Acid/Base Titrations
advertisement

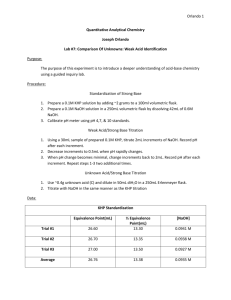
Acid/Base Titrations Titrations involving NaOH, KHP, Na2CO3 and weak acids (HA) H+1 + OH-1 H2O Vocabulary1) Aliquot 2) Back Titrate 3) Buret 4) End-Point 5) Equivalence point 6) Erlenmeyer Flask 7) Indicator 8) KHP 9) Meniscus 10) Parallax 11) Phenolphthalein 12) Pipet 13) Primary Standard 14) Standardization 15) TC 16) TD 17) Titer 18) Titration 19) Volumetric Flask Procedurea) Prepare 1000 mL of approximate 0.25 N HCl solution by adding 21.5 mL of 12 MHCl to 1000 mL of De-ionized water. Store in a clean glass bottle. b) Prepare 1000 mL of approximate 0.25 N NaOH solution by adding approximately 10 grams of solid NaOH pellets to 1000 mL of boiled and cooled De-ionized water. Store in a clean polyethylene bottle. c) Remember the magic number three. i. Always rinse rinse titration burets three times with 2 to 5 mL of the solution that they will contain. ii. Always rinse titration flask, Erlenmeyer Flask three with DI water. iii. Always add three drops of indicator. iv. Always do three good trials. d) Determine the Acid/Base ration by titrating approximately 20.00 mL of the HCl with the NaOH. i. Be sure to read and record the initial and final buret readings and report your answer to the nearest .01 mL. ii. Don’t forget to add the indicator. Use either methyl orange or phenolphthalein. iii. Be sure to rinse the sides of the E-Flask down with DI water just before the endpoint. e) Determination of a titer (You use the data from the first trial to speed up the process.) Remember, you should always use different amount in each trial. Titer mL Base needed Trial x = mL NaOH mL HCl X Trial #1 ml HCl Trial x Trial #1 Titer mL acid needed Trial x =(mL acid used mg Na2CO3 Trial #1 )(mg Na2CO3 Trial x) Trial #1 Titer mL base needed Trial x =(mL base used Trial #1)(mg KHP mg KHP Trial #1 Trial x) f) Standardize the HCl by titrating a sample of pure sodium carbonate ( a primary standard). Measure accurately between 0.4 to 0.5 grams of Na2CO3. Dissolve in 50 mL of DI water and add 3 drops of bromocresol green indicator solution. Fill both the acid and base buret. Titrate to a yellow color, then heat to drive off the CO2 formed the carbonate. If the indicator goes back to its original blue color finish titrating with acid to the faintest of yellow. If too much acid has been added back titrate with the NaOH. Do three trials. g) Or standardize the NaOH with KHP. Measure accurately 1.0 to 1.2 grams of KHP. Dissolve in 50 mL of DI water and add 3 drops of phenolphthalein indicator. Titrate to the faintest of pink endpoint. If necessary you can back titrate with the acid. Do three trials. Errors-The biggest error problem is the carbon dioxide dissolve in the water used to make the NaOH solution. Calculations- mL X N = meq = mg/GEM a) Acid Base Ratio Ratio = mlHCL/mLNaOH b) Standardization of HCl Normal-titration NHCl = (mgNa CO /GEMNa CO )(1/ mLHCl) 2 3 2 3 Back-titration meqacid - meqbase = (mgNa CO /GEMNa CO ) 2 3 2 3 (mLacid) (Nacid) - (mLbase) (Nbase) = (mgNa2CO3/GEMNa2CO3) (mLacid) (Nacid) = (mLbase) (Nbase) (Nbase) = (Nacid)(mLacid/mLbase) Titer (Nacid)(mLacid) - (mLbase) [(Nacid)(mLacid/mLbase)] = (mgNa2CO3/GEMNa2CO3) (Nacid) [(mLacid) - (mLbase) (mLacid/mLbase)] = (mgNa2CO3/GEMNa2CO3) (Nacid) = (mgNa2CO3/GEMNa2CO3) (1/[(mLacid) - (mLbase) (mLacid/mLbase)] ) c) Standardization of NaOH Normal titration NNaOH = (mgKHP/GEMKHP)(1/ mLNaOH) Back-titration meqbase - meqacid = (mgKHP/GEMKHP) (mLbase) (Nbase) - (mLacid) (Nacid) = (mgKHP/GEMKHP) (mLacid) (Nacid) = (mLbase) (Nbase) (Nacid) = (Nbase)(mLbase/mLacid) Titer (Nbase) (mLbase) - (mLacid) [(Nbase)(mLbase/mLacid)] = (mgKHP/GEMKHP) (Nbase) [ (mLbase) - (mLacid) (mLbase/mLacid)] = (mgKHP/GEMKHP) (Nbase) = (mgKHP/GEMKHP) (1/[(mLbase) - (mLacid) (mLbase/mLacid)] )