the medication to flow through a simplex periodically occluding the jugular and aspirating or lowering the bottle to a level below the venipuncture site to insure the needle remains in the lumen of the vein. To prevent catastrophic adverse reactions to Calcium containing solutions, these solutions should be administered slowly. If using a simplex to administer Calcium salts, the bottle should never be elevated above the cow’s shoulder and the heart rate should be constantly monitored. Following administration of the medication, withdraw the needle from the vein and skin and apply pressure to the venipuncture site until bleeding ceases to prevent hematoma formation, usually about a minute. Intradermal injection: Intradermal injections are most commonly utilized in bovine practice during Tuberculosis testing. A 0.1 ml volume of tuberculin is drawn up in a small syringe and a 25-27 gauge needle is used to inject the tuberculin within the dermal layer of the skin in the caudal fold of the tail. A small bump should appear within the skin upon injection. The caudal fold is inspected for swelling and the results recorded in 72 ± 6 hours. The presence of swelling, an indication of antibodies to Mycobacterium bovis, the causative agent of bovine Tuberculosis, requires the animal be identified as a TB suspect. If swelling appears the animal is quarantined and the Federal Veterinarian is called. A comparative cervical Tuberculin test is then performed by the Federal Veterinarian. The comparative cervical TB test is also a series of intradermal injections of antigens of M. bovis, and M. avium. The comparative cervical TB test can only be administered by a Federal Veterinarian. Subconjuntival injection: Subconjunctival injections are administered in the treatment of Infectious Bovine Keratoconjunctivitis (IBK). IBK is also known as pinkeye. To administer the subconjunctival injection approximately 1 ml of aqueous Procaine Penicillin G is drawn up in a 3 cc syringe with a 22 gauge needle. The head of the animal is restrained in a headgate and a halter is applied with the head extended laterally and tied tightly to the chute in a quick release manner with the affected eye exposed. The superior lid is retracted digitally and the bulbar conjunctiva is identified. The 22 gauge needle is inserted through the bulbar conjunctiva and 1 ml of the antibiotic is injected into the potential space bordered superficially by the bulbar conjunctiva and deeply by the sclera of the globe. The resulting “bleb” of penicillin is then slowly expressed with each blink of the eye resulting in a nearly constant bathing of the cornea with antibiotic suspension.