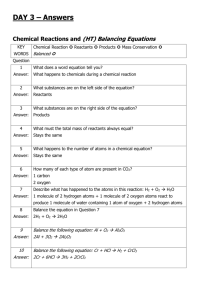
Chapter 2 Science
advertisement

Name _________________ Date_________ Chapter 2, Lesson 1 What are physical and chemical changes? ____________ before and after being carved. Physical Changes: Examples of Physical Changes: raindrops become _______________, a potato gets __________________, a piece of wood is _______________, ice cream that ___________ onto the ground, salt crystals ______________ into water. __________________ becomes _________________ when cooked. Chemical Changes: For example, pancake batter when cooked ________________, ____________, ______________, and _______________ differently. When a _____________ _______________ occurs, _______________ rearrange themselves to form ___________________ kinds of matter. A ____________ marshmallow shows evidence of a chemical change. Evidence of Chemical Changes: ___________________ is another chemical change. When a marshmallow burns is produces three new substances: ________, __________________________________, and ____________________________. Name _________________ Date_________ Chapter 2, Lesson 1 What are physical and chemical changes? Gemstones before and after being carved. Physical Changes: changes in size, shape, volume, and state of matter. Examples of Physical Changes: raindrops become frozen, a potato gets grated, a piece of wood is sawed in half, ice cream that drips onto the ground, salt crystals mixed into water. Pancake batter becomes pancakes when Chemical Changes: occur when one kind of matter changes into a different kind cooked. of matter with different properties. For example, pancake batter when cooked looks, tastes, feels and smells differently. When a chemical change occurs, atoms rearrange themselves to form different kinds of matter. A burning marshmallow shows evidence of a chemical change. Evidence of Chemical Changes: Chemical changes often produce a gas or a solid. Burning is another chemical change. When a marshmallow burns is produces three new substances: ash, carbon dioxide, and water vapor. Chapter 2, lesson 2 How does matter change state? An object is a ______________, ________________, or ____________ because of the _______________ and positions of its ____________ or _______________________. The three forms of water are called ______________ or states of matter. The state of a material at room temperature is a __________________ property. Compared to solids and liquids, particles of a __________ are _____________________________. As solids melt, their particles __________________. Freezing and Melting: The _________________ point is when a ___________ turns into a ___________. While the _____________________________ is when a _____________ turns into a _________________. Evaporation: The _____________________ is a physical property of a liquid. Condensation: Often this occurs when gas particles touch a ___________ surface. Sublimation: carbon dioxide in its solid form is known as _______________. Chapter 2, lesson 2 How does matter change state? An object is a solid, liquid , or gas because of the motions and positions of its atoms or molecules. The three forms of water are called phases or states of matter. The state of a material at room temperature is a physical property. Compared to solids and liquids, particles of a gas are very far apart. As solids melt, their particles gain energy. Freezing and Melting: Particles slow down and vibrate in place as liquids get colder and freeze. The freezing point is when a liquid turns into a solid. While the melting point is when a solid turns into a liquid. (This is a physical property) Evaporation: particles leave a liquid and become a gas. The boiling point is a physical property of a liquid. Condensation: a gas turns into a liquid. Often this occurs when gas particles touch a cold surface. Sublimation: solids change directly to gases – carbon dioxide in its solid form is known as dry ice. Chapter 2, Lesson 3 What are some kinds of chemical reactions? Water is the _______________, and _________________ changes occur during oxygen is the ______________. chemical _________________, and substances _____________ into other substances. Reactant: Product: Chemical equation: Matter cannot be _____________ or _____________ during chemical reactions. ___________________ breaks down in a ____________________reaction (water and oxygen gas) Three types of reactions: 1. Decomposition reaction: 2. Combination reaction: 3. Replacement reaction: During a chemical reaction, ____________ in the ________________ rearrange to form products that have _______________________ properties. No original atoms are __________ and no new atoms are _______________. The atoms simply __________________ in new ways to form new ____________________. __________ forms from a combination reaction. Chapter 2, Lesson 3 What are some kinds of chemical reactions? Water is the reactant, and oxygen is the Chemical changes occur during chemical product. reactions, and substances change into other substances. Reactant: the substance used in the reaction. Product: the substance made by the reaction. Chemical equation: shows what happened during a chemical reaction. (like a math problem with arrows) Hydrogen Peroxide breaks down in a decomposition reaction (water and oxygen gas) Matter cannot be created or destroyed during chemical reactions. Three types of reactions: 1. Decomposition reaction: compounds split apart to form smaller compounds or elements. Ex. AB = A + B 2. Combination reaction: elements or compounds come together to form new compounds. Ex. A + B = AB 3. Replacement reaction: two or more compounds split apart. The parts switch places. Ex. AB + CD = AD + CB During a chemical reaction, atoms in the reactants rearrange to form products that have different properties. No original atoms are lost and no new atoms are added. The atoms simply combine in new ways to form new substances. Rust forms from a combination reaction. Chapter 2, Lesson 4 How are chemical properties used? ________________ and ______________ properties may be used to ________________ mixtures and help __________________ materials. Procedure Place a hard boiled egg in each of two mason jars. Fill one of the jars with water and the other with vinegar. Next. place a well-washed chicken bone in each of the two mason jars. Fill one of the jars with water and the other with vinegar. Now, place a seashell in each of two mason jars. Fill one of the jars with water and the other with vinegar. Now wait about 4 days. The objects which have been sitting in vinegar have changed. The objects which have been sitting in water are the same. The bone will be bendy, the seashell will be squishy, and the egg shell will be gone making for a bouncy egg. What's Going On? Calcium carbonate is in egg shells, bones, and seashells. When calcium carbonate comes into contact with vinegar, which contains acetic acid, a chemical reaction occurs. Simply put, the calcium carbonate breaks down into a "calcium part" and a "carbonate part". The "calcium part" is a calcium salt that dissolves or precipitates and the "carbonate part" is a carbon dioxide which bubbles away. Water is also a product of the reaction. One might say the acetic acid dissolves the calcium from the bone, egg, and seashell. Chemical properties may be used to separate __________ from rocks with the use of __________________ which breaks down limestone. Separating Metals from Ores: Separating Solutions: ______________ and ________________ are two types of substances. Example of an acid __________ and a base ________________. The stronger the acid or base the greater the ______________. Acids and bases can be distinguished from each other by their different properties. For example, acids taste sour and turn blue litmus red. In contrast, bases are bitter and turn red litmus blue. Universal indicator paper: Chapter 2, Lesson 4 How are chemical properties used? Chemical and physical properties may be used to separate mixtures and help identify materials. Procedure Place a hard boiled egg in each of two mason jars. Fill one of the jars with water and the other with vinegar. Next. place a well-washed chicken bone in each of the two mason jars. Fill one of the jars with water and the other with vinegar. Now, place a seashell in each of two mason jars. Fill one of the jars with water and the other with vinegar. Now wait about 4 days. The objects which have been sitting in vinegar have changed. The objects which have been sitting in water are the same. The bone will be bendy, the seashell will be squishy, and the egg shell will be gone making for a bouncy egg. What's Going On? Calcium carbonate is in egg shells, bones, and seashells. When calcium carbonate comes into contact with vinegar, which contains acetic acid, a chemical reaction occurs. Simply put, the calcium carbonate breaks down into a "calcium part" and a "carbonate part". The "calcium part" is a calcium salt that dissolves or precipitates and the "carbonate part" is a carbon dioxide which bubbles away. Water is also a product of the reaction. One might say the acetic acid dissolves the calcium from the bone, egg, and seashell. Chemical properties may be used to separate fossils from rocks with the use of vinegar which breaks down limestone. Separating Metals from Ores: Heating iron ore allows the iron to separate from the oxygen in the ore. Separating Solutions: A reaction between two substances can be used to separate the original substance ex. Lead iodide Acids and bases are two types of substances. Example of an acid vinegar and a base soap. The stronger the acid or base the greater the reaction. Acids and bases can be distinguished from each other by their different properties. For example, acids taste sour and turn blue litmus red. In contrast, bases are bitter and turn red litmus blue. Universal indicator paper: This helps to identify a substance. Acids will turn indicator paper red, and bases turn it purple.