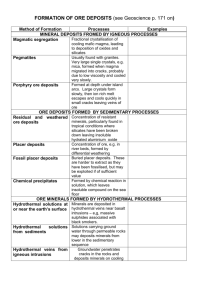
Hydrothermal mineral deposits are those in which hot water serves
advertisement

Hydrothermal mineral deposits are those in which hot water serves as a concentrating, transporting, and depositing agent. They are the most numerous of all classes of deposit. Hydrothermal deposits are never formed from pure water, because pure water is a poor solvent of most ore minerals. Rather, they are formed by hot brines, making it more appropriate to refer to them as products of hydrothermal solutions. Brines, and especially sodium-calcium chloride brines, are effective solvents of many sulfide and oxide ore minerals, and they are even capable of dissolving and transporting native metals such as gold and silver. The water in a hydrothermal solution can come from any of several sources. It may be released by a crystallizing magma; it can be expelled from a mass of rock undergoing metamorphism; or it may originate at the Earth's surface as rainwater or seawater and then trickle down to great depths through fractures and porous rocks, where it will be heated, react with adjacent rocks, and become a hydrothermal solution. Regardless of the origin and initial composition of the water, the final compositions of all hydrothermal solutions tend to converge, owing to reactions between solutions and the rocks they encounter. Hydrothermal solutions are sodium-calcium chloride brines with additions of magnesium and potassium salts, plus small amounts of many other chemical elements. The solutions range in concentration from a few percent to as much as 50 percent dissolved solids by weight. Existing hydrothermal solutions can be studied at hot springs, in subsurface brine reservoirs such as those in the Imperial Valley of California or the Cheleken Peninsula on the eastern edge of the Caspian Sea in Turkmenistan, and in oil-field brines. Fossil hydrothermal solutions can be studied in fluid inclusions, which are tiny samples of solution trapped in crystal imperfections by a growing mineral. Because hydrothermal solutions form as a result of many processes, they are quite common within the Earth's crust. Hydrothermal mineral deposits, on the other hand, are neither common nor very large compared to other geologic features. It is apparent from this that most solutions eventually mix in with the rest of the hydrosphere and leave few obvious traces of their former presence. Those solutions that do form mineral deposits (and thereby leave obvious evidence of their former presence) do so because some process causes them to deposit their dissolved loads in a restricted space or small volume of porous rock. It is most convenient, therefore, to discuss hydrothermal mineral deposits in the context of their settings. Veins The simplest hydrothermal deposit to visualize is a vein, which forms when a hydrothermal solution flows through an open fissure and deposits its dissolved load. A great many veins occur close to bodies of intrusive igneous rocks because the igneous rocks serve as heat sources that create convectively driven flows in hydrothermal solutions. Precipitation of the minerals is usually caused by cooling of the hydrothermal solution, by boiling, or by chemical reactions between the solution and rocks lining the fissure. Some famous deposits are the tincopper-lead-zinc veins of Cornwall, Eng.; the gold-quartz veins of Kalgoorlie, W. Aus., Australia, and Kirkland Lake, Ont., Can.; the tin-silver veins of Llallagua and Potosí, Bol.; and the silver-nickel-uranium veins of the Erzgebirge, Ger., which were first described by Georgius Agricola in his book De re metallica (1556). Hydrothermal deposits formed at shallow depths below a boiling hot spring system (see figure) are commonly referred to as epithermal, a term retained from an old system of classifying hydrothermal deposits based on the presumed temperature and depth of deposition. Epithermal veins tend not to have great vertical continuity, but many are exceedingly rich and deserving of the term bonanza. Many of the famous silver and gold deposits of the western United States, such as Comstock in Nevada and Cripple Creek in Colorado, are epithermal bonanzas.